Abstract
Objective
To compare the MRI findings between the localized- and diffuse-type tenosynovial giant cell tumors (TSGCTs) of digits with pathology correlation.
Methods
Twenty-eight patients with newly diagnosed TSGCTs of digits (22 localized and 6 diffuse types) who underwent preoperative MRI and surgical excision were included from Jan. 2015 to September 2021. MRI findings regarding nodularity, margins, morphology of hypointensity with pathology correlation, and disease extent (bone erosion, articular involvement, muscle involvement, tendon destruction, and neurovascular encasement) were assessed.
Results
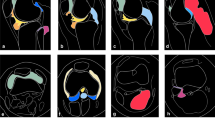
Diffuse type was significantly larger (P = 0.006), more multinodular on both MRI and pathology (P = 0.038, both) with significant agreement, and infiltrative on both MRI and pathology (P < 0.001, both) with substantial agreement, and showed central granular on MRI and strong hemosiderin deposition on pathology (P = 0.022 and P = 0.021) with moderate agreement than localized type. Localized type showed significantly more frequent peripheral capsules on both MRI and pathology (P < 0.001, both) with moderate agreement than diffuse type. However, the septum on both MRI and pathology showed no statistically significant difference between the two groups (P = 0.529 and P = 0.372) without significant agreement. The disease extent was more severe in the diffuse type than the localized type regarding articular involvement (P < 0.001), muscle involvement (P < 0.001), and tendon destruction (P = 0.010). No statistically significant differences were found between the two groups regarding bone erosion (P = 0.196) or neurovascular bundle encasement (P = 0.165).
Conclusions
Diffuse-type TSGCTs of digits presented as locally aggressive lesions with larger, multinodular, infiltrative masses exhibiting stronger hemosiderin deposition and more severe disease extents of articular, muscle, and tendon involvement than the localized type.






Similar content being viewed by others
Abbreviations
- AUC:
-
Areas under the curve
- H&E:
-
Hematoxylin and eosin
- MRI:
-
Magnetic resonance imaging
- PIP:
-
Proximal interphalangeal
- ROC:
-
Receiver operating characteristic
- TSGCT:
-
Tenosynovial giant cell tumor
References
World Health Organization WHO, Fletcher C, Bridge JA, Hogendoorn PCW, Mertens F, editors. WHO classification of tumours of soft tissue and bone: WHO classification of tumours, vol. 5. 4th ed. World Health Organization; 2013. p. 468.
Somerhausen NS, Fletcher CD. Diffuse-type giant cell tumor: clinicopathologic and immunohistochemical analysis of 50 cases with extraarticular disease. Am J Surg Pathol. 2000;24:479–92.
van der Heijden L, Gibbons CL, Dijkstra PD, et al. The management of diffuse-type giant cell tumour (pigmented villonodular synovitis) and giant cell tumour of tendon sheath (nodular tenosynovitis). J Bone Joint Surg Br. 2012;94:882–8.
Gouin F, Noailles T. Localized and diffuse forms of tenosynovial giant cell tumor (formerly giant cell tumor of the tendon sheath and pigmented villonodular synovitis). Orthop Traumatol Surg Res. 2017;103:S91–7.
Rateb K, Hassen BG, Leila A, Faten F, Med SD. Giant cell tumor of soft tissues: a case report of extra-articular diffuse-type giant cell tumor of the quadriceps. Int J Surg Case Rep. 2017;31:245–9.
Verspoor FG, van der Geest IC, Vegt E, Veth RP, van der Graaf WT, Schreuder HW. Pigmented villonodular synovitis: current concepts about diagnosis and management. Future Oncol. 2013;9:1515–31.
Al-Qattan MM. Giant cell tumours of tendon sheath: classification and recurrence rate. J Hand Surg Br. 2001;26:72–5.
Ozben H, Coskun T. Giant cell tumor of tendon sheath in the hand: analysis of risk factors for recurrence in 50 cases. BMC Musculoskelet Disord. 2019;20:457.
Serhal A, Samet J, Shah C, Omar I, Youngner J. MRI evaluation of solid soft tissue masses of the fingers with pathology correlation. Eur J Radiol. 2021;135:109465.
Ge Y, Guo G, You Y, et al. Magnetic resonance imaging features of fibromas and giant cell tumors of the tendon sheath: differential diagnosis. Eur Radiol. 2019;29:3441–9.
Ho CY, Maleki Z. Giant cell tumor of tendon sheath: cytomorphologic and radiologic findings in 41 patients. Diagn Cytopathol. 2012;40(Suppl 2):E94–8.
Chiari C, Pirich C, Brannath W, Kotz R, Trieb K. What affects the recurrence and clinical outcome of pigmented villonodular synovitis? Clin Orthop Relat Res. 2006;450:172–8.
Kim DE, Kim JM, Lee BS, Kim NK, Lee SH, Bin SI. Distinct extra-articular invasion patterns of diffuse pigmented villonodular synovitis/tenosynovial giant cell tumor in the knee joints. Knee Surg Sports Traumatol Arthrosc. 2018;26:3508–14.
van der Heijden L, Mastboom MJ, Dijkstra PD, van de Sande MA. Functional outcome and quality of life after the surgical treatment for diffuse-type giant-cell tumour around the knee: a retrospective analysis of 30 patients. Bone Joint J. 2014;96-B(8):1111–8.
Ota T, Nishida Y, Ikuta K, et al. Tumor location and type affect local recurrence and joint damage in tenosynovial giant cell tumor: a multi-center study. Sci Rep. 2021;11:17384.
Murphey MD, Rhee JH, Lewis RB, Fanburg-Smith JC, Flemming DJ, Walker EA. Pigmented villonodular synovitis: radiologic-pathologic correlation. Radiographics. 2008;28:1493–518.
Dundar A, Young JR, Wenger DE, Inwards CY, Broski SM. Unusual manifestations of diffuse-type tenosynovial giant cell tumor in two patients: importance of radiologic-pathologic correlation. Skeletal Radiol. 2020;49:483–9.
Wang C, Song RR, Kuang PD, Wang LH, Zhang MM. Giant cell tumor of the tendon sheath: Magnetic resonance imaging findings in 38 patients. Oncol Lett. 2017;13:4459–62.
Crim J, Dyroff SL, Stensby JD, Evenski A, Layfield LJ. Limited usefulness of classic MR findings in the diagnosis of tenosynovial giant cell tumor. Skeletal Radiol. 2021;50:1585–91.
Mastboom MJL, Verspoor FGM, Hanff DF, et al. Severity classification of Tenosynovial Giant Cell Tumours on MR imaging. Surg Oncol. 2018;27:544–50.
Palmerini E, Staals EL, Maki RG, et al. Tenosynovial giant cell tumour/pigmented villonodular synovitis: outcome of 294 patients before the era of kinase inhibitors. Eur J Cancer. 2015;51:210–7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval and informed consent
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Approval from the Institutional Review Board was obtained and in keeping with the policies for a retrospective review, informed consent was not required.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This manuscript has not previously been published.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jeong, H.S., Lee, S.K., Kim, JY. et al. Tenosynovial giant cell tumors of digits: MRI differentiation between localized types and diffuse types with pathology correlation. Skeletal Radiol 52, 593–603 (2023). https://doi.org/10.1007/s00256-022-04170-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-022-04170-x