Abstract
Background
Current guidelines recommend therapeutic drug monitoring as a critical component of valproic acid (VPA) therapy. Due to high protein binding, the active unbound (free) portion of VPA can be misrepresented by total VPA serum levels in certain clinical scenarios. Monitoring free VPA serum levels may be warranted when assessing the clinical response to VPA therapy.
Objectives
The aims were to conduct a systematic review to identify a therapeutic range for free VPA serum levels; to explore the correlation of free VPA serum levels with clinical toxicity and therapeutic benefit; and to examine predictors of discordance between free and total VPA levels.
Methods
Medline, EMBASE, Cochrane Central Register of Controlled Trials (CENTRAL), PsycINFO, BIOSIS Previews, and Cumulative Index to Nursing and Allied Health Literature (CINAHL) were searched from the time of database inception to June 20, 2021. Randomized controlled trials and observational studies that evaluated any patient receiving VPA with free VPA level monitoring were included.
Results
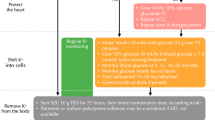
Of 189 citations, we identified 27 relevant studies, which included 14 observational studies, two case series, and 11 case reports. Three studies provided a therapeutic range for free VPA levels between 20 and 410 μmol/L. Two studies suggested the occurrence of hyperammonemia and thrombocytopenia at free VPA serum levels above 60 µmol/L and 103.3 µmol/L, respectively. Two studies suggested an upper limit for neurotoxicity at free VPA serum levels of 70 µmol/L and 207.9 µmol/L. Hypoalbuminemia was identified as a predictor of therapeutic discordance.
Conclusions
This review demonstrates a paucity of data informing the clinical utility of free VPA serum levels. Further high-quality trials are needed to validate an optimal therapeutic range for free VPA levels.

Similar content being viewed by others
References
Methaneethorn J. A systematic review of population pharmacokinetics of valproic acid. Br J Clin Pharmacol. 2018;84:816–34. https://doi.org/10.1111/bcp.13510.
Landmark CJ. Antiepileptic drugs in non-epilepsy disorders: relations between mechanisms of action and clinical efficacy. CNS Drugs. 2008;22:27–47. https://doi.org/10.2165/00023210-200822010-00003.
Loscher W. Basic pharmacology of valproate: a review after 35 years of clinical use for the treatment of epilepsy. CNS Drugs 2002;16:669–94. https://doi.org/10.2165/00023210-200216100-00003.
Ding J, Wang Y, Lin W, Wang C, Zhao L, Li X, et al. A population pharmacokinetic model of valproic acid in pediatric patients with epilepsy: a non-linear pharmacokinetic model based on protein-binding saturation. Clin Pharmacokinet. 2015;54:305–17. https://doi.org/10.1007/s40262-014-0212-8.
Tseng Y-J, Huang S-Y, Kuo C-H, Wang C-Y, Wang K-C, Wu C-C. Factors to influence the accuracy of albumin adjusted free valproic acid concentration. J Formos Med Assoc. 2020. https://doi.org/10.1016/j.jfma.2020.09.004.
Yu-Ju T, Shih-Ying H, Kuo C-H, Chen-Yu W, Kuo-Chuan W, Wu C-C. Safety range of free valproic acid serum concentration in adult patients. PLoS One San Franc 2020;15:e0238201. https://doi.org/10.1371/journal.pone.0238201.
Bellver MJG, Sánchez MJG, Gonzalez ACA, Buelga DS, Dominguez-Gil A. Plasma protein binding kinetics of valproic acid over a broad dosage range: therapeutic implications. J Clin Pharm Ther. 1993;18:191–7. https://doi.org/10.1111/j.1365-2710.1993.tb00612.x.
Free valproic acid assay. CADTHCa 2014. https://www.cadth.ca/free-valproic-acid-assay. Accessed 27 Oct 2020.
Hiemke C, Bergemann N, Clement H, Conca A, Deckert J, Domschke K, et al. Consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology: update 2017. Pharmacopsychiatry. 2018;51:9–62. https://doi.org/10.1055/s-0043-116492.
Kang J-S, Lee M-H. Overview of therapeutic drug monitoring. Korean J Intern Med. 2009;24:1–10. https://doi.org/10.3904/kjim.2009.24.1.1.
Gram L. Experimental studies and controlled clinical testing of valproate and vigabatrin. Acta Neurol Scand. 1988;78:241–70. https://doi.org/10.1111/j.1600-0404.1988.tb03655.x.
Hermida J, Tutor JC. A theoretical method for normalizing total serum valproic acid concentration in hypoalbuminemic patients. J Pharmacol Sci. 2005;97:489–93. https://doi.org/10.1254/jphs.fpe04007x.
Doré M, San Juan AE, Frenette AJ, Williamson D. Clinical importance of monitoring unbound valproic acid concentration in patients with hypoalbuminemia. Pharmacotherapy. 2017;37:900–7. https://doi.org/10.1002/phar.1965.
Nasreddine W, Dirani M, Atweh S, Makki A, Beydoun A. Determinants of free serum valproate concentration: a prospective study in patients on divalproex sodium monotherapy. Seizure. 2018;59:24–7. https://doi.org/10.1016/j.seizure.2018.04.012.
Parent X, Marzullo C. Gutbub AM [Valproic acid: a simple method for the estimation of free serum concentration]. Acide Valproique Estim Simple Conc Serique Libre. 1993;51:649–50.
Drisaldi A, Weeda E, Neyens R, Orvin N, Bonilha L, Campbell Z, et al. Accuracy of valproic acid concentration correction based on serum albumin. Neurocrit Care. 2019;30:301–6. https://doi.org/10.1007/s12028-018-0627-4.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372: n71. https://doi.org/10.1136/bmj.n71.
Chapter 25: Assessing risk of bias in a non-randomized study. https://training.cochrane.org/handbook/current/chapter-25 Accessed 6 Sep 2021.
Chapter 8: Assessing risk of bias in a randomized trial. https://training.cochrane.org/handbook/current/chapter-08. Accessed 6 Sep 2021.
Gagnier JJ, Kienle G, Altman DG, Moher D, Sox H, Riley D, et al. The CARE guidelines: consensus-based clinical case reporting guideline development. Headache. 2013;53:1541–7. https://doi.org/10.1111/head.12246.
Murad MH, Sultan S, Haffar S, Bazerbachi F. Methodological quality and synthesis of case series and case reports. BMJ Evid-Based Med. 2018;23:60–3. https://doi.org/10.1136/bmjebm-2017-110853.
Haidukewych D, Rodin EA. Monitoring free valproic acid in epilepsy patients medicated with coanticonvulsants. Ther Drug Monit. 1982;4:209–12.
Roman E, Ponniah P, Lambert J, Buchanan N. Free sodium valproate monitoring. Br J Clin Pharmacol. 1982;13:452–5. https://doi.org/10.1111/j.1365-2125.1982.tb01403.x.
Yu HY. Clinical implications of serum protein binding in epileptic children during sodium valproate maintenance therapy. Ther Drug Monit. 1984;6:414–23.
Bauer LA, Davis R, Wilensky A, Raisys V, Levy RH. Valproic acid clearance: unbound fraction and diurnal variation in young and elderly adults. Clin Pharmacol Ther. 1985;37:697–700.
Froscher W, Burr W, Penin H, Vohl J, Bulau P, Kreiten K. Free level monitoring of carbamazepine and valproic acid: clinical significance. Clin Neuropharmacol. 1985;8:362–71.
Cramer JA, Mattson RH, Bennett DM, Swick CT. Variable free and total valproic acid concentrations in sole- and multi-drug therapy. Ther Drug Monit. 1986;8:411–5.
Kilpatrick CJ, Bury RW, Fullinfaw RO, Moulds RF. Plasma concentrations of unbound valproate and the management of epilepsy. Aust N Z J Med. 1987;17:574–9.
Koerner M, Yerby M, Friel P, McCormick K. Valproic acid disposition and protein binding in pregnancy. Ther Drug Monit. 1989;11:228–30.
Felix S, Sproule BA, Hardy BG, Naranjo CA. Dose-related pharmacokinetics and pharmacodynamics of valproate in the elderly. J Clin Psychopharmacol. 2003;23:471–8.
Sriboonruang T, Panomvana D, Chamchitchun S, Silpakit O. The impact of dosage of sustained-release formulation on valproate clearance and plasma concentration in psychiatric patients: analysis based on routine therapeutic drug monitoring data. J Clin Psychopharmacol. 2011;31:115–9. https://doi.org/10.1097/JCP.0b013e318203b405.
Itoh H, Suzuki Y, Fujisaki K, Sato Y, Takeyama M. Correlation between plasma ammonia level and serum trough concentration of free valproic acid in patients with epilepsy. Biol Pharm Bull. 2012;35:971–4. https://doi.org/10.1248/bpb.35.971.
Gibbs HG, Zimmerman DE, Shermock KM, Clarke W, Mirski MA, Lewin Ii JJ. Comparison of free fraction serum valproic acid concentrations between inpatients and outpatients. Am J Health Syst Pharm. 2015;72:121–6. https://doi.org/10.2146/ajhp140191.
Gidal BE, Collins DM, Beinlich BR. Apparent valproic acid neurotoxicity in a hypoalbuminemic patient. Ann Pharmacother. 1993;27:32–5. https://doi.org/10.1177/106002809302700107.
Haroldson J, Kramer L, Wolff D, Lake K. Elevated free fractions of valproic acid in a heart transplant patient with hypoalbuminemia. Ann Pharmacother. 2000;34:183–7. https://doi.org/10.1345/aph.19147.
Ginsberg DL. Neurotoxicity with “normal” serum valproic acid levels. Prim Psychiatry. 2005;12:25–6.
Sandson NB, Marcucci C, Bourke DL, Smith-Lamacchia R. An interaction between aspirin and valproate: the relevance of plasma protein displacement drug-drug interactions. Am J Psychiatry. 2006;163:1891–6. https://doi.org/10.1176/appi.ajp.163.11.1891.
Xiong GL, Ferranti J, Leamon MH. Toxic interaction between valproate and oxcarbazepine: a case detected by the free valproate level. J Clin Psychopharmacol. 2008;28:472–3. https://doi.org/10.1097/JCP.0b013e31817ea9e6.
Chan K, Beran RG. Value of therapeutic drug level monitoring and unbound (free) levels. Seizure. 2008;17:572–5. https://doi.org/10.1016/j.seizure.2007.12.007.
de Leon J, Kiesel JL, Fleming MW, Strobl B. Valproic acid toxicity associated with low dose of aspirin and low total valproic acid levels: a case report. J Clin Psychopharmacol. 2009;29:509–11. https://doi.org/10.1097/JCP.0b013e3181b4b07c.
Sleegers MJM, Beutler JJ, Hardon WJ, Berden JHM, Verhave JC, Conemans JMH, et al. Reversible rapidly progressive dementia with parkinsonism induced by valproate in a patient with systemic lupus erythematosus. J Am Geriatr Soc. 2010;58:799–801. https://doi.org/10.1111/j.1532-5415.2010.02795.x.
de Maat MMR, van Leeuwen HJ, Edelbroek PM. High unbound fraction of valproic acid in a hypoalbuminemic critically ill patient on renal replacement therapy. Ann Pharmacother. 2011;45: e18. https://doi.org/10.1345/aph.1P308.
Sparla S, Hogeman P, van Gemert M, Swart E, Malingre M. Valproic acid during pregnancy: Case report of a child with congenital malformations due to fetal valproate syndrome, and a high unbound serum level of valproic acid at birth. Int J Epilepsy. 2017;4:94–7. https://doi.org/10.1016/j.ijep.2016.11.001.
Dautzenberg G, Nederlof M, Beekman A, Egberts T, Heerdink ER. Severe cognitive impairment associated with a high free but therapeutic total concentration of valproic acid due to hypoalbuminemia in an older patient with bipolar disorder. J Clin Psychopharmacol. 2018;38:265–8. https://doi.org/10.1097/JCP.0000000000000872.
Goulden KJ, Dooley JM, Camfield PR, Fraser AD. Clinical valproate toxicity induced by acetylsalicylic acid. Neurology. 1987;37:1392–4.
Hatton C, Riker RR, Gagnon DJ, May T, Seder DB, Fraser GL. Free serum valproate concentration more reliable than total concentration in critically ill patients. Resuscitation. 2016;105:e15–6. https://doi.org/10.1016/j.resuscitation.2016.05.027.
Johannessen Landmark C, Johannessen S, Patsalos P. Therapeutic drug monitoring of antiepileptic drugs: current status and future prospects. Expert Opin Drug Metab Toxicol. 2020;16:1–12. https://doi.org/10.1080/17425255.2020.1724956.
Aldaz A, Ferriols R, Aumente D, Calvo MV, Farre MR, García B, et al. Pharmacokinetic monitoring of antiepileptic drugs. Farmacia Hosp. 2014;35(6):326–39.
Hirschfeld RMA, Bowden CL, Gitlin MJ, Keck PE, Suppes T, Thase ME, et al. Work group on bipolar disorder. Focus. 2010;1:82.
Yatham LN, Kennedy SH, O’Donovan C, Parikh S, MacQueen G, McIntyre R, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) guidelines for the management of patients with bipolar disorder: consensus and controversies. Bipolar Disord. 2005;7:5–69. https://doi.org/10.1111/j.1399-5618.2005.00219.x.
Patsalos PN, Berry DJ, Bourgeois BFD, Cloyd JC, Glauser TA, Johannessen SI, et al. Antiepileptic drugsbest practice guidelines for therapeutic drug monitoring: a position paper by the subcommission on therapeutic drug monitoring, ILAE commission on therapeutic strategies. Epilepsia. 2008;49:1239–76. https://doi.org/10.1111/j.1528-1167.2008.01561.x.
Riker RR, Gagnon DJ, Hatton C, May T, Seder DB, Stokem K, et al. Valproate protein binding is highly variable in ICU patients and not predicted by total serum concentrations: a case series and literature review. Pharmacotherapy. 2017;37:500–8. https://doi.org/10.1002/phar.1912.
Beran RG, Lewis JH, Nolte JL, Westwood AP. Use of total and free anticonvulsant serum levels in clinical practice. Clin Exp Neurol. 1985;21:69–77.
Perucca E, Grimaldi R, Gatti G, Pirracchio S, Crema F, Frigo GM. Pharmacokinetics of valproic acid in the elderly. Br J Clin Pharmacol. 1984;17:665–9.
Mahmoud SH, Zhou XY, Ahmed SN. Managing the patient with epilepsy and renal impairment. Seizure. 2020;76:143–52. https://doi.org/10.1016/j.seizure.2020.02.006.
Ahmed SN, Siddiqi ZA. Antiepileptic drugs and liver disease. Seizure. 2006;15:156–64. https://doi.org/10.1016/j.seizure.2005.12.009.
Javadi S-S, Mahjub R, Taher A, Mohammadi Y, Mehrpooya M. Correlation between measured and calculated free phenytoin serum concentration in neurointensive care patients with hypoalbuminemia. Clin Pharmacol Adv Appl. 2018;10:183–90. https://doi.org/10.2147/CPAA.S186322.
Sheiner L. Clinical pharmacokinetics : the use of plasma concentrations of drugs. Clin Pharmacol Basic Princ Ther; 1978.
Anderson GD, Pak C, Doane KW, Griffy KG, Temkin NR, Wilensky AJ, et al. Revised Winter-Tozer equation for normalized phenytoin concentrations in trauma and elderly patients with hypoalbuminemia. Ann Pharmacother. 1997;31:279–84. https://doi.org/10.1177/106002809703100301.
Kane SP, Bress AP, Tesoro EP. Characterization of unbound phenytoin concentrations in neurointensive care unit patients using a revised Winter-Tozer equation. Ann Pharmacother. 2013;47:628–36. https://doi.org/10.1345/aph.1R651.
Beck DE, Farringer JA, Ravis WR, Robinson CA. Accuracy of three methods for predicting concentrations of free phenytoin. Clin Pharm. 1987;6:888–94.
Cheng W, Kiang TKL, Bring P, Ensom MHH. Predictive performance of the Winter-Tozer and derivative equations for estimating free phenytoin concentration. Can J Hosp Pharm. 2016;69:269–79. https://doi.org/10.4212/cjhp.v69i4.1573.
Kiang TKL, Ensom MHH. A comprehensive review on the predictive performance of the Sheiner-Tozer and derivative equations for the correction of phenytoin concentrations. Ann Pharmacother. 2016;50:311–25. https://doi.org/10.1177/1060028016628166.
Orr JM, Abbott FS, Farrell K, Ferguson S, Sheppard I, Godolphin W. Interaction between valproic acid and aspirin in epileptic children: serum protein binding and metabolic effects. Clin Pharmacol Ther. 1982;31:642–9. https://doi.org/10.1038/clpt.1982.89.
Toma CM, Imre S, Vari CE, Muntean DL, Tero-Vescan A. Ultrafiltration method for plasma protein binding studies and its limitations. Processes. 2021;9(2):382. https://doi.org/10.3390/pr9020382.
Barton P, Austin RP, Fessey RE. 5.14—In vitro models for plasma binding and tissue storage. In: Taylor JB, Triggle DJ, editors. Comprehensive medicinal chemistry II. Elsevier; 2007:321–340. https://doi.org/10.1016/B0-08-045044-X/00129-2.
Seyfinejad B, Ozkan SA, Jouyban A. Recent advances in the determination of unbound concentration and plasma protein binding of drugs: analytical methods. Talanta. 2021;225: 122052. https://doi.org/10.1016/j.talanta.2020.122052.
Barré J, Chamouard JM, Houin G, Tillement JP. Equilibrium dialysis, ultrafiltration, and ultracentrifugation compared for determining the plasma-protein-binding characteristics of valproic acid. Clin Chem. 1985;31(1):60–4.
Price CP. Analytical techniques for therapeutic drug monitoring. Clin Biochem. 1984;17(1):52–6. https://doi.org/10.1016/S0009-9120(84)90510-1.
Perucca E. Free level monitoring of antiepileptic drugs. Clinical usefulness and case studies. Clin Pharmacokinet 1984;9(Suppl 1):71–8.
Acknowledgements
The authors would like to thank Ursula Ellis, reference librarian at the University of British Columbia, for their help with the literature search.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts or financial interest in any product or service mentioned in the article, including grants, medications, employment, gifts, and honoraria. The views expressed in this article are those of the authors and do not reflect those of the Department of Pharmacy or the Faculty of Pharmaceutical Sciences.
Funding
The authors declare no specific funding for this work.
Author contributions
VC provided the concept for this systematic review. KL and VC performed the literature search and data analysis, and drafted the original manuscript. KD, VC, and CA critically reviewed and edited the manuscript.
Availability of data and material
The datasets generated during and/or analyzed during the study are available from the corresponding author on reasonable request.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Code availability
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, K., Cao, V.F.S., Au, C. et al. Clinical Pharmacokinetic Monitoring of Free Valproic Acid Levels: A Systematic Review. Clin Pharmacokinet 61, 1345–1363 (2022). https://doi.org/10.1007/s40262-022-01171-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40262-022-01171-w