Abstract
Purpose
Obstructive sleep apnea (OSA) is related to increased risk of cardiovascular disease. Ferroptosis is a form of programmed cell death characterized by iron overload and plays critical roles in myocardial injury. This study aimed to investigate the role of ferroptosis in intermittent hypoxia (IH)-induced myocardial injury involving endoplasmic reticulum stress (ERS).
Methods
AC16 human cardiomyocytes were exposed to IH or normoxia conditions. Mice were randomly grouped as follows: normal control (NC), IH, ferrostatin-1 + IH (FIH), and N-acetylcysteine + IH (AIH). The mRNA levels of GPX4, xCT, FTH1, and FACL4 in AC16 cells were detected by qRT-PCR. The protein levels of GPX4, xCT, NOX4, ATF4, CHOP, Bcl-2, and Bax in myocardial tissue were detected by Western blot analysis.
Results
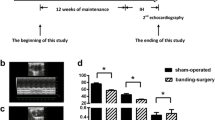
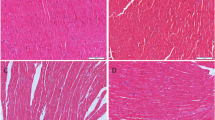
The mRNA expression levels of GPX4 and xCT in AC16 cells were significantly lower in IH group than that of NC group. In IH mice, myocardial tissues were injured accompanied by increased level of ferroptosis and ERS. Inhibition of ferroptosis and treatment of N-acetylcysteine reduced ERS and myocardial injury in mice exposed to IH. In addition, compared to ferrostatin-1, N-acetylcysteine exerted a greater effect in relieving IH-induced myocardial damage and ERS.
Conclusions
Ferroptosis was involved in IH-related myocardial injury accompanied by the activation of ERS. Inhibition of ferroptosis and acetylcysteine treatment alleviated IH-related myocardial injury, which may be a potential target for therapeutic approaches to OSA-induced myocardial injury.




Similar content being viewed by others
Data availability
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
References
Somers VK et al (2008) Sleep apnea and cardiovascular disease: an American Heart Association/american College Of Cardiology Foundation Scientific Statement from the American Heart Association Council for High Blood Pressure Research Professional Education Committee, Council on Clinical Cardiology, Stroke Council, and Council On Cardiovascular Nursing. In collaboration with the National Heart, Lung, and Blood Institute National Center on Sleep Disorders Research (National Institutes of Health). Circulation 118(10):1080–111
Floras JS (2014) Sleep apnea and cardiovascular risk. J Cardiol 63(1):3–8
Lui MMS et al (2018) Untreated obstructive sleep apnea is associated with myocardial injury independent of blood pressure control in hypertension. J Clin Sleep Med 14(11):1841–1847
Almendros I et al (2011) Early and mid-term effects of obstructive apneas in myocardial injury and inflammation. Sleep Med 12(10):1037–1040
Dixon SJ et al (2012) Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149(5):1060–1072
Li W et al (2020) Ferroptosis is involved in diabetes myocardial ischemia/reperfusion injury through endoplasmic reticulum stress. DNA Cell Biol 39(2):210–225
Chen LD et al (2020) The role of ferroptosis in chronic intermittent hypoxia-induced liver injury in rats. Sleep Breath 24(4):1767–1773
Lee YS et al (2018) Ferroptosis-induced endoplasmic reticulum stress: cross-talk between ferroptosis and apoptosis. Mol Cancer Res 16(7):1073–1076
Hong SH et al (2017) Molecular crosstalk between ferroptosis and apoptosis: emerging role of ER stress-induced p53-independent PUMA expression. Oncotarget 8(70):115164–115178
Toffoli S et al (2009) Intermittent hypoxia is an angiogenic inducer for endothelial cells: role of HIF-1. Angiogenesis 12(1):47–67
Baguet JP et al (2005) The severity of oxygen desaturation is predictive of carotid wall thickening and plaque occurrence. Chest 128(5):3407–3412
Drager LF et al (2017) Sleep apnea and cardiovascular disease: lessons from recent trials and need for team science. Circulation 136(19):1840–1850
Zhou X et al (2018) DL-Propargylglycine protects against myocardial injury induced by chronic intermittent hypoxia through inhibition of endoplasmic reticulum stress. Sleep Breath 22(3):853–863
Park MW et al (2021) NOX4 promotes ferroptosis of astrocytes by oxidative stress-induced lipid peroxidation via the impairment of mitochondrial metabolism in Alzheimer’s diseases. Redox Biol 41:101947
Lu B et al (2017) The role of ferroptosis in cancer development and treatment response. Front Pharmacol 8:992
Friedmann Angeli JP et al (2014) Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice. Nat Cell Biol 16(12):1180–1191
Linkermann A et al (2014) Synchronized renal tubular cell death involves ferroptosis. Proc Natl Acad Sci U S A 111(47):16836–16841
Yamada N et al (2020) Iron overload as a risk factor for hepatic ischemia-reperfusion injury in liver transplantation: Potential role of ferroptosis. Am J Transplant 20(6):1606–1618
Chen Y et al (2020) Distinct types of cell death and the implication in diabetic cardiomyopathy. Front Pharmacol 11:42
Dixon SJ et al (2014) Pharmacological inhibition of cystine-glutamate exchange induces endoplasmic reticulum stress and ferroptosis. Elife 3:e02523
Lee YS et al (2019) Ferroptosis-inducing agents enhance TRAIL-induced apoptosis through upregulation of death receptor 5. J Cell Biochem 120(1):928–939
Su N, Kilberg MS (2008) C/EBP homology protein (CHOP) interacts with activating transcription factor 4 (ATF4) and negatively regulates the stress-dependent induction of the asparagine synthetase gene. J Biol Chem 283(50):35106–35117
Polimeno L et al (2013) Augmenter of liver regeneration, a protective factor against ROS-induced oxidative damage in muscle tissue of mitochondrial myopathy affected patients. Int J Biochem Cell Biol 45(11):2410–2419
Funding
The work was supported by the National Natural Science Foundation of China (No. 81900090), Natural Science Foundation of Fujian Province (No. 2020J01987), and Youth Research Fund from Health and Family Planning Commission of Fujian Province (No. 2020QNA053).
Author information
Authors and Affiliations
Contributions
Jiefeng Huang, sequence/data analysis and preparation of the manuscript; Hansheng Xie, study design and preparation of the manuscript; Yisong Yang, PCR and WB; Lida Chen, animal experiments; Biying Wang, manuscript preparation; Gongping Chen, analyzed the data; Qichang Lin, contributed in the experimental design; Jiefeng Huang, Hansheng Xie and Yisong Yang contributed equally to this work. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The study protocols were according to the standard ethical guidelines and approved by Xiamen University Animal Care and Use Committee (XMULAC20201002).
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, J., Xie, H., Yang, Y. et al. The role of ferroptosis and endoplasmic reticulum stress in intermittent hypoxia-induced myocardial injury. Sleep Breath 27, 1005–1011 (2023). https://doi.org/10.1007/s11325-022-02692-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-022-02692-1