Abstract
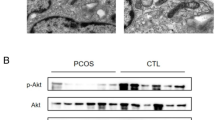
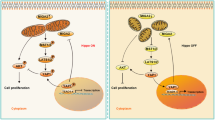
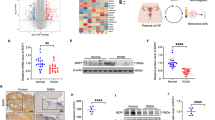
Mitochondrial dysfunction plays a crucial role in the pathological physiology of polycystic ovary syndrome (PCOS). Mitochondrial quality control system is vital to maintaining mitochondrial function, includes mitochondrial biosynthesis, dynamics and mitophagy. While mitophagy as a specific autophagy, plays an important role in the mitochondrial quality control system and is mediated by some signaling pathways to eliminate the excessive production of reactive oxygen species (ROS), such as hypoxia-inducible factor (HIF)-1α/B-cell lymphoma-2 adenovirus E1B 19 kDa interacting protein 3 (BNIP3). Our previous studies have found that excessive production of ROS and the decreased expression of HIF-1α in the ovaries of PCOS rats. Thus, we hypothesized that excessive ROS leads to mitochondrial dysfunction, attenuates HIF-1α/BNIP3-mediated mitophagy in the ovaries of PCOS rats, and further reduces the mitophagic defense. Firstly, the oxidative stress status was detected and found excessive ROS damages ovarian tissue in PCOS rats. Secondly, the marker proteins of mitochondrial biosynthesis/dynamics and amount were examined and found that their expression levels were abnormal, which showed that the abnormal mitochondrial quality control system leads to accumulate the excess or damaged mitochondria in PCOS ovaries. Finally, we detected the HIF-1α/BNIP3 pathway and found HIF-1α-mediated mitophagy is impaired in the ovaries of PCOS rats. Together, these results clearly demonstrated excessive ROS causes mitochondrial dysfunction via the abnormal mitochondrial quality control system, and attenuates HIF-1α/BNIP3-mediated mitophagic defense in the granulosa cells of PCOS rats, which will provide a new direction for further understanding the role of HIF-1α in the molecular mechanism of mitochondrial dysfunction in PCOS ovaries.









Similar content being viewed by others
Data availability
Data that support the findings of this study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
References
Bannigida DM, Nayak BS, Vijayaraghavan R (2020) Insulin resistance and oxidative marker in women with PCOS. Arch Physiol Biochem 126(2):183–186. https://doi.org/10.1080/13813455.2018.1499120
Bhargava D, Wu W, Komrskova K, Maňásková-Postlerová P (2020) Mitochondrial function in modulating human granulosa cell steroidogenesis and female fertility. Int J Mol Sci 21(10):1–20. https://doi.org/10.3390/ijms21103592
Carvalho KF, Machado TS, Garcia BM, Zangirolamo AF, Macabelli CH, Sugiyama FHC, Grejo MP, Neto JDA, Tostes K, Ribeiro FKS, Sarapião FD, Pandey AK, Nociti RP, Tizioto P, Coutinho LL, Meirelles FV, Guimarães FEG, Pernas L, Seneda MM (2020) Mitofusin 1 is required for oocyte growth and communication with follicular somatic cells. FASEB J 34(6):7644–7660. https://doi.org/10.1096/fj.201901761R
Fu Z, Wang Z, Xu L, Chen X, Li X, Liao W, Ma H, Jiang M, Xu T, Xua J, Shen Y, Song B, Gao P, Han W, Zhang W (2020) HIF-1α-BNIP3-mediated mitophagy in tubular cells protects against renal ischemia/reperfusion injury. Rwdox Biol 36:1–16. https://doi.org/10.1016/j.redox.2020.101671
Gao A, Jiang J, Xie F, Chen L (2020) Bnip3 in mitophagy: novel insights and potential therapeutic target for diseases of secondary mitochondrial dysfunction. Clin Chim Acta 506:72–83. https://doi.org/10.1016/j.cca.2020.02.024
González F, Considine RV, Abdelhadi OA, Acton AJ (2019) Oxidative stress in response to saturated fat ingestion is linked to insulin resistance and hyperandrogenism in polycystic ovary syndrome. JCEM 11:5360–5371. https://doi.org/10.1210/jc.2019-00987
He Chuan LuS, Wang X, Wang C, Wang L, Liang S, Luo T, Wang Z, Piao M, Chi G, Ge P (2021) FOXO3a protects glioma cells against temozolomide-induced DNA double strand breaks via promotion of BNIP3-mediated mitophagy. Acta Pharmacol Sin 42(8):1324–1337. https://doi.org/10.1038/s41401-021-00663-y
Ilie IR (2018) Advances in PCOS pathogenesis and progression—mitochondrial mutations and dysfunction. Adv Clin Chem 86:127–155. https://doi.org/10.1016/bs.acc.2018.05.003
KimI R-E, Lemasters JJ (2007) Selective degradation of mitochondria by mitophagy. Arch Biochem Biophys 462(2):245–253. https://doi.org/10.1016/j.abb.2007.03.034
Li C, Zhou J, Liu Z, Zhou J, Yao W, Tao J, Shen M, Liu H (2020) FSH prevents porcine granulosa cells from hypoxia-induced apoptosis via activating mitophagy through the HIF-1α-PINK1-Parkin pathway. FASEB J 34(3):3631–3645. https://doi.org/10.1096/fj.201901808RRR
Li W, Fan R, Xu Q, Chen J, Wu X, Kuang H, Wang Y, Li Y (2021) Mitochondrial dynamics of ovarian cells and polycystic ovary syndrome. Int J Gynecol Obstet 48(4):403–408. https://doi.org/10.12280/gjfckx.20200838
Maqbool M, Dar AM, Gani I, Geer MI (2019) Insulin resistance and polycystic ovary syndrome: a review. J Drug Deliv 9(1):433–436. https://doi.org/10.22270/jddt.v9i1-s.2275
Meo SD, Susanna L, Venditti P (2017) Skeletal muscle insulin resistance: role of mitochondria and other ROS sources. J Endocrinol 233(1):15–42. https://doi.org/10.1530/JOE-16-0598
Robb EL, Christoff CA, Maddalena LA, Stuart JA (2014) Mitochondrial reactive oxygen species (ROS) in animal cells: relevance to aging and normal physiology. Can J Zool 92(7):603–613. https://doi.org/10.1139/cjz-2013-0131
Roca A, Tait SWG (2021) Mitochondrial quality control: from molecule to organelle. Cell Mol Life Sci 78(6):3853–3866. https://doi.org/10.1007/s00018-021-03775-0
Salehi R, Mazier HL, Nivet AL, Reunov AA, Lima P, Wang Q, Fiocco A, Isidoro C, Tsang BK (2020) Ovarian mitochondrial dynamics and cell fate regulation in an androgen-induced rat model of polycystic ovarian syndrome. Sci Rep 10(1):1–13. https://doi.org/10.1038/s41598-020-57672-w
Sedlackova L, Korolchuk VI (2018) Mitochondrial quality control as a key determinant of cell survival. BBA-Mol Cell Res 1866(4):575–587. https://doi.org/10.1016/j.bbamcr.2018.12.012
Shukla P, Mukherjee S (2020) Mitochondrial dysfunction: an emerging link in the pathophysiology of polycystic ovary syndrome. Mitochondrion 52(10):24–39. https://doi.org/10.1016/j.mito.2020.02.006
Tang Z, Xu R, Zhang Z, Shi C, Wang Z (2021a) HIF-1α protects granulosa cells from hypoxia-induced apoptosis during follicular development by inducing autophagy. Front Cell Dev Biol 9:1–12. https://doi.org/10.3389/fcell.2021.631016
Tang Z, Zhang Z, Lin Q, Xu R, Chen J, Wang Y, Zhang Y, Tang Y, Shi C, Liu Y, Yang H, Wang Z (2021b) HIF-1α/BNIP3-mediated autophagy contributes to the luteinization of granulosa cells during the formation of corpus luteum. Front Cell Dev Biol 8:1–19. https://doi.org/10.3389/fcell.2020.619924
Wang F, Zhang Z, Zhang Z, Wang Z, Xiao K, Wang Q, Su J, Wang Z (2015) Expression and clinical significance of the HIF-1a/ET-2 signaling pathway during the development and treatment of polycystic ovary syndrome. J Mol Histol 46(2):173–181. https://doi.org/10.1007/s10735-015-9609-4
Wang F, Wang S, Zhang Z, Lin Q, Liu Y, Xiao Y, Xiao K, Wang Z (2017a) Activation of NLRP3 inflammasome in the ovaries during the development and treatment of polycystic ovary syndrome. Int J Exp Pathol 10(5):5022–5030
Wang F, Zhang Z, Chen J, Wang S, Lin Q, Xiao K, Xiao Y, Wang Z (2017b) Effects of DMBG on Akt/HIF-1a signaling in the ovaries of polycystic ovary syndrome rats. Int J Clin Exp Med 10(3):4435–4442
Wang F, Wang X, Liu Y, Zhang Z (2021a) Effects of exercise-induced ROS on the pathophysiological functions of skeletal muscle. Oxid Med Cell Longev 2021:1–5. https://doi.org/10.1155/2021/3846122
Wang Y, Yang Q, Wang H, Zhu J, Cong L, Li H, Sun Y (2021b) NAD+ deficiency and mitochondrial dysfunction in granulosa cells of women with polycystic ovary syndrome. Biol Reprod 105(2):371–380. https://doi.org/10.1093/biolre/ioab078
Wu H, Chen Q (2015) Hypoxia activation of mitophagy and its role in disease pathogenesis. Antioxid Redox Signal 22(12):1032–1046. https://doi.org/10.1089/ars.2014.6204
Yao R, Ren C, Xia Z, Yao Y (2020) Organelle-specific autophagy in inflammatory diseases: a potential therapeutic target underlying the quality control of multiple organelles. Autophagy 17(2):385–401. https://doi.org/10.1080/15548627.2020.1725377
Yi S, Zheng B, Zhu Y, Cai Y, Sun H, Zhou J (2020) Melatonin ameliorates excessive PINK1/Parkin-mediated mitophagy by enhancing SIRT1 expression in granulosa cells of PCOS. Endocrinol Metab 319(1):91–101. https://doi.org/10.1152/ajpendo.00006.2020
Zhang G, Deng M, Zhang Y, Fan Y (2016) Effect of PGC-1α overexpression or silencing on mitochondrial apoptosis of goat luteinized granulosa cells. J Bioenerg Biomembr 48(5):493–507. https://doi.org/10.1007/s10863-016-9684-6
Zhang J, Bao Y, Zhou X, Zheng L (2019) Polycystic ovary syndrome and mitochondrial dysfunction. Reprod Biol Endocrin 17(1):1–15. https://doi.org/10.1186/s12958-019-0509-4
Zhang Chun Xu, Xiao YL (2016) Research on the expression of apoptosis regulatory proteins Bcl-2, bax in ovarian follicle granulosa cells of improved PCOS rat model. J Tradit Chin Med 6:34–37
Zhou J, Yao W, Li C, Wu W, Li Q, Liu H (2017) Administration of follicle-stimulating hormone induces autophagy via upregulation of HIF-1α in mouse granulosa cells. Cell Death Did 8(8):e3001. https://doi.org/10.1038/cddis.2017.371
Zuo T, Zhu M, Xu W (2015) Roles of oxidative stress in polycystic ovary syndrome and cancers. Oxid Med Cell Longev 2016:1–14. https://doi.org/10.1155/2016/8589318
Funding
This research was funded by the Key Projects of Scientific and Technological Innovation in Fujian Province (Grant Nos. 2021G02021 and 2021G02023), Special Funds of the Central Government Guilding Local Science and Technology Development (2020L3008), Fujian Provincial Natural Science Foundation (2019R1011-3 and 2020J01176), and Scientific Research Talent Project of Fujian Provincial Health Commission (2018-ZQN-20).
Author information
Authors and Affiliations
Contributions
FW and YL: designed the experiment. JH and XW: performed the experiment. FW: wrote the manuscript. FW: analyzed the data. ZZ: revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Ethical approval
All animal procedures performed in this study were approved by the Institutional Animal Care and Use Committee of the College of Fujian Normal University.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, F., Han, J., Wang, X. et al. Roles of HIF-1α/BNIP3 mediated mitophagy in mitochondrial dysfunction of letrozole-induced PCOS rats. J Mol Histol 53, 833–842 (2022). https://doi.org/10.1007/s10735-022-10096-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-022-10096-4