Abstract
The gold standard to measure intra-abdominal pressure (IAP) is intra-vesical measurement via the urinary bladder. However, this technique is restricted in ambulatory settings because of the risk of iatrogenic urinary tract infections. Rectal IAP measurements (IAPrect) may overcome these limitations, but requires validation. This validation study compares the IAPrect technique against gold standard intra-vesical IAP measurements (IAPves). IAPrect using an air-filled balloon catheter and IAPves using Foley Manometer Low Volume were measured simultaneously in sedated and ventilated patients. Measurements were performed twice in different positions (supine and HOB 45° elevated head of bed) and with an external abdominal pressure belt. Sixteen patients were included. Seven were not eligible for analysis due to unreliable IAPrect values. IAPrect was significantly higher than IAPves for all body positions (p < 0.01) and the correlation between IAPves and IAPrect was poor and not significant in each position (p ≥ 0.25, R2 < 0.6, Lin’s CCC < 0.8, bias − 8.1 mmHg and precision of 5.6 mmHg with large limits of agreement between − 19 to 2.9 mmHg, high percentage error 67.3%, and low concordance 86.2%). Repeatability of IAPrect was not reliable (R = 0.539, p = 0.315). For both techniques, measurements with the external abdominal pressure belt were significantly higher compared to those without (p < 0.03). IAPrect has important shortcomings making IAP estimation using a rectal catheter unfeasible because the numbers cannot be trusted nor validated.
Similar content being viewed by others
1 Introduction
Intra-abdominal pressure (IAP) is the steady state pressure inside the abdominal cavity and is normally less than 7 mmHg in healthy adults with higher physiological baseline levels (9 to 14 mmHg) in morbidly obese patients [1, 2]. Critically ill patients usually have baseline IAP of approximately 10 mmHg [3]. Intra-abdominal hypertension (IAH) is defined by the Abdominal Compartment Society (WSACS), formerly known as the World Society of the Abdominal Compartment Syndrome (www.wsacs.org) [4, 5], as a sustained increased in IAP ≥ 12 mmHg, and abdominal compartment syndrome as IAP > 20 mmHg with new onset organ failure [6,7,8]. A pathologically increased IAP is often seen in critically ill patients and this may have detrimental consequences such as acute renal failure, hemodynamic instability, inadequate ventilation and decreased blood flow to organs [9, 10]. It has been suggested that gestational complications such as preeclampsia might also be associated with intra-abdominal hypertension [11,12,13], making IAP an important consideration in obstetric (patho) physiology as well.
Urinary bladder pressure measurement, by using a FoleyManometer Low Volume (FMLV), is recognised as the gold standard to measure IAP [14]. This technique is easily applicable in catheterized patients, but its use is restricted in ambulatory settings because of risks of iatrogenic urinary tract infections. The abdomen behaves according to Pascal’s Law, thus rectal pressure measurements proximal of the pelvic floor muscles should also represent IAP similar to intravesical pressure. Rectal pressures are used routinely as estimates for IAP during urodynamic studies to calculate transmural detrusor muscle pressure (intravesical pressure minus IAP measured rectally) [15,16,17]. From a theoretical point-of-view, measurement using a rectal catheter seems less invasive and could potentially be used in ambulatory settings and in pregnant patients, however, validation of this technique is required. This validation study compares the rectal intra-abdominal pressure (IAPrect) technique against standard intra-vesical IAP measurements (IAPves).
2 Material and methods
2.1 Ethical approval
The study was conducted at the Ziekenhuis Netwerk Antwerpen (ZNA Campus Stuivenberg, Antwerp, Belgium) in accordance with the study protocol, the Declaration of Helsinki and applicable regulatory requirements. The study was approved by the local Institutional Review Board and Ethics Committee of ZNA (Antwerp, Belgium) (EC Approval number 3001) and Ziekenhuis Oost-Limburg (Genk, Belgium) (EC 12/084U). Oral and written informed consent was obtained from the relatives of all patients and there were no deviations from standard clinical practice.
2.2 Patient selection
Sedated and ventilated patients admitted to the ICU (Ziekenhuis Netwerk Antwerpen, ZNA Campus Stuivenberg, Antwerp, Belgium) were included from December 2014 to May 2015. Exclusion criteria were patients younger than 18 years and those in whom there was a medical contraindication for rectal or urinary bladder catheterisation. Demographic data were recorded for all patients.
2.3 Pressure measurements
In most patients, a urinary catheter was already in place with a Foley Manometer Low Volume (FMLV, Holtech, Medical, Charlottenlund, Denmark) attached. If not, a urinary catheter was inserted prior to FMLV attachment. In case of an empty urinary bladder or the presence of air-bubbles obstructing a continuous fluid column in the FMLV, 20 ml of 0.9% sterile sodium chloride solution was injected via the FMLV urine sample port using an aseptic technique. Baseline IAP was measured in the supine position using the FMLV (IAPves) with the zero-reference point in the midaxillary line at the level of the iliac crest (as recommended by WSACS) [14, 18]. IAP was noted at end-expiration, when the meniscus of the fluid column had stabilized and oscillated with the breathing efforts.
The IAPrect was measured using a rectal T-DOC 7Fr air-filled balloon catheter (Laborie Medical Technologies, Mississauga, Canada) connected to a computer displaying the IAP (Audact Pro database version 7.11, Ellipse Andromeda, Urotex, The Netherlands) (ESM Fig. 1). The balloon was inflated with air using a switch, zeroed at atmospheric pressure and inserted 15 cm into the rectum after digital rectal palpation to remove impacted faeces. The catheter was attached to the patient’s leg to prevent displacement.
2.4 Study protocol
IAP is most accurate when measured in a supine position [8, 16]. To validate the accuracy of IAPrect with increasing IAP, measurements were performed in 2 positions in an attempt to artificially increase IAP; the 45° elevated head of bed semirecumbent position, followed with an external abdominal pressure belt (similar to that used by surgeons to prevent incisional hernias). The abdominal belt was put on manually and fastened with a velcro tape and was not released during the protocol. IAPves and IAPrect were measured simultaneously according to a standardized protocol (Fig. 1). All positions (except the application of the external abdominal pressure belt) were repeated twice, including the insertion of the rectal catheter.
Standardized protocol to measure intra-abdominal pressure. Intra-abdominal pressure measurements were performed in different positions: supine position (Supine1 and Supine2) and 45° semirecumbent (45°1 and 45°2) without an external abdominal pressure belt, and 45° semirecumbent (45°APB) and supine position (SupineAPB) with an external abdominal pressure belt (marked with a red spot)
2.5 Statistical analysis
All statistical analyses were performed using SPSS 28.0 software (SPSS inc., Chicago, USA). Results of continuous data that were normally distributed are presented as mean (± SD) unless otherwise stated. Mean values were compared using a paired or independent sample Student’s t test whenever appropriate. A p value of < 0.05 was considered significant. Paired measurements by the two different IAP methods were compared using five different statistical methods.
First, correlations between IAPves and IAPrect were evaluated using univariate linear regression analysis and Pearson correlation coefficient. Two methods are considered equal if the line of identity crosses the origin of X and Y-axis and if R2 (R = Pearson’s correlation coefficient) is > 0.6. Second, we calculated bias (mean difference between reference technique IAPves and IAPrect), precision (SD of the bias) and limits of agreement (bias ± 1.96 × precision) according to Bland and Altman. We followed the Abdominal Compartment Society (WSACS, www.wsacs.org) guidelines and recommendations for research from the international conference of experts on intra-abdominal hypertension and abdominal compartment syndrome on validation of new IAP technology against the gold standard [19]. The bias should be maximal 1 mmHg with a precision less than 2 mmHg to allow two techniques to be used interchangeably. Using a t test and assuming equal standard deviations and an anticipated mean for IAPves around 13.6 ± 3.1 mmHg in the supine position and assuming rectal pressures overestimating IAPves with a mean IAPrect of 18 to 19 mmHg, and assuming a type I error rate alpha of 0.05, with a type II error rate or power (1-β) of 80% an adequate sample size should be 10 to 16, depending on a mean IAPrect of 19 vs. 18 mmHg respectively. Power and sample size calculation was performed with Clincalc (https://clincalc.com/stats/samplesize.aspx).
Third, the percentage error (two times precision of the bias divided by the mean of the reference IAP technique) was calculated as described previously [19]. Based on previous reports, the percentage error for IAP should be less than 35%.
Fourth, Lin’s Concordance Correlation Coefficient (CCC) was calculated as previously described as an extra method for comparing two measurements (rectal vs. vesical) of the same variable (IAP). Ideally the CCC should be above 0.94.
Fifth, the ability of IAPrect to track changes or trends in IAPves was assessed by plotting ΔIAPrect against ΔIAPves during the same time interval (four quadrants trend plot). The concordance is calculated as the percentage of pairs with the same direction of change after exclusion of pairs with both a ΔIAPrect and ΔIAPves ≤ 2.5 mmHg (or less than 15% of change) or with either ΔIAPrect and ΔIAPves equal to zero. Based on clinical relevance, the concordance should be > 90% after exclusion of the pairs falling within the exclusion zone with ΔIAP from − 2.5 to + 2.5 mmHg.
3 Results
3.1 Patient demographics
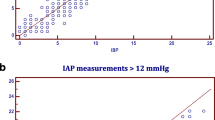
Sixteen patients were included, of whom 7 (43.8%) were not eligible for analysis as all IAPrect measurements were unreliable due to IAPrect values out of physiological range (> 40 mmHg) or strongly fluctuating (> 50%). These patients were found to have profound diarrhoea (n = 1), faecal impaction (n = 2), abdominal muscle contractions in a subconscious patient (n = 1) and difficult placement of the catheter due to anal skin tags and haemorrhoids (n = 1) or morbid obesity (n = 2). An example of both invalid and valid IAPrect measurement is shown in Fig. 2. The men/women ratio of the of the 9 remaining patients (56.2%) eligible for further analysis was 7/2, with mean age 59.0 ± 13.5 years and mean BMI 26.9 ± 6.8 kg/m2. Patients were admitted to ICU for sepsis (n = 3), vascular disease, liver cirrhosis, exacerbation of COPD, pneumonia (n = 4), and major trauma (n = 2).
Output of a rectal intra-abdominal pressure measurement. Intra-abdominal pressure measurement in cmH2O (y-axis) presented over time (x-axis). a invalid measurement due to improper placement of the catheter (first part) and active abdominal muscle contraction (pushing) (last part). b valid measurement in which the influence of the breathing is observed: supine position (Supine1 and supine2) and 45° semirecumbent (45°1 and 45°2) without an external abdominal pressure belt, 45° semirecumbent (45°APB) and supine position (SupineAPB) with an external abdominal pressure belt
3.2 Effect of interventions (position and pressure belt)
Four patients had only one set of measurements and the external abdominal pressure belt was contra-indicated in one patient because of rib fractures, therefore there were in total 14 paired measurements without and 8 measurements with the abdominal belt in supine position. One patient was not able to be put in HOB 45° position because of pain, therefore there were in total 13 paired measurements without and 7 measurements with the abdominal belt in HOB 45° position. Table 1 shows the mean IAPves and IAPrect in both positions, and with or without the external abdominal pressure belt. IAPves was lowest in the supine position (13.6 ± 3.1 mmHg), however, not significantly different from measurement obtained in the semi-recumbent HOB 45° position (15.7 ± 4.4 mmHg, p = 0.103). Measurements with the abdominal pressure belt were significantly higher compared to those without (p < 0.03). Figure 3 shows a boxplot of the median IAPves and IAPrect values in the different body positions and with or without the external abdominal pressure belt.
Boxplots comparing vesical (IAPves) and rectal intra-abdominal pressure (IAPrect) measurements in different body positions. Box and whisker plots comparing IAPves and IAPrect in different body positions. The error bars are the 95% confidence interval, the bottom and top of the box are the 25th and 75th percentiles, the line inside the box is the 50th percentile (median), and any outliers are shown as open circles, p value < 0.05 for all vesical vs. rectal IAP comparisons
IAPrect in the supine position was significantly lower compared to measurements in the semi-recumbent position (20.8 ± 5.0 mmHg versus 24.4 ± 4.8 mmHg (p = 0.002) for supine and semirecumbent position, respectively) and measurements with the abdominal pressure belt were significantly higher than without, (p = 0.032 and p = 0.003 for IAPrect in supine and semi-recumbent positions, respectively).
3.3 Correlation between bladder and rectal pressure measurement techniques
Correlation (Pearson and Lin concordance correlation coefficient) between IAPves and IAPrect was poor (Table 1, Fig. 4, and ESM Fig. 2). An aggregated Bland and Altman analysis for IAPrect versus IAPves (n = 42) shows an abnormal bias of − 8.1 mmHg and precision of 5.6 mmHg with large limits of agreement between − 19 and 2.9 mmHg (Fig. 5, and ESM Fig. 3). The percentage error (LA divided by mean IAP) was 67.3% and too high (should be below 35%) (Table 2).
Correlation scatter plot comparing vesical and rectal intra-abdominal pressure measurements. Correlation plot for all IAPves and IAPrect comparisons in different positions (supine, HOB45°) and with/without abdominal pressure belt (n = 42). Line of identity in blue and linear regression line in black. IAPrect: rectal intra-abdominal pressure; IAPves: vesical intra-abdominal pressure
Bland and Altman plot comparing vesical and rectal intra-abdominal pressure measurements. Aggregated Bland and Altman plot for the mean difference between all IAPrect and IAPves, in different body positions (supine and HOB 45°) and with/without abdominal Pressure belt (n = 42), and their 95% limits of agreement. IAPrect: rectal intra-abdominal pressure; IAPves: vesical intra-abdominal pressure
IAPrect was significantly higher than IAPves for all positions (p < 0.001) and reached the threshold of IAH (≥ 12 mmHg) in all patients, against 64.3% for the IAPves measurements. The IAP difference when moving from supine to 45° HOB semi-recumbent position was different between the two techniques: 1.2 ± 3.1 mmHg versus 3.5 ± 3.1 mmHg, for IAPves and IAPrect, respectively (p = 0.046). The concordance was insufficient (86.2%) after exclusion of the pairs falling within the exclusion zone with ΔIAP from -2.5 to + 2.5 mmHg (Fig. 6).
Four quadrant concordance plot looking at changes in IAP. Four quadrants trend plot for 33 paired measurements of ΔIAPves and ΔIAPrect. From the 33 initial paired measurements, 4 pairs were excluded because either ΔIAP was ≤ ± 2.5 mmHg or equal to zero (exclusion zone, blue shaded area). The calculated level of concordance was 86.2%. See text for explanation
There was a no correlation between Supine1 and Supine2 (Fig. 1) for IAPrect (R = 0.539, p = 0.315). When repeating the protocol, IAPrect was out of physiological range (> 40 mmHg) or unstable in 4/7 patients that were not included for further analysis (57.1%).
4 Discussion
4.1 Correlation between bladder and rectal pressure measurement techniques
Various techniques have been developed to measure IAP, of which the intra-vesical approach is regarded as the gold standard, but it is relatively invasive in an ambulatory setting. There is a need for minimally- or non-invasive techniques of IAP monitoring, especially in obstetrics. Recently different techniques like microwave reflection and transient radar method have been suggested however these are not yet available for clinical use [20, 21]. IAP estimation via rectal measurements seems, at least from a theoretical or hypothetical point-of-view, appropriate and feasible in the pregnant population because of the low-infection risk and lower risk of trauma compared to vesical measurements. However, taking into account the many disadvantages and limitations we cannot recommend its routine ambulatory use, as will be discussed further.
This validation study found that IAPrect is higher when compared to IAPves, and thus may overestimate the true IAH incidence. Furthermore, the IAP differences and trend evolution after position change, or the application of an external abdominal pressure belt, are not similar to the gold standard technique. Correlation was poor, concordance was low, percentage error was too high, and Bland and Altman analysis showed too large LA, hence the two techniques cannot be used interchangeably. We also observed a very high failure rate in obtaining a reproducible IAPrect measurement.
4.2 Effect of body position
Change in body position has a significant impact on IAP measurement [22, 23]. We found that HOB elevation increased both the IAPrect and IAPves, (Fig. 3) which is in keeping with results from the literature. Previous studies confirmed that even a slight elevation in HOB results in a clinically apparent increase in IAP measured through the bladder [2, 22, 23]. Similar investigations were performed to check the impact of body position on IAP measurement not only through the bladder, but also through the stomach (intra-gastric pressure) [24]. HOB elevation increases the intra-gastric pressure (IGP) as well as the intra-bladder pressure, however, the IGP changes were observed to a smaller degree compared to IAPves. In contrast to these studies that have investigated the impact of the upper body position on IAP measurement, the impact of the lower body position on IAP measurement has been assessed recently [25]. In this experiment, IAP measurement through the vagina and (in some cases) rectum at supine, low lithotomy, and high lithotomy positions were evaluated. Based on the results of this study, there is no clinically remarkable change in IAP when the legs are positioned differently. However, the IAP with the patient’s legs in the supine position were lower compared to the low and high lithotomy positions.
4.3 Strengths and limitations of the study
This study is the first to attempt validation of IAPrect measurements against the gold standard IAPves in an ICU-setting [9]. The inclusion of sedated patients, in whom confounding variables are lower, and the strict protocol are strengths of this study [26]. However, the small sample size, the poor description of patient demographics, high drop-out rate and the incompletely performed protocol due to patient or technique related issues are weaknesses and may have underpowered our study results.
The results from this validation study are similar to a study by McCarthy et al., who validated IAPrect in 12 patients but found excessively high or unreliable values in 4 patients (33.3%) due to abdominal traction and technical difficulties on catheter insertion. They concluded that the rectal catheter should be inserted at least 10 cm deep to prevent pressure changes inside the rectum that may result in overestimated readings [27].
Significantly higher IAPrect measurements were observed compared to IAPves, even when IAP is within the physiological range, and as a result IAPrect over-diagnoses IAH. This is in keeping with IAPrect obtained with a fluid-filled rectal catheter balloon in which residual faecal mass can block the catheter-tip opening leading to overestimation of IAP [16]. Correcting this overestimation with a correction factor or the use of a different reference range might not be appropriate as there was no significant correlation between supine1 and supine2. Also, after re-insertion of the rectal catheter, measurement was not repeatable in more than half of the patients. This is in agreement with the results of Lacey et al., who evaluated different indirect techniques against invasive direct IAP measurement in rabbits [28]. Regression analysis showed good correlation with measurements performed in the inferior vena cava (R = 0.87) and the urinary bladder (R = 0.85), but not with intrarectal measurements (R = 0.10) [28]. On the contrary, Shafik et al. found IAPrect to be similar to direct IAP measurement [15]. Note that IAPrect was measured using a fluid filled rectal catheter.
4.4 Limitations of rectal pressure measurement
Several factors may affect IAPrect measured via an air-filled balloon. First, previous studies showed that body temperature is higher in the rectum compared to the urinary bladder [29, 30]. In the present study a small amount of air (at ambient temperature) was used to fill the rectal balloon and air is very sensitive to temperature changes. The higher rectal temperature will result in an increased air temperature in the balloon. In relation to the constant volume this may lead to a significantly increase in rectal pressure measured via the balloon-tipped catheter.
Second, the muscles in the rectum are stronger than the muscles in the urinary bladder. Physiologically, each rectal manipulation and filling stimulates the contraction of the rectal muscles. In a similar way, the insertion and filling of the (even small) balloon could trigger this reflex. Additionally, IAPrect is also affected by the internal anal sphincter tension which contributes about 85% of the pressure in the anal canal [31]. Studies in healthy volunteers showed a significant increase in the internal anal sphincter tension followed by an increase in IAPrect after insertion of an artificial manometer for IAPrect measurement [31, 32]. Therefore, we can assume that rectal insertion of the T-DOC 7Fr air-filled balloon catheter can increase IAPrect per se.
Third, the high failure rate experienced was largely due to IAPrect measuring values out of the physiological range, or due to difficulties with rectal catheter insertion. These excessive IAP values may be caused by interference from faecal masses or bowel movements on the catheter-tip opening, or an incorrect catheter position at the level of the rectal sphincter. Measuring pressure at the level of the rectal sphincter is used in anorectal manometry but it does not yield information regarding true IAP.
Fourth, as pregnant women have an increased risk of constipation and haemorrhoids, therefore, this IAP measurement technique is not suitable to perform in a pregnant population. Laxatives might help to overcome the problem of obstructing stool; however, this is not appropriate in an ambulatory setting.
Fifth, although we perceive rectal pressure measurement as being less or even minimal invasive compared to bladder pressure measurement because of the virtual absence of infection risk it must be noted that rectal manipulation can induce parasympathetic hyperactivity with severe bradycardia and cardiac arrhythmias [33].
Sixth, continuous IAP (CIAP) monitoring is the future [34, 35] and even if rectal pressures would be accurate and comparable to bladder pressures (which was not the case) continuous IAPrect monitoring would be difficult because of probe positioning, displacement, and faecal interference.
5 Conclusion
This validation study found that IAPrect is higher when compared to IAPves. The observed IAP changes induced by position change or the application of an external abdominal pressure belt are not similar when measured via the rectum versus the bladder. Overall correlation was poor, concordance was low, percentage error was too high, and Bland and Altman analysis showed too large LA, hence the two techniques cannot be used interchangeably. IAPrect has important shortcomings making IAP estimation using a rectal catheter unfeasible, largely because the numbers cannot be trusted nor validated. Future studies should be done in different patient populations and/or ambulatory patients to confirm or refute our findings.
Data availability
The datasets used and/or analyzed during the current study are available from the first author on reasonable request.
Abbreviations
- ACS:
-
Abdominal compartment syndrome
- IAP:
-
Intra-abdominal pressure
- IAH:
-
Intra-abdominal hypertension
- IAPves :
-
Vesical intra-abdominal pressure
- IAPrect :
-
Rectal intra-abdominal pressure
- HOB:
-
Head of bed
- FMLV:
-
Foley Manometer Low Volume
- LA:
-
Limits of agreement
- COPD:
-
Chronic obstructive pulmonary disease
- ICU:
-
Intensive care unit
References
Malbrain ML, Cheatham ML, Kirkpatrick A, Sugrue M, Parr M, De Waele J, et al. Results from the international conference of experts on intra-abdominal hypertension and abdominal compartment syndrome. I Definitions Intensive Care Med. 2006;32(11):1722–32.
De Keulenaer BL, De Waele JJ, Powell B, Malbrain ML. What is normal intra-abdominal pressure and how is it affected by positioning, body mass and positive end-expiratory pressure? Intensive Care Med. 2009;35(6):969–76.
Malbrain ML, Chiumello D, Cesana BM, Reintam Blaser A, Starkopf J, Sugrue M, et al. A systematic review and individual patient data meta-analysis on intra-abdominal hypertension in critically ill patients: the wake-up project. World initiative on Abdominal Hypertension Epidemiology, a Unifying Project (WAKE-Up!). Minerva Anestesiol. 2014;80(3):293–306.
Kirkpatrick AW, De Waele JJ, De Laet I, De Keulenaer BL, D’Amours S, Bjorck M, et al. WSACS—The Abdominal Compartment Society. A Society dedicated to the study of the physiology and pathophysiology of the abdominal compartment and its interactions with all organ systems. Anaesthesiol Intensive Ther. 2015;47(3):191–4.
De Waele JJ, Malbrain ML, Kirkpatrick AW. The abdominal compartment syndrome: evolving concepts and future directions. Crit Care. 2015;19:211.
Cheatham ML, Safcsak K, Sugrue M. Long-term implications of intra-abdominal hypertension and abdominal compartment syndrome: physical, mental, and financial. Am Surg. 2011;77(Suppl 1):S78-82.
Malbrain ML, Chiumello D, Pelosi P, Wilmer A, Brienza N, Malcangi V, et al. Prevalence of intra-abdominal hypertension in critically ill patients: a multicentre epidemiological study. Intensive Care Med. 2004;30(5):822–9.
Malbrain ML, De Laet IE, De Waele JJ, Kirkpatrick AW. Intra-abdominal hypertension: definitions, monitoring, interpretation and management. Best Pract Res Clin Anaesthesiol. 2013;27(2):249–70.
Malbrain ML, Deeren D, De Potter TJ. Intra-abdominal hypertension in the critically ill: it is time to pay attention. Curr Opin Crit Care. 2005;11(2):156–71.
Malbrain ML, Roberts DJ, Sugrue M, De Keulenaer BL, Ivatury R, Pelosi P, et al. The polycompartment syndrome: a concise state-of-the-art review. Anaesthesiol Intensive Ther. 2014;46(5):433–50.
Sugerman HJ. Hypothesis: preeclampsia is a venous disease secondary to an increased intra-abdominal pressure. Med Hypotheses. 2011;77(5):841–9.
Paramore RH. The intra-abdominal pressure in pregnancy. Proc R Soc Med. 1913;6:291–334.
Lozada MJ, Goyal V, Levin D, Walden RL, Osmundson SS, Pacheco LD, et al. Management of peripartum intra-abdominal hypertension and abdominal compartment syndrome. Acta Obstet Gynecol Scand. 2019;98(11):1386–97.
Kirkpatrick AW, Roberts DJ, De Waele J, Jaeschke R, Malbrain ML, De Keulenaer B, et al. Intra-abdominal hypertension and the abdominal compartment syndrome: updated consensus definitions and clinical practice guidelines from the World Society of the Abdominal Compartment Syndrome. Intensive Care Med. 2013;39(7):1190–206.
Shafik A, El-Sharkawy A, Sharaf WM. Direct measurement of intra-abdominal pressure in various conditions. Eur J Surg. 1997;163(12):883–7.
Malbrain ML. Different techniques to measure intra-abdominal pressure (IAP): time for a critical re-appraisal. Intensive Care Med. 2004;30(3):357–71.
James ED, Niblett PG, MacNaughton JA, Shaldon C. The vagina as an alternative to the rectum in measuring abdominal pressure during urodynamic investigations. Br J Urol. 1987;60(3):212–6.
Cheatham ML, Malbrain ML, Kirkpatrick A, Sugrue M, Parr M, De Waele J, et al. Results from the international conference of experts on intra-abdominal hypertension and abdominal compartment syndrome. II. Recommendations. Intensive Care Med. 2007;33(6):951–62.
De Waele JJ, Cheatham ML, Malbrain MLNG, Kirkpatrick AW, Sugrue M, Balogh Z, et al. Recommendations for research from the international conference of experts on intra-abdominal hypertension and abdominal compartment syndrome. Acta Clin Belg. 2009;64(3):203–9.
Tayebi S, Pourkazemi A, Malbrain M, Stiens J. Non-invasive intra-abdominal pressure measurement by means of transient radar method: in vitro validation of a novel radar-based sensor. Sensors. 2021;21(18):5999.
Tayebi S, Gutierrez A, Mohout I, Smets E, Wise R, Stiens J, et al. A concise overview of non-invasive intra-abdominal pressure measurement techniques: from bench to bedside. J Clin Monit Comput. 2021;35(1):51–70.
Cheatham ML, De Waele JJ, De Laet I, De Keulenaer B, Widder S, Kirkpatrick AW, et al. The impact of body position on intra-abdominal pressure measurement: a multicenter analysis. Crit Care Med. 2009;37(7):2187–90.
Cresswell AB, Jassem W, Srinivasan P, Prachalias AA, Sizer E, Burnal W, et al. The effect of body position on compartmental intra-abdominal pressure following liver transplantation. Ann Intensive Care. 2012;2(Suppl 1):S12.
Rooban N, Regli A, Davis WA, De Keulenaer BL. Comparing intra-abdominal pressures in different body positions via a urinary catheter and nasogastric tube: a pilot study. Ann Intensive Care. 2012;2(Suppl 1):S11.
Young K, Mou T, Geynisman-Tan J, Tavathia M, Collins S, Mueller M, et al. Truth or myth: intra-abdominal pressure increases in the lithotomy position. J Minim Invasive Gynecol. 2021;28(1):26–9.
Regli A, Nanda R, Braun J, Girardis M, Max M, Malbrain ML, et al. The effect of non-invasive ventilation on intra-abdominal pressure. Anaesthesiol Intensive Ther. 2022;54(1):30–3.
McCarthy TA. Validity of rectal pressure measurements as indication of intra-abdominal pressure changes during urodynamic evaluation. Urology. 1982;20(6):657–60.
Lacey SR, Bruce J, Brooks SP, Griswald J, Ferguson W, Allen JE, et al. The relative merits of various methods of indirect measurement of intraabdominal pressure as a guide to closure of abdominal wall defects. J Pediatr Surg. 1987;22(12):1207–11.
Lefrant JY, Muller L, de La Coussaye JE, Benbabaali M, Lebris C, Zeitoun N, et al. Temperature measurement in intensive care patients: comparison of urinary bladder, oesophageal, rectal, axillary, and inguinal methods versus pulmonary artery core method. Intensive Care Med. 2003;29(3):414–8.
Schell-Chaple HM, Liu KD, Matthay MA, Puntillo KA. Rectal and bladder temperatures vs forehead core temperatures measured with SpotOn monitoring system. Am J Crit Care. 2018;27(1):43–50.
Frenckner B, Euler CV. Influence of pudendal block on the function of the anal sphincters. Gut. 1975;16(6):482–9.
Lestar B, Penninckx FM, Kerremans RP. Defecometry. A new method for determining the parameters of rectal evacuation. Dis Colon Rectum. 1989;32(3):197–201.
Sampson CS, Shea CM. Death by disimpaction: a bradycardic arrest secondary to rectal manipulation. Case Rep Emerg Med. 2016;2016:5489325.
Kaussen T, Gutting M, Lasch F, Boethig D, von Gise A, Dingemann J, et al. Continuous intra-gastral monitoring of intra-abdominal pressure in critically ill children: a validation study. Intensive Care Med Exp. 2021;9(1):24.
Khanna A, Minear S, Prabhakar A, Kurz A, Stanton K, Essakalli L, et al. Intra-abdominal hypertension in cardiac surgery patients: a multicenter observational pilot study. Journal of Clinical Monitoring and Computing. 2022.
Acknowledgements
The authors want to thank Robert Wise, Sharona Vonck, Inneke de Laet, Karen Schoonheydt, Hilde Dits, Salar Tayebi, and Wojciech Dabrowski for their help and advice performing the study and editing and revising this manuscript. Manu Malbrain is professor at the Medical University of Lublin and member of the Executive Committee of the Abdominal Compartment Society, formerly known as the World Society of Abdominal Compartment Syndrome (https://www.wsacs.org/). He is co-founder, past-president and current treasurer of WSACS. He is co-founder of the International Fluid Academy (IFA). The mission statement of the IFA is to foster education, promote research on fluid management and hemodynamic monitoring, and thereby improve survival of critically ill by bringing together physicians, nurses, and others from throughout the world and from a variety of clinical disciplines. The IFA is integrated within the not-for-profit charitable organization iMERiT, International Medical Education and Research Initiative, under Belgian law. The content of the IFA website (http://www.fluidacademy.org) is based on the philosophy of FOAM (Free Open Access Medical education—#FOAMed).
Funding
This work is part of a PhD-thesis, which is supported by the Limburg Clinical Research Program (LCRP) UHasselt-ZOL-Jessa, supported by the foundation Limburg Sterk Merk, Hasselt University, Ziekenhuis Oost-Limburg and Jessa Hospital.
Author information
Authors and Affiliations
Contributions
ASS created the study protocol, performed the measurements, analysed the data, searched for relevant literature and wrote the manuscript. AH performed the measurements, analysed the data and searched for relevant literature. SC revised the manuscript. NVR edited and revised the manuscript. WG created the study protocol and revised the manuscript. MLNGM created the study protocol, performed the measurements, searched for relevant literature, analysed the data, and revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
MLNGM is a member of the medical advisory Board of Pulsion Medical Systems (now fully integrated in Getinge, Solna, Sweden), Baxter and Serenno Medical (Tel Aviv, Israel), and consults for Maltron, ConvaTec, BD, Spiegelberg and Holtech Medical. All other authors declare that they have no competing interests in relation to the content published in this manuscript.
Ethical approval
The study was conducted in accordance with the study protocol, the Declaration of Helsinki and applicable regulatory requirements. We applied for approval of the protocol by the local Institutional Review Board and Ethics Committee of the Ziekenhuis Netwerk Antwerpen, ZNA Stuivenberg approved the protocol (EC approval number: 3001 with insurance policy Ethias 45.313.314) and Ziekenhuis Oost-Limburg, Genk, Belgium (EC 12/084U).
Consent to participate
Oral and written informed consent was obtained from the relatives of all patients. There were no deviations from standard care.
Consent for publication
Consent for publication of figures and manuscript was obtained from the persons involved.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Staelens, A.S., Heymans, A., Christiaens, S. et al. Is it feasible to measure intra-abdominal pressure using a balloon-tipped rectal catheter? Results of a validation study. J Clin Monit Comput 37, 287–296 (2023). https://doi.org/10.1007/s10877-022-00890-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-022-00890-6