Abstract
Purpose
Noninvasive detection of high-risk plaques is still challenging. In this study, we aimed to noninvasively assess αvβ3-integrin expression using a customed photoacoustic (PA) computed tomography (PACT)/ultrasound (US) system in atherosclerotic lesions of varying degrees of severity and to explore its potential value for detecting high-risk plaques.
Methods
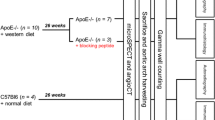
We constructed αvβ3-integrin-targeted ultrasmall gold nanorods (AuNRs) with cyclo Arg-Gly-Asp (cRGD) and tested their properties. Employing C57BL/6 J (wild-type, WT) mice and apolipoprotein E gene knockout (ApoE−/−) mice fed either a chow diet or a high-fat/high-cholesterol diet (HFHCD), we established varying degrees of lesion severity. In vivo PACT/US imaging was performed to assess αvβ3-integrin expression in the 4 groups by cRGD-AuNRs. Further histopathologic examination was conducted to evaluate the plaque vulnerability indicators.
Results
The data showed that cRGD-AuNRs exhibited excellent photothermal conversion capacity, stability, targeting ability, and biocompatibility. The immunohistochemical results indicated that αvβ3-integrin was upregulated with increasing aggravation of the lesions. In vivo PACT/US imaging showed good consistency with αvβ3-integrin expression. Notably, ApoE−/− mice fed a HFHCD showed an abrupt PA intensity increase compared with the other groups. The histopathologic examination verified that the atherosclerotic plaques of ApoE−/− mice fed the HFHCD developed unstable phenotypes. Correlation analysis showed that PA intensity was mainly related to inflammation and angiogenesis among all of the indicators.
Conclusion
Our data indicated that αvβ3-integrin is an effective indicator of plaque instability, and noninvasive PACT/US molecular imaging assessment of αvβ3-integrin holds promise in detecting high-risk plaques.








Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding authors on reasonable request.
References
Fiolet ATL, Opstal TSJ, Mosterd A, Eikelboom JW, Jolly SS, Keech AC, et al. Efficacy and safety of low-dose colchicine in patients with coronary disease: a systematic review and meta-analysis of randomized trials. Eur Heart J. 2021;42:2765–75. https://doi.org/10.1093/eurheartj/ehab115.
Campbell BCV, Khatri P. Stroke Lancet. 2020;396:129–42. https://doi.org/10.1016/S0140-6736(20)31179-X.
van der Wal AC, Becker AE, van der Loos CM, Das PK. Site of intimal rupture or erosion of thrombosed coronary atherosclerotic plaques is characterized by an inflammatory process irrespective of the dominant plaque morphology. Circulation. 1994;89:36–44. https://doi.org/10.1161/01.cir.89.1.36.
Sugizaki Y, Otake H, Kawamori H, Toba T, Nagano Y, Tsukiyama Y, et al. Adding alirocumab to rosuvastatin helps reduce the vulnerability of thin-cap fibroatheroma: an ALTAIR trial report. JACC Cardiovasc Imaging. 2020;13:1452–4. https://doi.org/10.1016/j.jcmg.2020.01.021.
Kedhi E, Berta B, Roleder T, Hermanides RS, Fabris E, AJJ IJ, et al. Thin-cap fibroatheroma predicts clinical events in diabetic patients with normal fractional flow reserve: the COMBINE OCT-FFR trial. Eur Heart J. 2021. https://doi.org/10.1093/eurheartj/ehab433.
Tomaniak M, Katagiri Y, Modolo R, de Silva R, Khamis RY, Bourantas CV, et al. Vulnerable plaques and patients: state-of-the-art. Eur Heart J. 2020;41:2997–3004. https://doi.org/10.1093/eurheartj/ehaa227.
Stone GW, Maehara A, Lansky AJ, de Bruyne B, Cristea E, Mintz GS, et al. A prospective natural-history study of coronary atherosclerosis. N Engl J Med. 2011;364:226–35. https://doi.org/10.1056/NEJMoa1002358.
Vancraeynest D, Pasquet A, Roelants V, Gerber BL, Vanoverschelde JL. Imaging the vulnerable plaque. J Am Coll Cardiol. 2011;57:1961–79. https://doi.org/10.1016/j.jacc.2011.02.018.
Osborn EA, Kessinger CW, Tawakol A, Jaffer FA. Metabolic and molecular imaging of atherosclerosis and venous thromboembolism. J Nucl Med. 2017;58:871–7. https://doi.org/10.2967/jnumed.116.182873.
Beer AJ, Pelisek J, Heider P, Saraste A, Reeps C, Metz S, et al. PET/CT imaging of integrin alphavbeta3 expression in human carotid atherosclerosis. JACC Cardiovasc Imaging. 2014;7:178–87. https://doi.org/10.1016/j.jcmg.2013.12.003.
Vancraeynest D, Roelants V, Bouzin C, Hanin FX, Walrand S, Bol V, et al. alphaVbeta3 integrin-targeted microSPECT/CT imaging of inflamed atherosclerotic plaques in mice. EJNMMI Res. 2016;6:29. https://doi.org/10.1186/s13550-016-0184-9.
Winter PM, Morawski AM, Caruthers SD, Fuhrhop RW, Zhang H, Williams TA, et al. Molecular imaging of angiogenesis in early-stage atherosclerosis with alpha(v)beta3-integrin-targeted nanoparticles. Circulation. 2003;108:2270–4. https://doi.org/10.1161/01.CIR.0000093185.16083.95.
Daeichin V, Kooiman K, Skachkov I, Bosch JG, Theelen TL, Steiger K, et al. Quantification of endothelial alphavbeta3 expression with high-frequency ultrasound and targeted microbubbles: in vitro and in vivo studies. Ultrasound Med Biol. 2016;42:2283–93. https://doi.org/10.1016/j.ultrasmedbio.2016.05.005.
Zhang P, Li L, Lin L, Shi J, Wang LV. In vivo superresolution photoacoustic computed tomography by localization of single dyed droplets. Light Sci Appl. 2019;8:36. https://doi.org/10.1038/s41377-019-0147-9.
Zhang J, Yang S, Ji X, Zhou Q, Xing D. Characterization of lipid-rich aortic plaques by intravascular photoacoustic tomography: ex vivo and in vivo validation in a rabbit atherosclerosis model with histologic correlation. J Am Coll Cardiol. 2014;64:385–90. https://doi.org/10.1016/j.jacc.2014.04.053.
Wang B, Yantsen E, Larson T, Karpiouk AB, Sethuraman S, Su JL, et al. Plasmonic intravascular photoacoustic imaging for detection of macrophages in atherosclerotic plaques. Nano Lett. 2009;9:2212–7. https://doi.org/10.1021/nl801852e.
Lin L, Xie Z, Xu M, Wang Y, Li S, Yang N, et al. IVUS\IVPA hybrid intravascular molecular imaging of angiogenesis in atherosclerotic plaques via RGDfk peptide-targeted nanoprobes. Photoacoustics. 2021;22:100262. https://doi.org/10.1016/j.pacs.2021.100262.
Razansky D, Harlaar NJ, Hillebrands JL, Taruttis A, Herzog E, Zeebregts CJ, et al. Multispectral optoacoustic tomography of matrix metalloproteinase activity in vulnerable human carotid plaques. Mol Imaging Biol. 2012;14:277–85. https://doi.org/10.1007/s11307-011-0502-6.
Johnson JL, Merrilees M, Shragge J, van Wijk K. All-optical extravascular laser-ultrasound and photoacoustic imaging of calcified atherosclerotic plaque in excised carotid artery. Photoacoustics. 2018;9:62–72. https://doi.org/10.1016/j.pacs.2018.01.002.
Wu C, Zhang Y, Li Z, Li C, Wang Q. A novel photoacoustic nanoprobe of ICG@PEG-Ag2S for atherosclerosis targeting and imaging in vivo. Nanoscale. 2016;8:12531–9. https://doi.org/10.1039/c6nr00060f.
Ge X, Cui H, Kong J, Lu SY, Zhan R, Gao J, et al. A non-invasive nanoprobe for in vivo photoacoustic imaging of vulnerable atherosclerotic plaque. Adv Mater. 2020;32:e2000037. https://doi.org/10.1002/adma.202000037.
Xie Z, Yang Y, He Y, Shu C, Chen D, Zhang J, et al. In vivo assessment of inflammation in carotid atherosclerosis by noninvasive photoacoustic imaging. Theranostics. 2020;10:4694–704. https://doi.org/10.7150/thno.41211.
Liu C, Gong X, Lin R, Liu F, Chen J, Wang Z, et al. Advances in imaging techniques and genetically encoded probes for photoacoustic imaging. Theranostics. 2016;6:2414–30. https://doi.org/10.7150/thno.15878.
Cho S, Baik J, Managuli R, Kim C. 3D PHOVIS: 3D photoacoustic visualization studio. Photoacoustics. 2020;18:100168. https://doi.org/10.1016/j.pacs.2020.100168.
Lin LY, Keeler EG. Progress of MEMS scanning micromirrors for optical bio-imaging. Micromachines. 2015;6:1675–89.
Wang D, Liang P, Samuelson S, Jia H, Ma J, Xie H. Correction of image distortions in endoscopic optical coherence tomography based on two-axis scanning MEMS mirrors. Biomed Opt Express. 2013;4:2066–77. https://doi.org/10.1364/BOE.4.002066.
Lu C, Xiong K, Ma Y, Zhang W, Cheng Z, Yang S. Electrothermal-MEMS-induced nonlinear distortion correction in photoacoustic laparoscopy. Opt Express. 2020;28:15300–13. https://doi.org/10.1364/OE.392493.
Zhou Y, Wang D, Zhang Y, Chitgupi U, Geng J, Wang Y, et al. A phosphorus phthalocyanine formulation with intense absorbance at 1000 nm for deep optical imaging. Theranostics. 2016;6:688–97. https://doi.org/10.7150/thno.14555.
Wang LV, Hu S. Photoacoustic tomography: in vivo imaging from organelles to organs. Science. 2012;335:1458–62. https://doi.org/10.1126/science.1216210.
Mantri Y, Jokerst JV. Engineering plasmonic nanoparticles for enhanced photoacoustic imaging. ACS Nano. 2020;14:9408–22. https://doi.org/10.1021/acsnano.0c05215.
Li Z, Huang P, Zhang X, Lin J, Yang S, Liu B, et al. RGD-conjugated dendrimer-modified gold nanorods for in vivo tumor targeting and photothermal therapy. Mol Pharm. 2010;7:94–104. https://doi.org/10.1021/mp9001415.
Chen YS, Zhao Y, Yoon SJ, Gambhir SS, Emelianov S. Miniature gold nanorods for photoacoustic molecular imaging in the second near-infrared optical window. Nat Nanotechnol. 2019;14:465–72. https://doi.org/10.1038/s41565-019-0392-3.
Reynolds LE, Wyder L, Lively JC, Taverna D, Robinson SD, Huang X, et al. Enhanced pathological angiogenesis in mice lacking beta3 integrin or beta3 and beta5 integrins. Nat Med. 2002;8:27–34. https://doi.org/10.1038/nm0102-27.
Maile LA, Busby WH, Nichols TC, Bellinger DA, Merricks EP, Rowland M, et al. A monoclonal antibody against alphaVbeta3 integrin inhibits development of atherosclerotic lesions in diabetic pigs. Sci Transl Med. 2010;2:18ra1. https://doi.org/10.1126/scitranslmed.3000476.
Jenkins WS, Vesey AT, Vickers A, Neale A, Moles C, Connell M, et al. In vivo alpha-V beta-3 integrin expression in human aortic atherosclerosis. Heart. 2019;105:1868–75. https://doi.org/10.1136/heartjnl-2019-315103.
Ali MR, Snyder B, El-Sayed MA. Synthesis and optical properties of small Au nanorods using a seedless growth technique. Langmuir. 2012;28:9807–15. https://doi.org/10.1021/la301387p.
Kumthekar P, Ko CH, Paunesku T, Dixit K, Sonabend AM, Bloch O, et al. A first-in-human phase 0 clinical study of RNA interference-based spherical nucleic acids in patients with recurrent glioblastoma. Sci Transl Med. 2021;13. https://doi.org/10.1126/scitranslmed.abb3945.
Khoobchandani M, Katti KK, Karikachery AR, Thipe VC, Srisrimal D, Dhurvas Mohandoss DK, et al. New approaches in breast cancer therapy through green nanotechnology and nano-ayurvedic medicine - pre-clinical and pilot human clinical investigations. Int J Nanomedicine. 2020;15:181–97. https://doi.org/10.2147/IJN.S219042.
Ramirez-Nava G, Santos-Cuevas C, Ferro-Flores G, Ocampo-Garcia B, Chairez I, Gomez-Argumosa E, et al. Hybrid (2D/3D) Dosimetry of radiolabeled gold nanoparticles for sentinel lymph node detection in patients with breast cancer. Contrast Media Mol Imaging. 2020;2020:2728134. https://doi.org/10.1155/2020/2728134.
Kharlamov AN, Tyurnina AE, Veselova VS, Kovtun OP, Shur VY, Gabinsky JL. Silica-gold nanoparticles for atheroprotective management of plaques: results of the NANOM-FIM trial. Nanoscale. 2015;7:8003–15. https://doi.org/10.1039/c5nr01050k.
Lankveld DP, Rayavarapu RG, Krystek P, Oomen AG, Verharen HW, van Leeuwen TG, et al. Blood clearance and tissue distribution of PEGylated and non-PEGylated gold nanorods after intravenous administration in rats. Nanomedicine (Lond). 2011;6:339–49. https://doi.org/10.2217/nnm.10.122.
Zhu XM, Fang C, Jia H, Huang Y, Cheng CH, Ko CH, et al. Cellular uptake behaviour, photothermal therapy performance, and cytotoxicity of gold nanorods with various coatings. Nanoscale. 2014;6:11462–72. https://doi.org/10.1039/c4nr03865g.
Panchatcharam M, Miriyala S, Yang F, Leitges M, Chrzanowska-Wodnicka M, Quilliam LA, et al. Enhanced proliferation and migration of vascular smooth muscle cells in response to vascular injury under hyperglycemic conditions is controlled by beta3 integrin signaling. Int J Biochem Cell Biol. 2010;42:965–74. https://doi.org/10.1016/j.biocel.2010.02.009.
Fu Q, Zhu R, Song J, Yang H, Chen X. Photoacoustic imaging: contrast agents and their biomedical applications. Adv Mater. 2019;31:e1805875. https://doi.org/10.1002/adma.201805875.
Acknowledgements
We thank Prof Jianping, Bin and Prof Longquan, Shao for their valuable advice on our manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (No. 81974266, No. 81470598, No.82122034).
Author information
Authors and Affiliations
Contributions
Xuewei Liu, Rongkang Gao, and Chiyun Chen designed the research studies, conducted the PACT/US experiments, AuNRs synthesis, analyzed data, and assisted with preparing the manuscript. Xiaobo Li, Chen Yu, and Yejia Chen performed histopathological experiments. Hongbin Liang and Min Xiao analyzes the offline data of PACT/US imaging. Lei Dai, Shifeng Qiu, and Xiaoyu Xin provide reagents. Liang Song provided conceptual and technical advice. Jiancheng Xiu, Chengbo Liu, and Jinbin Liu conceived the idea, designed research studies, conducted experiments, supervised the study, and prepared the manuscript. All authors contributed to the writing and review of the manuscript.
Corresponding authors
Ethics declarations
Conflicts of Interest
The authors declared no competing interests.
Ethics approval
The animal study protocols were approved by the Nanfang hospital animal ethic committee.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Cardiology
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, X., Gao, R., Chen, C. et al. Noninvasive photoacoustic computed tomography/ultrasound imaging to identify high-risk atherosclerotic plaques. Eur J Nucl Med Mol Imaging 49, 4601–4615 (2022). https://doi.org/10.1007/s00259-022-05911-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-022-05911-9