Abstract
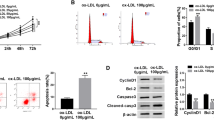
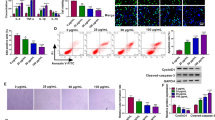
Atherosclerosis (AS) is a chronic inflammatory disease with high morbidity and mortality rates worldwide. This study aimed to investigate the role of circular RNA protein tyrosine phosphatase receptor type A (circRNA_PTPRA) in oxidized low-density lipoprotein (ox-LDL)-induced human umbilical vein endothelial cell (HUVECs) injury and its underlying molecular mechanism. The expression of circRNA-PTPRA and microRNA (miR)-671-5p was assessed by quantitative reverse transcription PCR (qRT-PCR). The interaction between circRNA-PTPRA and miR-671-5p was predicted using bioinformatic analysis. Cell viability and apoptosis were determined using the Cell Counting Kit-8 (CCK-8) assay and flow cytometry, respectively. Inflammation in HUVECs was analyzed by measuring the secretion of tumor necrosis factor alpha (TNF-α), interleukin-1beta (IL-1β), and IL-6 using enzyme-linked immunosorbent assay (ELISA). Cleaved-caspase-3 expression was assessed using western blotting. The results indicated that circRNA-PTPRA expression was significantly increased and miR-671-5p expression was decreased in the serum of patients with AS and in ox-LDL-treated HUVECs. The interaction between circRNA-PTPRA and miR-671-5p was verified by dual luciferase reporter and RNA pull-down assays. In HUVECs, downregulation of circRNA-PTPRA reversed ox-LDL-induced reduction in cell viability, increase in apoptosis, and enhanced inflammation, whereas all these effects mediated by circRNA-PTPRA downregulation in ox-LDL-treated HUVECs were abolished by miR-671-5p downregulation. In conclusion, circRNA-PTPRA downregulation protects against ox-LDL-induced HUVECs injury by upregulating miR-671-5p, thereby providing potential therapeutic targets for AS.







Similar content being viewed by others
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Bahn JH, Zhang Q, Li F, Chan TM, Lin X, Kim Y, Wong DT, Xiao X (2015) The landscape of microRNA, Piwi-interacting RNA, and circular RNA in human saliva. Clin Chem 61(1):221–230. https://doi.org/10.1373/clinchem.2014.230433
Burd CE, Jeck WR, Liu Y, Sanoff HK, Wang Z, Sharpless NE (2010) Expression of linear and novel circular forms of an INK4/ARF-associated non-coding RNA correlates with atherosclerosis risk. PLoS Genet 6(12):e1001233. https://doi.org/10.1371/journal.pgen.1001233
Chaichian S, Shafabakhsh R, Mirhashemi SM, Moazzami B, Asemi Z (2020) Circular RNAs: a novel biomarker for cervical cancer. J Cell Physiol 235(2):718–724. https://doi.org/10.1002/jcp.29009
de Yébenes VG, Briones AM, Martos-Folgado I, Mur SM, Oller J, Bilal F, González-Amor M, Méndez-Barbero N, Silla-Castro JC, Were F, Jiménez-Borreguero LJ, Sánchez-Cabo F, Bueno H, Salaices M, Redondo JM, Ramiro AR (2020) Aging-associated miR-217 aggravates atherosclerosis and promotes cardiovascular dysfunction. Arterioscler Thromb Vasc Biol 40(10):2408–2424. https://doi.org/10.1161/ATVBAHA.120.314333
Du WW, Zhang C, Yang W, Yong T, Awan FM, Yang BB (2017) Identifying and characterizing circRNA-protein interaction. Theranostics 7(17):4183–4191. https://doi.org/10.7150/thno.21299
Esteller M (2011) Non-coding RNAs in human disease. Nat Rev Genet 12(12):861–874. https://doi.org/10.1038/nrg3074
Falk E (2006) Pathogenesis of atherosclerosis. J Am Coll Cardiol 47(8 Suppl):C7-12. https://doi.org/10.1016/j.jacc.2005.09.068
Fasolo F, Di Gregoli K, Maegdefessel L, Johnson JL (2019) Non-coding RNAs in cardiovascular cell biology and atherosclerosis. Cardiovasc Res 115(12):1732–1756. https://doi.org/10.1093/cvr/cvz203
Gao J, Yang S, Wang K, Zhong Q, Ma A, Pan X (2019) Plasma miR-126 and miR-143 as potential novel biomarkers for cerebral atherosclerosis. J Stroke Cerebrovasc Dis 28(1):38–43. https://doi.org/10.1016/j.jstrokecerebrovasdis
GBD 2015 Risk Factors Collaborators (2016) Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet 388(10053):1659–1724. https://doi.org/10.1016/S0140-6736(16)31679-8
Hansen TB, Jensen TI, Clausen BH, Bramsen JB, Finsen B, Damgaard CK, Kjems J (2013) Natural RNA circles function as efficient microRNA sponges. Nature 495(7441):384–388. https://doi.org/10.1038/nature11993
Hopkins PN (2013) Molecular biology of atherosclerosis. Physiol Rev 93(3):1317–1542. https://doi.org/10.1152/physrev.00004.2012
Jiang T, Jiang D, You D, Zhang L, Liu L, Zhao Q (2020) Agonism of GPR120 prevents ox-LDL-induced attachment of monocytes to endothelial cells. Chem Biol Interact 316:108916. https://doi.org/10.1016/j.cbi.2019.108916
Jin W, Shi J, Liu M (2019) Overexpression of miR-671-5p indicates a poor prognosis in colon cancer and accelerates proliferation, migration, and invasion of colon cancer cells. Onco Targets Ther 12:6865–6873. https://doi.org/10.2147/OTT.S219421
Lechner K, von Schacky C, McKenzie AL, Worm N, Nixdorff U, Lechner B, Kränkel N, Halle M, Krauss RM, Scherr J (2020) Lifestyle factors and high-risk atherosclerosis: pathways and mechanisms beyond traditional risk factors. Eur J Prev Cardiol 27(4):394–406. https://doi.org/10.1177/2047487319869400
Li X, Diao H (2019) Circular RNA circ_0001946 acts as a competing endogenous RNA to inhibit glioblastoma progression by modulating miR-671-5p and CDR1. J Cell Physiol 234(8):13807–13819. https://doi.org/10.1002/jcp.28061
Li Y, Tian L, Sun D, Yin D (2019a) Curcumin ameliorates atherosclerosis through upregulation of miR-126. J Cell Physiol 234(11):21049–21059. https://doi.org/10.1002/jcp.28708
Li X, Nie C, Tian B, Tan X, Han W, Wang J, Jin Y, Li Y, Guan X, Hong A, Chen X (2019b) miR-671-5P blocks the progression of human esophageal squamous cell carcinoma by suppressing FGFR2. Int J Biol Sci 15(9):1892–1904. https://doi.org/10.7150/ijbs.32429
Libby P, Ridker PM, Hansson GK (2009) Leducq transatlantic network on atherothrombosis. Inflammation in atherosclerosis: from pathophysiology to practice. J Am Coll Cardiol 54(23):2129–2138. https://doi.org/10.1016/j.jacc.2009.09.009
Libby P, Buring JE, Badimon L, Hansson GK, Deanfield J, Bittencourt MS, Tokgözoğlu L, Lewis EF (2019a) Atherosclerosis. Nat Rev Dis Primers 5(1):56. https://doi.org/10.1038/s41572-019-0106-z
Libby P, Pasterkamp G, Crea F, Jang IK (2019b) Reassessing the mechanisms of acute coronary syndromes. Circ Res 124(1):150–160. https://doi.org/10.1161/CIRCRESAHA.118.311098
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Ma S, Wang S, Li M, Zhang Y, Zhu P (2018) The effects of pigment epithelium-derived factor on atherosclerosis: putative mechanisms of the process. Lipids Health Dis 17(1):240. https://doi.org/10.1186/s12944-018-0889-z
Novak J, Vašků JB, Souček M (2018) Dlouhé nekódující RNA v patofyziologii aterosklerózy [Long non-coding RNAs in the pathophysiology of atherosclerosis]. Vnitr Lek 64(1):77–82
Pan RY, Liu P, Zhou HT, Sun WX, Song J, Shu J, Cui GJ, Yang ZJ, Jia EZ (2017) Circular RNAs promote TRPM3 expression by inhibiting hsa-miR-130a-3p in coronary artery disease patients. Oncotarget 8(36):60280–60290. https://doi.org/10.18632/oncotarget.19941
Qiu T, Wang K, Li X, Jin J (2018) miR-671-5p inhibits gastric cancer cell proliferation and promotes cell apoptosis by targeting URGCP. Exp Ther Med 16(6):4753–4758. https://doi.org/10.3892/etm.2018.6813
Ross R (1999) Atherosclerosis: an inflammatory disease. N Engl J Med 340(2):115–126. https://doi.org/10.1056/NEJM199901143400207
Shao BZ, Han BZ, Zeng YX, Su DF, Liu C (2016) The roles of macrophage autophagy in atherosclerosis. Acta Pharmacol Sin 37(2):150–156. https://doi.org/10.1038/aps.2015.87
Sharif H, Akash MSH, Rehman K, Irshad K, Imran I (2020) Pathophysiology of atherosclerosis: association of risk factors and treatment strategies using plant-based bioactive compounds. J Food Biochem 44(11):e13449. https://doi.org/10.1111/jfbc.13449
Shen L, Hu Y, Lou J, Yin S, Wang W, Wang Y, Xia Y, Wu W (2019) CircRNA-0044073 is upregulated in atherosclerosis and increases the proliferation and invasion of cells by targeting miR-107. Mol Med Rep 19(5):3923–3932. https://doi.org/10.3892/mmr.2019.10011
Sun L, Dou F, Chen J, Chi H, Xing S, Liu T, Sun S, Chen C (2018) Salidroside slows the progression of EA.hy926 cell senescence by regulating the cell cycle in an atherosclerosis model. Mol Med Rep 17(1):257–263. https://doi.org/10.3892/mmr.2017.7872
Tan X, Fu Y, Chen L, Lee W, Lai Y, Rezaei K, Tabbara S, Latham P, Teal CB, Man YG, Siegel RS, Brem RF, Fu SW (2016) miR-671-5p inhibits epithelial-to-mesenchymal transition by downregulating FOXM1 expression in breast cancer. Oncotarget 7(1):293–307. https://doi.org/10.18632/oncotarget.6344
Torres N, Guevara-Cruz M, Velázquez-Villegas LA, Tovar AR (2015) Nutrition and atherosclerosis. Arch Med Res 46(5):408–426. https://doi.org/10.1016/j.arcmed.2015.05.010
Verduci L, Strano S, Yarden Y, Blandino G (2019) The circRNA-microRNA code: emerging implications for cancer diagnosis and treatment. Mol Oncol 13(4):669–680. https://doi.org/10.1002/1878-0261.12468
Wei S, Zheng Y, Jiang Y, Li X, Geng J, Shen Y, Li Q, Wang X, Zhao C, Chen Y, Qian Z, Zhou J, Li W (2019) The circRNA circPTPRA suppresses epithelial-mesenchymal transitioning and metastasis of NSCLC cells by sponging miR-96-5p. EBioMedicine 44:182–193. https://doi.org/10.1016/j.ebiom.2019.05.032
Wei MY, Lv RR, Teng Z (2020) Circular RNA circHIPK3 as a novel circRNA regulator of autophagy and endothelial cell dysfunction in atherosclerosis. Eur Rev Med Pharmacol Sci 24(24):12849–12858. https://doi.org/10.26355/eurrev_202012_24187
Yuan Y, Xu L, Geng Z, Liu J, Zhang L, Wu Y, He D, Qu P (2021) The role of non-coding RNA network in atherosclerosis. Life Sci 265:118756. https://doi.org/10.1016/j.lfs.2020.118756
Zhang LL (2020) CircRNA-PTPRA promoted the progression of atherosclerosis through sponging with miR-636 and upregulating the transcription factor SP1. Eur Rev Med Pharmacol Sci 24(23):12437–12449. https://doi.org/10.26355/eurrev_202012_24039
Zhang HD, Jiang LH, Sun DW, Hou JC, Ji ZL (2018) CircRNA: a novel type of biomarker for cancer. Breast Cancer 25(1):1–7. https://doi.org/10.1007/s12282-017-0793-9
Zhang S, Song G, Yuan J, Qiao S, Xu S, Si Z, Yang Y, Xu X, Wang A (2020) Circular RNA circ_0003204 inhibits proliferation, migration and tube formation of endothelial cell in atherosclerosis via miR-370-3p/TGFβR2/phosph-SMAD3 axis. J Biomed Sci 27(1):11. https://doi.org/10.1186/s12929-019-0595-9
Zhu J, Liu B, Wang Z, Wang D, Ni H, Zhang L, Wang Y (2019) Exosomes from nicotine-stimulated macrophages accelerate atherosclerosis through miR-21-3p/PTEN-mediated VSMC migration and proliferation. Theranostics 9(23):6901–6919. https://doi.org/10.7150/thno.37357
Funding
No funding was received.
Author information
Authors and Affiliations
Contributions
XL contributed to the conception and design of the study, data acquisition, analysis and interpretation, and also drafted and critically revised the manuscript. XZ contributed to data collection, statistical analysis, and manuscript preparation. All the authors declare that they have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors declare that they have no competing interest.
Ethical Approval
This study was supported by the Ethics Committee of Hubei University of Traditional Chinese Medicine.
Consent to Participate and for Publication
All patients provided written informed consent before using serum for scientific research. And all patients agreed for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Luo, X., Zhou, X. CircRNA-PTPRA Knockdown Inhibits Atherosclerosis Progression by Repressing ox-LDL-Induced Endothelial Cell Injury via Sponging of miR-671-5p. Biochem Genet 61, 187–201 (2023). https://doi.org/10.1007/s10528-022-10256-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-022-10256-x