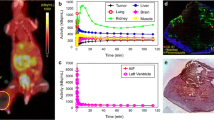
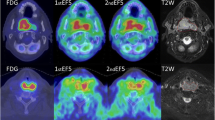
Abstract
Purpose
To identify the optimal threshold in 18F-fluoromisonidazole (FMISO) PET images to accurately locate tumor hypoxia by using electron paramagnetic resonance imaging (pO2 EPRI) as ground truth for hypoxia, defined by pO2 \(\le\) 10 mmHg.
Methods
Tumor hypoxia images in mouse models of SCCVII squamous cell carcinoma (n = 16) were acquired in a hybrid PET/EPRI imaging system 2 h post-injection of FMISO. T2-weighted MRI was used to delineate tumor and muscle tissue. Dynamic contrast enhanced (DCE) MRI parametric images of Ktrans and ve were generated to model tumor vascular properties. Images from PET/EPR/MRI were co-registered and resampled to isotropic 0.5 mm voxel resolution for analysis. PET images were converted to standardized uptake value (SUV) and tumor-to-muscle ratio (TMR) units. FMISO uptake thresholds were evaluated using receiver operating characteristic (ROC) curve analysis to find the optimal FMISO threshold and unit with maximum overall hypoxia similarity (OHS) with pO2 EPRI, where OHS = 1 shows perfect overlap and OHS = 0 shows no overlap. The means of dice similarity coefficient, normalized Hausdorff distance, and accuracy were used to define the OHS. Monotonic relationships between EPRI/PET/DCE-MRI were evaluated with the Spearman correlation coefficient (\(\rho\)) to quantify association of vasculature on hypoxia imaged with both FMISO PET and pO2 EPRI.
Results
FMISO PET thresholds to define hypoxia with maximum OHS (both OHS = 0.728 \(\pm\) 0.2) were SUV \(\ge\) 1.4 \(\times\) SUVmean and SUV \(\ge\) 0.6 \(\times\) SUVmax. Weak-to-moderate correlations (|\(\rho\)|< 0.70) were observed between PET/EPRI hypoxia images with vascular permeability (Ktrans) or fractional extracellular-extravascular space (ve) from DCE-MRI.
Conclusion
This is the first in vivo comparison of FMISO uptake with pO2 EPRI to identify the optimal FMISO threshold to define tumor hypoxia, which may successfully direct hypoxic tumor boosts in patients, thereby enhancing tumor control.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the first author or corresponding author on reasonable request.
References
Hockel M, Vaupel P. Tumor hypoxia: definitions and current clinical, biologic, and molecular aspects. J Natl Cancer Inst. 2001;93(4):266–76.
Vaupel P, Mayer A. Hypoxia in cancer: significance and impact on clinical outcome. Cancer Metastasis Rev. 2007;26(2):225–39.
Overgaard J. Hypoxic radiosensitization: adored and ignored. J Clin Oncol. 2007;25(26):4066–74.
Epel B, Maggio MC, Barth ED, et al. Oxygen-guided radiation therapy. Int J Radiat Oncol Biol Phys. 2019;103(4):977–84.
Dewhirst MW. Relationships between cycling hypoxia, HIF-1, angiogenesis and oxidative stress. Radiat Res. 2009;172(6):653–65.
Muz B, Azab F, Azab AK. The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy. Hypoxia (Auckl). 2015;3:83–92.
Schwarz G. Uber Desensibilisierung gegen Rontgen- und Radiumstrahlen. Munch Med Wochenschr. 1909;56:1217–8.
Crabtree HG, Cramer W. The action of radium on cancer cells II - some factors determining the susceptibility of cancer cells to radium. Proc R Soc Lond B. 1933;113(782):238–50.
Thomlinson RH, Gray LH. The histological structure of some human lung cancers and the possible implications for radiotherapy. Br J Cancer. 1955;9:539–63.
Kishimoto S, Saito K, Enomoto A, et al. Pulsed electron paramagnetic resonance imaging: applications in the studies of tumor physiology. Antioxid Redox Signal. 2018;28(15):1378–93.
Epel B, Redler G, Tormyshev V, Halpern HJ. Towards human oxygen images with electron paramagnetic resonance imaging. Adv Exp Med Biol. 2016;876:363–9.
Krohn KA, Link JM, Mason RP. Molecular imaging of hypoxia. J Nucl Med. 2008;49(Suppl 2):129S-S148.
Lopci E, Grassi I, Chiti A, et al. PET radiopharmaceuticals for imaging of tumor hypoxia: a review of the evidence. Am J Nucl Med Mol Imaging. 2014;4:365–84.
Vera P, Thureau S, Chaumet-Riffaud P, et al. Phase II Study of a radiotherapy total dose increase in hypoxic lesions identified by 18F-misonidazole PET/CT in patients with non-small cell lung carcinoma (RTEP5 study). J Nucl Med. 2017;58(7):1045–53.
Riaz N, Sherman E, Pei X, et al. Precision radiotherapy: reduction in radiation for oropharyngeal cancer in the 30 ROC Trial. J Natl Cancer Inst. 2021;113(6):742–51.
Fleming IN, Manavaki R, Blower PJ, et al. Imaging tumour hypoxia with positron emission tomography. Br J Cancer. 2015;112(2):238–50.
Ferini G, Valenti V, Tripoli A, et al. Lattice or oxygen-guided radiotherapy: what if they converge? Possible future directions in the era of immunotherapy. Cancers (Basel). 2021;13(13):3290.
Lindblom E, Dasu A, Uhrdin J, et al. Defining the hypoxic target volume based on positron emission tomography for image guided radiotherapy - the influence of the choice of the reference region and conversion function. Acta Oncol. 2017;56(6):819–25.
Chang JH, Wada M, Anderson NJ, et al. Hypoxia-targeted radiotherapy dose painting for head and neck cancer using (18)F-FMISO PET: a biological modeling study. Acta Oncol. 2013;52(8):1723–9.
de Figueiredo HB, Zacharatou C, Galland-Girodet S, et al. Hypoxia imaging with [18F]-FMISO-PET for guided dose escalation with intensity-modulated radiotherapy in head-and-neck cancers. Strahlenther Onkol. 2015;191(3):217–24.
Hendrickson K, Phillips M, Smith W, Peterson L, Krohn K, Rajendran J. Hypoxia imaging with [F-18] FMISO-PET in head and neck cancer: potential for guiding intensity modulated radiation therapy in overcoming hypoxia-induced treatment resistance. Radiother Oncol. 2011;101(3):369–75.
Dewhirst MW, Cao YT, Moeller B. Cycling hypoxia and free radicals regulate angiogenesis and radiotherapy response. Nat Rev Cancer. 2008;8(6):425–37.
Kim H, Epel B, Sundramoorthy S, et al. Development of a PET/EPRI combined imaging system for assessing tumor hypoxia. J Instrum. 2021;16(P03031). https://doi.org/10.1088/1748-0221/16/03/p03031.
Elas M, Bell R, Hleihel D, et al. Electron paramagnetic resonance oxygen image hypoxic fraction plus radiation dose strongly correlates with tumor cure in FSa fibrosarcomas. Int J Radiat Oncol Biol Phys. 2008;71(2):542–9.
Lim JL, Berridge MS. An efficient radiosynthesis of [18F]fluoromisonidazole. Appl Radiat Isot. 1993;44(8):1085–91.
Gertsenshteyn I, Epel B, Barth E, et al. improving tumor hypoxia location in 18F-misonidazole PET with dynamic contrast-enhanced MRI using quantitative electron paramagnetic resonance partial oxygen pressure images. Radiol Imaging Cancer. 2021;3(2):e200104. https://doi.org/10.1148/rycan.2021200104.
Tofts PS, Brix G, Buckley DL, et al. Estimating kinetic parameters from dynamic contrast-enhanced T1-weighted MRI of a diffusable tracer: standardized quantities and symbols. J Magn Reson Imaging. 1999;10:223–32.
Epel B, Halpern HJ. EPR oxygen imaging workflow with MATLAB Image Registration Toolbox. Appl Magn Reson. 2021;52(10):1311–9.
Gertsenshteyn I, Giurcanu M, Vaupel P, Halpern H. Biological validation of electron paramagnetic resonance (EPR) image oxygen thresholds in tissue. J Physiol. 2021;599(6):1759–67.
Metz CE. Basic principles of ROC analysis. Semin Nucl Med. 1978;8(4):283–98.
Grkovski M, Schöder H, Lee N, et al. multiparametric imaging of tumor hypoxia and perfusion with 18F-fluoromisonidazole dynamic PET in head and neck cancer. J Nucl Med. 2017;58(7):1072–80.
Thorwarth D, Eschmann SM, Paulsen F, Alber M. A kinetic model for 18F-Fmiso PET data to analyse tumour hypoxia. Phys Med Biol. 2005;50(10):2209–24.
Horsman MR, Vaupel P. Pathophysiological basis for the formation of the tumor microenvironment. Front Oncol. 2016;6(66). https://doi.org/10.3389/fonc.2016.00066.
Krock B, Skuli N, Simon CM. Hypoxia-induced angiogenesis: good and evil. Genes Cancer. 2011;2(12):1117–33.
Karth J, Ferrer FA, Perman E, et al. Coexpression of hypoxia-inducible factor 1-alpha and vascular endothelial growth factor in Wilms’ tumor. J Pediatr Surg. 2000;35(12):1749–53.
Simoncic U, Leibfarth S, Welz S, et al. Comparison of DCE-MRI kinetic parameters and FMISO-PET uptake parameters in head and neck cancer patients. Med phys. 2017;44(6):2358–68.
Carmona-Bozo JC, Manavaki R, Woitek R, et al. Hypoxia and perfusion in breast cancer: simultaneous assessment using PET/MR imaging. Eur Radiol. 2021;31(1):333–44.
Acknowledgements
We would like to thank the University of Chicago’s Cyclotron Facility, the Integrated Small Animal Imaging Research Resource, and the Human Tissue Resource Center. We would also like to thank Dr. Scott Trinkle for his helpful comments after reading over the manuscript, Dr. Nicole Cipriani’s expertise in pathology, and Dr. Hannah Zhang’s advice in early histological imaging sample preparation.
Funding
This study was funded by National Institutes of Health grants R01 CA098575, R01 CA 236385, P30 CA014599, P41 EB002034, F31 CA254223, S10 OD025265, and R01 EB029948.
Author information
Authors and Affiliations
Contributions
The following authors contributed to acquiring data: Inna Gertsenshteyn, Boris Epel, Eugene Barth, John Lukens, Xiaobing Fan, Hsui-Ming Tsai, Lara Leoni, Heejong Kim, Marta Zamora, Erica Markiewicz, Subramanian Sundramoorthy, Richard Freifelder, Mohammed Bhuiyan, and Anna Kucharski. The following authors contributed to the conception and design of the data: Inna Gertsenshteyn, Boris Epel, Brian Roman, Gregory Karczmar, Chien-Min Kao, Howard Halpern, Chin-Tu Chen. The following authors contributed to analyzing and interpreting data: Inna Gertsenshteyn, Amandeep Ahluwalia, Mihai Giurcanu, Brian Roman, Gregory Karczmar, Howard Halpern, Chin-Tu Chen. The first draft of the manuscript was written by Inna Gertsenshteyn, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Conflict of interest
Author Chin-Tu Chen (CTC) is a PI for the following grants from NIH/NIBIB (R01 EB022388 and R01 EB029948), NIH/NIDA (R01 DA044760), and NIH/NCI (P30 CA14599 facility). CTC receives a personal fee from the American Institute of Physics for editorial responsibilities. Other relationships include institute licenses patents (CTC is a co-inventor) to RefleXion Medical, Inc. and Incom. CTC is a co-founder and on the board of directors of EVO Worldwide LLC and AEPX Imaging, Inc. Author Howard Halpern (HH) is a PI for the following grants from NIH/NCI (R01 CA098575 and P30 CA014599) and NIH/NIBIB (P41 EB002034). HH also holds two US patents (8,664,955 and 9,392,957) to him and one (9,392,957) to author Boris Epel (BE) for aspects of the pO2 imaging technology; HH and BE are also members of a start-up company O2M to market the pO2 imaging technology in preclinical models. No other potential conflicts of interest relevant to this article exist. The rest of the authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Preclinical Imaging.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gertsenshteyn, I., Epel, B., Ahluwalia, A. et al. The optimal 18F-fluoromisonidazole PET threshold to define tumor hypoxia in preclinical squamous cell carcinomas using pO2 electron paramagnetic resonance imaging as reference truth. Eur J Nucl Med Mol Imaging 49, 4014–4024 (2022). https://doi.org/10.1007/s00259-022-05889-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-022-05889-4