Abstract
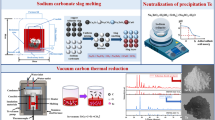
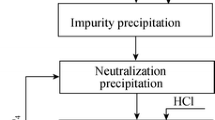
Copper telluride slag from copper smelting process is an important source of tellurium (Te). In this work, an efficient and economical hydrometallurgical process for extracting Te from copper telluride slag was proposed. First, H2SO4 and NaClO3 were demonstrated to be effective for separating Te and Cu, and the leaching efficiency of Cu(II) reached 99.44 pct, while that of Te(IV) was only 3.00 pct. Tellurium oxide in the oxidative acid leaching residue was dissolved in NaOH solution, and Na2S was added as precipitant to purify alkali leaching solution. Valuable metals (Bi, Se, Pb, Sb, Ag, Au) were enriched in alkaline leaching residue. Furthermore, the separation of Te and Se was realized by adding H2O2 in alkali leaching solution, the precipitation efficiency of Te(VI) was up to 99.43 pct. Finally, Te(VI) was reduced by Na2S-Na2SO3, and the reduction efficiency was 99.85 pct under optimized conditions. The results show that 96.18 pct of Te can be extracted as elemental Te powder with a purity of 99.98 pct, indicating that the process achieves an efficient extraction efficiency of Te from copper telluride slag.













Similar content being viewed by others
References
L. Xu, Y.H. Xiong, G.G. Zhang, F.Y. Zhang, Y.X. Yang, Z.S. Hua, Y.P. Tian, J.L. You, Z. Zhao: J. Clean. Prod, 2020, vol. 272, art. No.122723.
S.J. Wang: JOM, 2011, vol. 63, pp. 90–93.
K. Zweibel: Science, 2010, vol. 328, pp. 699–701.
H.J. Zhang, C.X. Liu, X.L. Qi, X. Dai, Z. Fang, and S.C. Zhang: Nat. Phys, 2009, vol. 5, pp. 438–42.
H. Choi, K. Jeong, J. Chae, H. Park, J. Baeck, T.H. Kim, J.Y. Song, J. Park, K.H. Jeong, and M.H. Cho: Nano Energy, 2018, vol. 47, pp. 374–84.
Y.K. Zhu, J. Guo, L. Chen, S.W. Gu, Y.X. Zhang, Q. Shan, J. Feng, Z.H. Ge: Chem. Eng. J, 2021, vol. 407, art. No.126407.
Z. Pan, A. Ueda, R. Mu, and S.H. Morgan: J. Lumin, 2007, vol. 126, pp. 251–56.
W.C. Wang, B. Zhou, S.H. Xu, Z.M. Yang, and Q.Y. Zhang: Prog. Mater. Sci, 2019, vol. 101, pp. 90–171.
M. Lankhorst, B. Ketelaars, and R. Wolters: Nat. Mater, 2005, vol. 4, pp. 347–52.
G. Kavlak and T.E. Graedel: Resour. Conserv. Recy, 2013, vol. 76, pp. 21–26.
Y.W. Li, X.X. Fan, and K.B. Yang: Chem. Eng. Commun., 2020, vol. 9, pp. 1–7.
F.M. Makuei and G. Senanayake: Min. Eng, 2018, vol. 115, pp. 79–87.
W.F. Liu, R. Jia, B. Sun, D. Zhang, L. Chen, T. Yang, and S. Lu: J. Clean. Prod, 2020, vol. 264, p. 121637.
Z.Y. Ma, H.Y. Yang, S.T. Huang, Y. Lü, and L. Xiong: Int. J. Min. Met. Mater, 2015, vol. 22, pp. 582–88.
S. Rao, Y. Liu, D.X. Wang, H.Y. Cao, W. Zhu, R. Yang, L.J. Duan, and Z.Q. Liu: J. Clean. Prod., 2021, vol. 278, p. 123989.
M. Mokmeli, D. Dreisinger, B: Hydrometallurgy, 2014, vol. 147-148, pp. 20–29.
W.A. Dutton and W.C. Cooper: Chem. Rev, 1966, vol. 66, pp. 657–75.
M. Mokmeli, D.B. Dreisinger, B. Wassink, and B. Difley: Int. J. Chem. Kinet, 2016, vol. 48, pp. 204–11.
J.E. Hoffmann: JOM, 1989, vol. 41, pp. 33–38.
L. Xu, Y.H. Xiong, Y. Song, G.G. Zhang, F.Y. Zhang, Y.X. Yang, Z.S. Hua, Y.P. Tian, J.L. You, and Z. Zhao: Hydrometallurgy, 2020, vol. 196, p. 105436.
U.B. Barache, B.T. Khogare, A.B. Shaikh, S.A. Deodware, B.N. Kokare, A.G.P. Rodriguez, T.N. Lokhande, and S.H. Gaikwad: Chem. Data. Collect., 2019, vol. 19, p. 100173.
Y.H.Q. Yu, Y.H. Chu, T. Zhang, L.B. Yu, D.Y. Yang, F.X. Qiu, and D.S. Yuan: Hydrometallurgy, 2018, vol. 177, pp. 1–8.
Z. Zhao, Y.H. Xiong, X.K. Cheng, X. Hou, Y.X. Yang, Y.P. Tian, J.L. You, and L. Xu: J. Hazard. Mater, 2020, vol. 393, p. 122378.
D. Park, H. Ju, T. Oh, and J. Kim: Cryst. Eng. Commun., 2019, vol. 21, pp. 1555–63.
G.W. She, X.H. Zhang, W.S. Shi, Y. Cai, N. Wang, P. Liu, and D.M. Chen: Cryst. Growth Des, 2008, vol. 8, pp. 1789–91.
Y.K. Shen, H.B. Zhang, H.Z. Cao, L.K. Wu, and G.Q. Zheng: Hydrometallurgy, 2019, vol. 189, p. 105121.
S.K. Lower: 1998. Reference text, Simon Fraser University.
Z.M. Sun and Y.J. Zheng: T. Nonferr. Metal. Soc, 2011, vol. 21, pp. 665–72.
L.X. Shao, J. Diao, C.Q. Ji, G. Li: Hydrometallurgy, 2020, vol. 191, art. No. 105205.
J.A. Dean, N.A. Lange: Lange's Handbook of Chemistry, McGraw-Hill, 1999.
A. Alemi, S.W. Joo, Y. Hanifehpour, A. Khandar, A. Morsali, and B.K. Min: J. Nanomater, 2011, vol. 2011, pp. 1–5.
Y. Liu, Y.J. Zheng, and Z.M. Sun: Rare Met, 2014, vol. 33, pp. 479–84.
M.S.S. Alireza, M. Mohammad, and A.F. Rashchi: Metall. Mater. Trans. B, 2020, vol. 51B, pp. 2555–75.
S. Aydogan: Chem. Eng. J, 2006, vol. 123, pp. 65–70.
Z.L. Dong, T. Jiang, B. Xu, J.K. Yang, Y.Z. Chen, Q. Li, and Y.B. Yang: Chem. Eng. J, 2020, vol. 393, p. 124762.
Z.P. Xu, X.Y. Guo, Q.H. Tian, D. Li, Z. Zhang, and L. Zhu: Hydrometallurgy, 2020, vol. 193, p. 105316.
Z.P. Xu, X.Y. Guo, D. Li, Q.H. Tian, L. Zhu: Hydrometallurgy, 2020, vol. 191, art. No. 105219.
J.C. Lee, K. Kurniawan, K.W. Chung, and S. Kim: Met. Mater. Int, 2020, vol. 27, pp. 2160–87.
S.J. Lu, J. Li, D.L. Chen, W. Sun, J. Zhang, and Y. Yang: J. Clean. Prod., 2020, vol. 261, p. 121214.
P. Ostermeyer, L. Bonin, K. Folens, F. Verbruggen, C. García-Timermans, K. Verbeken, K. Rabaey, and T. Hennebel: J. Hazard. Mater, 2021, vol. 409, p. 124418.
B. Hu, T.Z. Yang, W.F. Liu, D.C. Zhang, and L. Chen: Trans. Nonferr. Metal. Soc., 2019, vol. 26, pp. 2411–21.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (No. 51374185).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shen, Y., Xu, S., Feng, W. et al. An Efficient Hydrometallurgical Process for Extracting Tellurium From Copper Telluride Slag. Metall Mater Trans B 53, 2838–2851 (2022). https://doi.org/10.1007/s11663-022-02568-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-022-02568-5