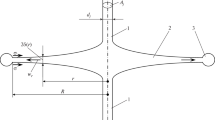
Abstract
A comparative study of macro- and micromixing in a 1-L laboratory beaker, a 0.5-L flask, and a microreactor with intensively swirled flows (two-stage jet vortex microreactor (TSJVMR)) is presented. Stirring in the beaker and flask is accomplished with a magnetic stirrer. Mixing is studied with two methods: a 4 M NaOH solution is added to a 0.05 M HCl solution in macromixing studies and the iodide–iodate method is used in micromixing studies. Flows are photographed to establish the mixing behavior in all of the cases and to calculate the mixing zone in the TSJVMR. The results of our studies show that when even macromixing is unsatisfactory with the use of a magnetic stirrer, and the quality of micromixing (as indicated by the segregation index) in the 0.5-L flask equipped with a magnetic follower is 250 times lower than that in the TSJVMR. The results of this study have a significant impact on our understanding of the effects that micromixing conditions have on the solution synthesis of nanoscale particles.




Similar content being viewed by others
REFERENCES
Teychené, S., Rodríguez-Ruiz, I., and Ramamoorthy, R.K., Reactive crystallization: From mixing to control of kinetics by additives, Curr. Opin. Colloid Interface Sci., 2020, vol. 46, pp. 1–19. https://doi.org/10.1016/j.cocis.2020.01.003
Nightingale, A.M. and DeMello, J.C., Segmented flow reactors for nanocrystal synthesis, Adv. Mater., 2013, vol. 25, pp. 1813–1821. https://doi.org/10.1002/adma.201203252
Patil, S., Kate, P.R., Deshpande, J.B., and Kulkarni, A.A., Quantitative understanding of nucleation and growth kinetics of silver nanowires, Chem. Eng. J., 2021, vol. 414, Article 128711. https://doi.org/10.1016/j.cej.2021.128711
Kudryashova, Yu.S., Zdravkov A.V., and Abiev R.Sh., Synthesis of yttrium–aluminum garnet using a microreactor with impinging jets, Glass Phys. Chem., 2021, vol. 47, no. 3, pp. 260–264. https://doi.org/10.1134/S108765962103007X
Abiev, R.Sh., Zdravkov, A.V., Kudryashova, Yu.S., et al., Synthesis of calcium fluoride nanoparticles in a microreactor with intensely swirling flows, Russ. J. Inorg. Chem., 2021, vol. 66, no. 7, p. 1047. https://doi.org/10.1134/S0036023621070020
Sidorov, V.I. and Malyavskii, N.I., Stroit. Mater., Oborud., Tekhnol. XXI Veka, 2012, No. 1, p. 42.
Chemical Methods for Producing Ceramic and Polymer Nanomaterials from the Liquid Phase: A Study Guide, Luchinin, V.V. and Shilova, O.A., Eds., St. Petersburg: SPbGETU “LETI,” 2013.
Proskurina, O.V., Abiev, R.S., Danilovich, D.P., Panchuk, V.V., Semenov, V.G., Nevedomsky, V.N., and Gusarov. V.V., Formation of nanocrystalline BiFeO3 during heat treatment of hydroxides co-precipitated in an impinging-jets microreactor, Chem. Eng. Proc.: Proc. Intens., 2019, vol. 143, Article 107598. https://doi.org/10.1016/j.cep.2019.107598
Tacsi, K., Joo, A., Pusztai, E., Domokos, A., Nagy, Z.K., Marosi, G., and Pataki, H., Development of a triple impinging jet mixer for continuous antisolvent crystallization of acetylsalicylic acid reaction mixture, Chem. Eng. Proc.: Proc. Intens., 2021, vol. 165, Article 108446. https://doi.org/10.1016/j.cep.2021.108446
Johnson, B.K. and Prud’homme, R.K., Chemical processing and micromixing in confined impinging jets, AIChE J., 2003, vol. 49, pp. 2264–2282.
Ravi Kumar, D.V., Prasad, B.L.V., and Kulkarni, A.A., Impinging jet micromixer for flow synthesis of nanocrystalline MgO: Role of mixing/impingement zone, Ind. Eng. Chem. Res., 2013, vol. 52, pp. 17376–17382. https://doi.org/10.1021/ie402012x
Marchisio, D.L., Rivautella, L., and Barresi, A.A., Design and scale-up of chemical reactors for nanoparticle precipitation, AIChE J., 2006, vol. 52, p. 1877–1887.
Nightingale, A.M., PhD Thesis, Imperial College London, Department of Chemistry, 2010.
Zhao, C.-X., He, L., Qiao, S.Z., and Middelberg, A.P.J., Nanoparticle synthesis in microreactors, Chem. Eng. Sci., 2011, vol. 66, p. 1463–1479. https://doi.org/10.1016/j.ces.2010.08.039
Bałdyga, J. and Bourne, J.R., Simplification of micromixing calculations. I. Derivation and application of new model, Chem. Eng. J., 1989, vol. 42, p. 83–92.
Morozov, M.I., Mezentseva, L.P., and Gusarov, V.V., Mechanism of formation of Bi4Ti3O12, Russ. J. Gen. Chem., 2002, vol. 72, no. 7, pp. 1038–1040.
Morozov, M.I., Lomanova, N.A., and Gusarov, V.V., Specific features of BiFeO3 formation in a mixture of bismuth(III) and iron(III) oxides, Russ. J. Gen. Chem., 2003, vol. 73, no. 11, pp. 1676–1680. https://doi.org/10.1023/B:RUGC.0000018640.30953.70
Artamonova, O.V., Al’myasheva, O.V., Mittova, I.Ya., et al., Hydrothermal synthesis of zirconia-based nanocrystals in the ZrO2–In2O3 system, Russ. J. Inorg. Chem., 2004, vol. 49, no. 11, p. 1657–1660.
Krzhizhanovskaya, M., Filatov, S., Gusarov, V.V., et al., Aurivillius phases in the Bi4Ti3O12/BiFeO3 system: Thermal behaviour and crystal structure, Z. Anorg. Allg. Chem., 2005, vol. 631, no. 9, pp. 1603–1608.
Tugova, E.A., Popova, V.F., Zvereva, I.A., et al., Mechanism and kinetics of formation of La2SrFe2O7 and Nb2SrFe2O7, Russ. J. Gen. Chem., 2007, vol. 77, no. 6, p. 979–982.
Almjasheva, O.V. and Gusarov, V.V., Hydrothermal synthesis of nanosized and amorphous alumina in the ZrO2–Al2O3–H2O system, Russ. J. Inorg. Chem., 2007, vol. 52, no. 8, pp. 1194–1200. https://doi.org/10.1134/S0036023607080062
Chivilikhin, S.A., Popov, I.Yu., Svitenkov, A.I., et al., Dokl. Akad. Nauk, 2009, vol. 429, no. 2, p. 185.
Al’myashev, O.V. and Gusarov, V.V., Features of the phase formation in the nanocomposites, Russ. J. Gen. Chem., 2010, vol. 80, no. 3, pp. 385–390. https://doi.org/10.1134/S1070363210030023
Komlev, A.A., and Gusarov, V.V., Mechanism of the nanocrystals formation of the spinel structure in the MgO–Al2O3–H2O system under the hydrothermal conditions, Russ. J. Gen. Chem., 2011, vol. 81, no. 11, p. 2222. https://doi.org/10.1134/S1070363211110028
Kirillova, S.A., Al’myashev, V.I., and Gusarov, V.V., Spinodal decomposition in the SiO2–TiO2 system and hierarchically organized nanostructures formation, Nanosyst.: Phys., Chem., Math., 2012, vol. 3, no. 2, p. 100.
Smirnov, A.V., Fedorov, B.A., Tomkovich, M.V., et al., Core-shell nanoparticles forming in the ZrO2–Gd2O3–H2O system under hydrothermal conditions, Dokl. Phys. Chem., 2014, vol. 456, no. 1, pp. 71–73. https://doi.org/10.1134/S0012501614050042
Popkov, V.I., Almjasheva, O.V., and Gusarov, V.V., The investigation of the structure control possibility of nanocrystalline yttrium orthoferrite in its synthesis from amorphous powders, Russ. J. Appl. Chem., 2014, vol. 87, pp. 1417–1421. https://doi.org/10.1134/S1070427214100048
Popkov V.I., Almjasheva O.V., Schmidt M.P., et al., Formation mechanism of nanocrystalline yttrium orthoferrite under heat treatment of the coprecipitated hydroxides, Russ. J. Gen. Chem., 2015, vol. 85, pp. 1370–1375. https://doi.org/10.1134/S107036321506002X
Almjasheva, O.V. and Gusarov, V.V., Prenucleation formations in control over synthesis of CoFe2O4 nanocrystalline powders, Russ. J. Appl. Chem., 2016, vol. 89, no. 6, pp. 851–856. https://doi.org/10.1134/S107042721606001X
Komlev, A.A., Panchuk, V.V., Semenov V.G., et al., Effect of the sequence of chemical transformations on the spatial segregation of components and formation of periclase–spinel nanopowders in the MgO–Fe2O3–H2O system, Russ. J. Appl. Chem., 2016, vol. 89, no. 12, pp. 1932–1938. https://doi.org/10.1134/S1070427216120028
Proskurina, O.V., Nogovitsin, I.V., Il’ina, T.S., et al., Formation of BiFeO3 nanoparticles using impinging jets microreactor, Russ. J. Gen. Chem., 2018, vol. 88, no. 10, pp. 2139–2143. https://doi.org/10.1134/S1070363218100183
Proskurina, O.V., Sokolova, A.N., Sirotkin, A.A., et al., Role of hydroxide precipitation conditions in the formation of nanocrystalline BiFeO3, Russ. J. Inorg. Chem., 2021, vol. 66, no. 2, pp. 163–169. https://doi.org/10.1134/S0036023621020157
Gyurik, L., Ulbert, Zs., Molnár, B., et al., CFD based nozzle design for a multijet mixer, Chem. Eng. Proc.: Proc. Intens., 2020, vol. 157, Article 108121. https://doi.org/10.1016/j.cep.2020.108121
Chen, J., Jiang, W., and Liu, Y., Study on energy distribution characteristics of cyclone in Laval nozzle, Chem. Eng. Proc.: Proc. Intens., 2020, vol. 157, Article 108149. https://doi.org/10.1016/j.cep.2020.108149
Guichardon, P. and Falk, L., Characterisation of micromixing efficiency by the iodide–iodate reaction system. Part I: Experimental procedure, Chem. Eng. Sci., 2000, vol. 55, pp. 4233–4243. https://doi.org/10.1016/S0009-2509(00)00068-3
Falk L. and Commenge, J.-M., Performance comparison of micromixers, Chem. Eng. Sci., 2010, vol. 65, pp. 405–411. https://doi.org/10.1016/j.ces.2009.05.045
Abiev, R.Sh. and Sirotkin A.A., Influence of hydrodynamic conditions on micromixing in microreactors with free impinging jets, Fluids, 2020, vol. 5, p. 179. https://doi.org/10.3390/fluids5040179
Schaer, E., Guichardon, P., Falk, L., and Plasari, E., Determination of local energy dissipation rates in impinging jets by a chemical reaction method, Chem. Eng. J., 1999, vol. 72, pp. 125–138.
Baldyga, J. and Bourne, J. R., Chem. Eng. J., 1990, vol. 45, pp. 25–31.
Ditl, P., Šulc, R., Pešava, V., Jašíkova, D., Kotek, M., Kopecký, V., and Kysela, B., Local turbulent energy dissipation rate in an agitated vessel: Experimental and turbulence scaling, Theor. Found. Chem. Eng., 2018, vol. 52, p. 122–134.
Alopaeus, V., Koskinen, J., and Keskinen, K.I., Simulation of the population balances for liquid–liquid systems in a nonideal stirred tank. Part 1: Description and qualitative validation of the model, Chem. Eng. Sci., 1999, vol. 54, p. 5887–5899.
ACKNOWLEDGMENTS
We are grateful to D.A. Potekhin for assistance with the photography in the studies of macromixing in a laboratory beaker.
Funding
The work was supported by the Russian Science Foundation (project no. 20-63-47016).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by A. Kukharuk
Rights and permissions
About this article
Cite this article
Abiev, R.S., Makusheva, I.V. Effect of Macro- and Micromixing on Processes Involved in Solution Synthesis of Oxide Particles in Mocroreactors with Intensively Swirling Flows. Theor Found Chem Eng 56, 141–151 (2022). https://doi.org/10.1134/S0040579522020014
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0040579522020014