Abstract
Background
Mitophagy protects against cerebral ischemia/reperfusion (CI/R)–induced neuronal apoptosis via mitochondrial clearance. Although taurine-upregulated gene 1 (lncRNA TUG1) has been proposed to be involved in the neuronal apoptosis evoked by CI/R, its specific role in mitophagy during the progression of CI/R injury remains unknown.
Methods
The CI/R rat model was established using middle cerebral artery occlusion/reperfusion (MCAO/R). Human neuroblastoma cell line SH-SY5Y was subjected to oxygen-glucose deprivation and reoxygenation (OGD/R). Ubiquitination assay, co-immunoprecipitation assay, RNA pull-down, and RNA immunoprecipitation were used to determine the interplay among TUG1, sirtuin 1 (SIRT1), and F-box and WD repeat domain-containing 7 (FBXW7).
Results
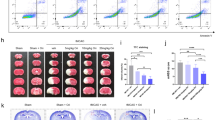
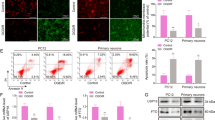
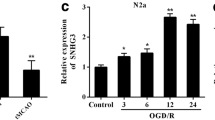
The upregulation of the TUG1 level and downregulation of the mitophagy were observed in both MCAO/R-treated rats and OGD/R-treated cells. The administration of si-TUG1 (a siRNA directed against TUG1) potentiated mitophagy and suppressed neuronal apoptosis in OGD/R-treated cells. However, the neuroprotective effect of si-TUG1 was reversed by mitophagy inhibitor or SIRT1 knockdown in vitro. Functionally, TUG1 enhanced FBXW7-mediated SIRT1 ubiquitination by upregulating FBXW7 expression. The overexpression of FBXW7 abrogated the si-TUG1-reinforced mitophagy by decreasing SIRT1 expression, thus aggravating neuronal apoptosis in the OGD/R+si-TUG1-treated cells. In rats with MCAO/R, the interference of TUG1 clearly decreased neuronal apoptosis, lessened the infarct volume, and relieved the neurological deficits.
Conclusion
TUG1 knockdown promotes SIRT1-induced mitophagy by suppressing FBXW7-mediated SIRT1 degradation, thus relieving the neuronal apoptosis induced by CI/R injury.
Graphical abstract
LncRNA TUG1 promotes neuronal apoptosis through inhibition of mitophagy. TUG1 decreased SIRT1 expression by promoting FBXW7-mediated SIRT1 ubiquitination. FBXW7/SIRT1 axis mediated the effect of TUG1 on OGD/R-induced neuronal apoptosis by regulating mitophagy.







Similar content being viewed by others
References
Bao MH, et al. Long non-coding RNAs in ischemic stroke. Cell Death Dis. 2018;9(3):281.
Barthels D, Das H. Current advances in ischemic stroke research and therapies. Biochim Biophys Acta Mol basis Dis. 2020;1866(4):165260.
Bhattacharya A, et al. Mitochondrial-targeted methionine sulfoxide reductase overexpression increases the production of oxidative stress in mitochondria from skeletal muscle. Aging Pathobiol Ther. 2020;2(1):45–51.
Chen S, et al. LncRNA TUG1 sponges microRNA-9 to promote neurons apoptosis by up-regulated Bcl2l11 under ischemia. Biochem Biophys Res Commun. 2017;485(1):167–73.
Di Sante G, et al. Loss of Sirt1 promotes prostatic intraepithelial neoplasia, reduces mitophagy, and delays PARK2 translocation to mitochondria. Am J Pathol. 2015;185(1):266–79.
Di Y, et al. Methylene blue reduces acute cerebral ischemic injury via the induction of mitophagy. Mol Med. 2015;21(1):420–9.
Fang EF, et al. Defective mitophagy in XPA via PARP-1 hyperactivation and NAD(+)/SIRT1 reduction. Cell. 2014;157(4):882–96.
Garcia JH, et al. Neurological deficit and extent of neuronal necrosis attributable to middle cerebral artery occlusion in rats. Statistical validation. Stroke. 1995;26(4):627–34 discussion 635.
He Q, et al. Resveratrol alleviates cerebral ischemia/reperfusion injury in rats by inhibiting NLRP3 inflammasome activation through Sirt1-dependent autophagy induction. Int Immunopharmacol. 2017;50:208–15.
Hong T, et al. Knocking down peroxiredoxin 6 aggravates cerebral ischemia-reperfusion injury by enhancing mitophagy. Neuroscience. 2022;482:30–42.
Klionsky DJ, et al. Autophagy in major human diseases. EMBO J. 2021;40(19):e108863.
Lazarou M, et al. The ubiquitin kinase PINK1 recruits autophagy receptors to induce mitophagy. Nature. 2015;524(7565):309–14.
Lemasters JJ. Selective mitochondrial autophagy, or mitophagy, as a targeted defense against oxidative stress, mitochondrial dysfunction, and aging. Rejuvenation Res. 2005;8(1):3–5.
Liang W, et al. Preactivation of Notch1 in remote ischemic preconditioning reduces cerebral ischemia-reperfusion injury through crosstalk with the NF-κB pathway. J Neuroinflammation. 2019;16(1):181.
Lin Y, et al. Glucosamine promotes hepatitis B virus replication through its dual effects in suppressing autophagic degradation and inhibiting MTORC1 signaling. Autophagy. 2020;16(3):548–61.
Lu H, Wang B. SIRT1 exerts neuroprotective effects by attenuating cerebral ischemia/reperfusion-induced injury via targeting p53/microRNA-22. Int J Mol Med. 2017;39(1):208–16.
Rabinstein AA, et al. Factors that may contribute to poor outcome despite good reperfusion after acute endovascular stroke therapy. Int J Stroke. 2019;14(1):23–31.
Rao G, Zhang W, Song S. MicroRNA-217 inhibition relieves cerebral ischemia/reperfusion injury by targeting SIRT1. Mol Med Rep. 2019;20(2):1221–9.
Shan W, et al. Long noncoding RNA TUG1 contributes to cerebral ischaemia/reperfusion injury by sponging mir-145 to up-regulate AQP4 expression. J Cell Mol Med. 2020;24(1):250–9.
Shen Z, et al. PARK2-dependent mitophagy induced by acidic postconditioning protects against focal cerebral ischemia and extends the reperfusion window. Autophagy. 2017;13(3):473–85.
Shen T, et al. Ube2v1-mediated ubiquitination and degradation of Sirt1 promotes metastasis of colorectal cancer by epigenetically suppressing autophagy. J Hematol Oncol. 2018;11(1):95.
Stegner D, Klaus V, Nieswandt B. Platelets as modulators of cerebral ischemia/reperfusion injury. Front Immunol. 2019;10:2505.
Tang BL. Sirt1 and the mitochondria. Mol Cell. 2016;39(2):87–95.
Um JH, Yun J. Emerging role of mitophagy in human diseases and physiology. BMB Rep. 2017;50(6):299–307.
Vosler PS, et al. Mitochondrial targets for stroke: focusing basic science research toward development of clinically translatable therapeutics. Stroke. 2009;40(9):3149–55.
Wei YS, et al. A functional polymorphism in the promoter of TUG1 is associated with an increased risk of ischaemic stroke. J Cell Mol Med. 2019;23(9):6173–81.
White BC, et al. Brain ischemia and reperfusion: molecular mechanisms of neuronal injury. J Neurol Sci. 2000;179(S 1-2):1–33.
Xian W, et al. Maresin 1 attenuates the inflammatory response and mitochondrial damage in mice with cerebral ischemia/reperfusion in a SIRT1-dependent manner. Brain Res. 2019;1711:83–90.
Xiao Z, et al. Blocking lncRNA H19-miR-19a-Id2 axis attenuates hypoxia/ischemia induced neuronal injury. Aging (Albany NY). 2019;11(11):3585–600.
Xiong ZJ, et al. Overexpression of TUG1 promotes neuronal death after cerebral infarction by regulating microRNA-9. Eur Rev Med Pharmacol Sci. 2018;22(21):7393–400.
Xu L, et al. Salidroside attenuates hypoxia/reoxygenation-induced human brain vascular smooth muscle cell injury by activating the SIRT1/FOXO3α pathway. Exp Ther Med. 2018;15(1):822–30.
Yan H, et al. Yap regulates gastric cancer survival and migration via SIRT1/Mfn2/mitophagy. Oncol Rep. 2018;39(4):1671–81.
Yan X, et al. Calycosin-7-O-β-D-glucoside attenuates OGD/R-induced damage by preventing oxidative stress and neuronal apoptosis via the SIRT1/FOXO1/PGC-1α pathway in HT22 cells. Neural Plast. 2019;2019:8798069–9.
Yeh CH, Bellon M, Nicot C. FBXW7: a critical tumor suppressor of human cancers. Mol Cancer. 2018;17(1):115.
Young TL, Matsuda T, Cepko CL. The noncoding RNA taurine upregulated gene 1 is required for differentiation of the murine retina. Curr Biol. 2005;15(6):501–12.
Yu L, et al. Ubiquitination-mediated degradation of SIRT1 by SMURF2 suppresses CRC cell proliferation and tumorigenesis. Oncogene. 2020;39(22):4450–64.
Yuan Y, et al. BNIP3L/NIX-mediated mitophagy protects against ischemic brain injury independent of PARK2. Autophagy. 2017;13(10):1754–66.
Zhang X, et al. Cerebral ischemia-reperfusion-induced autophagy protects against neuronal injury by mitochondrial clearance. Autophagy. 2013;9(9):1321–33.
Zhang F, et al. Novel oncogene COPS3 interacts with Beclin1 and Raf-1 to regulate metastasis of osteosarcoma through autophagy. J Exp Clin Cancer Res. 2018;37(1):135.
Zheng XW, et al. Buyang Huanwu decoction targets SIRT1/VEGF pathway to promote angiogenesis after cerebral ischemia/reperfusion injury. Front Neurosci. 2018;12:911.
Data availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Funding
This work was supported by the Medical Science and Technology Program of Henan Province of China (No. SB201901018), Joint Funds of the National Natural Science Foundation of China (No. U2004128), and Joint Project of Medical Science and Technology Program of Henan Province of China (No. 2018020070).
Author information
Authors and Affiliations
Contributions
LX & SC conceived and designed the study and drafted the manuscript. SX collected the data and PL contributed to the statistical analysis. HL interpreted the data and put forward the concept of the study and reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All animal experiments were carried out with the approval of the ethics committee of The First Affiliated Hospital of Zhengzhou University.
Consent for publication
The study was undertaken with the patient’s consent.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information

Supplementary Figure 1
The effect of TUG1 knockdown on cell apoptosis in OGD/R-treated SH-SY5Y cells. SH-SY5Y cells were grouped into control, OGD/R, OGD/R + si-TUG1, OGD/R + si-NC, OGD/R + si-TUG1 + mdivi-1 (a mitophagy inhibitor). The reoxygenation duration of OGD/R was set at 12 h and cells were incubated with 25 μmol/L mdivi-1 during reoxygenation. TUNEL assay was performed to evaluate cell apoptosis in SH-SY5Y cells. Scale bar=50 μm. (PNG 407 kb)

Supplementary Figure 2
The effect of FBXW7/SIRT1 axis on cell apoptosis in OGD/R-treated SH-SY5Y cells. SH-SY5Y cells were grouped into OGD/R + si-NC, OGD/R + si-TUG1, OGD/R + si-TUG1 + pcDNA, OGD/R + si-TUG1 + pcDNA-FBXW7, OGD/R + si-TUG1 + pcDNA-FBXW7 + pcDNA-SIRT1. The reoxygenation duration of OGD/R was performed for 12 h. TUNEL assay was carried out to assess the apoptosis of SH-SY5Y cells. Scale bar=50 μm. (PNG 904 kb)
Rights and permissions
About this article
Cite this article
Xue, Lx., Chen, Sf., Xue, Sx. et al. LncRNA TUG1 compromised neuronal mitophagy in cerebral ischemia/reperfusion injury by targeting sirtuin 1. Cell Biol Toxicol 38, 1121–1136 (2022). https://doi.org/10.1007/s10565-022-09700-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-022-09700-w