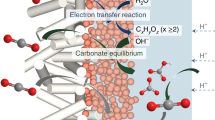
Abstract
Electrochemical CO2 reduction provides a promising route to the sustainable generation of valuable chemicals and fuels. Tandem catalysts enable sequential CO2-to-CO and CO-to-multicarbon (C2+) product conversions on complementary active sites, to produce high C2+ Faradaic efficiency (FE). Unfortunately, previous tandem catalysts exhibit poor management of CO intermediates, which diminishes C2+ FE. Here, we design segmented gas-diffusion electrodes (s-GDEs) in which a CO-selective catalyst layer (CL) segment at the inlet prolongs CO residence time in the subsequent C2+-selective segment, enhancing conversion. This phenomenon enables increases in both the CO utilization and C2+ current density for a Cu/Ag s-GDE compared to pure Cu, by increasing the *CO coverage within the Cu CL. Lastly, we develop a Cu/Fe-N-C s-GDE with 90% C2+ FE at C2+ partial current density (jC2+) exceeding 1 A cm−2. These results prove the importance of transport and establish design principles to improve C2+ FE and jC2+ in tandem CO2 reduction.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Source data are provided with this paper. All data generated and analysed during the present study are available from the authors upon reasonable request.
References
Jouny, M., Luc, W. W. & Jiao, F. A general techno-economic analysis of CO2 electrolysis systems. Ind. Eng. Chem. Res. 57, 2165–2177 (2018).
Nitopi, S. et al. Progress and perspectives of electrochemical CO2 reduction on copper in aqueous electrolyte. Chem. Rev. 119, 7610–7672 (2019).
Dinh, C.-T. et al. CO2 electroreduction to ethylene via hydroxide-mediated copper catalysis at an abrupt interface. Science 360, 783–787 (2018).
Gao, Y. et al. Recent advances in intensified ethylene production—a review. ACS Catal. 9, 8592–8621 (2019).
Montoya, J. H., Peterson, A. A. & Nørskov, J. K. Insights into C-C coupling in CO2 electroreduction on copper electrodes. ChemCatChem 5, 737–742 (2013).
Montoya, J. H., Shi, C., Chan, K. & Norskov, J. K. Theoretical insights into a CO dimerization mechanism in CO2 electroreduction. J. Phys. Chem. Lett. 6, 2032–2037 (2015).
Schouten, K. J. P., Kwon, Y., van der Ham, C. J. M., Qin, Z. & Koper, M. T. M. A new mechanism for the selectivity to C1 and C2 species in the electrochemical reduction of carbon dioxide on copper electrodes. Chem. Sci. 2, 1902–1909 (2011).
Schouten, K. J. P., Pérez Gallent, E. & Koper, M. T. M. Structure sensitivity of the electrochemical reduction of carbon monoxide on copper single crystals. ACS Catal. 3, 1292–1295 (2013).
Liu, X. et al. pH effects on the electrochemical reduction of CO2 towards C2 products on stepped copper. Nat. Commun. 10, 32 (2019).
Garza, A. J., Bell, A. T. & Head-Gordon, M. Mechanism of CO2 reduction at copper surfaces: pathways to C2 products. ACS Catal. 8, 1490–1499 (2018).
Resasco, J. et al. Promoter effects of alkali metal cations on the electrochemical reduction of carbon dioxide. J. Am. Chem. Soc. 139, 11277–11287 (2017).
Li, J. et al. Constraining CO coverage on copper promotes high-efficiency ethylene electroproduction. Nat. Catal. 2, 1124–1131 (2019).
Choi, C. et al. Highly active and stable stepped Cu surface for enhanced electrochemical CO2 reduction to C2H4. Nat. Catal. 3, 804–812 (2020).
Gu, Z. et al. Efficient electrocatalytic CO2 reduction to C2+ alcohols at defect-site-rich Cu surface. Joule 5, 429–440 (2021).
Zhong, M. et al. Accelerated discovery of CO2 electrocatalysts using active machine learning. Nature 581, 178–183 (2020).
Vasileff, A., Xu, C., Jiao, Y., Zheng, Y. & Qiao, S.-Z. Surface and interface engineering in copper-based bimetallic materials for selective CO2 electroreduction. Chem 4, 1809–1831 (2018).
Huang, Y., Handoko, A. D., Hirunsit, P. & Yeo, B. S. Electrochemical reduction of CO2 using copper single-crystal surfaces: effects of CO* coverage on the selective formation of ethylene. ACS Catal. 7, 1749–1756 (2017).
Monzó, J. et al. Enhanced electrocatalytic activity of Au@Cu core-shell nanoparticles towards CO2 reduction. J. Mater. Chem. A 3, 23690–23698 (2015).
Morales-Guio, C. G. et al. Improved CO2 reduction activity towards C2+ alcohols on a tandem gold on copper electrocatalyst. Nat. Catal. 1, 764–771 (2018).
Chen, C. et al. Cu-Ag tandem catalysts for high-rate CO2 electrolysis toward multicarbons. Joule 4, 1688–1699 (2020).
Wang, X. et al. Mechanistic reaction pathways of enhanced ethylene yields during electroreduction of CO2–CO co-feeds on Cu and Cu-tandem electrocatalysts. Nat. Nanotechnol. 14, 1063–1070 (2019).
Gao, J. et al. Selective C-C coupling in carbon dioxide electroreduction via efficient spillover of intermediates as supported by operando Raman spectroscopy. J. Am. Chem. Soc. 141, 18704–18714 (2019).
Fu, J. et al. Bipyridine‐assisted assembly of Au nanoparticles on Cu nanowires to enhance electrochemical reduction of CO2. Angew. Chem. Int. Ed. Engl. 131, 14238–14241 (2019).
Hoang, T. T. H. et al. Nanoporous copper-silver alloys by additive-controlled electrodeposition for the selective electroreduction of CO2 to ethylene and ethanol. J. Am. Chem. Soc. 140, 5791–5797 (2018).
Li, F. et al. Cooperative CO2-to-ethanol conversion via enriched intermediates at molecule–metal catalyst interfaces. Nat. Catal. 3, 75–82 (2019).
Wang, L. et al. Electrochemical carbon monoxide reduction on polycrystalline copper: effects of potential, pressure, and pH on selectivity toward multicarbon and oxygenated products. ACS Catal. 8, 7445–7454 (2018).
Schreier, M., Yoon, Y., Jackson, M. N. & Surendranath, Y. Competition between H and CO for active sites governs copper-mediated electrosynthesis of hydrocarbon fuels. Angew. Chem. Int. Ed. Engl. 57, 10221–10225 (2018).
Ren, D. et al. Atomic layer deposition of ZnO on CuO enables selective and efficient electroreduction of carbon dioxide to liquid fuels. Angew. Chem. Int. Ed. Engl. 58, 15036–15040 (2019).
She, X. et al. Tandem electrodes for carbon dioxide reduction into C2+ products at simultaneously high production efficiency and rate. Cell Rep. Phys. Sci. 1, 100051 (2020).
Zhang, T., Li, Z., Zhang, J. & Wu, J. Enhance CO2-to-C2+ products yield through spatial management of CO transport in Cu/ZnO tandem electrodes. J. Catal. 387, 163–169 (2020).
Levenspiel, O. Chemical Reaction Engineering 3rd edn, Ch. 5 (John Wiley & Sons, 1999).
Weng, L.-C., Bell, A. T. & Weber, A. Z. A systematic analysis of Cu-based membrane-electrode assemblies for CO2 reduction through multiphysics simulation. Energy Environ. Sci. 13, 3592–3606 (2020).
Kas, R. et al. Along the channel gradients impact on the spatioactivity of gas diffusion electrodes at high conversions during CO2 electroreduction. ACS Sustain. Chem. Eng. 9, 1286–1296 (2021).
Bui, J. C., Kim, C., Weber, A. Z. & Bell, A. T. Dynamic boundary layer simulation of pulsed CO2 electrolysis on a copper catalyst. ACS Energy Lett. 6, 1181–1188 (2021).
Dunwell, M., Luc, W., Yan, Y., Jiao, F. & Xu, B. Understanding surface-mediated electrochemical reactions: CO2 reduction and beyond. ACS Catal. 8, 8121–8129 (2018).
Moradzaman, M., Martínez, C. S. & Mul, G. Effect of partial pressure on product selectivity in Cu-catalyzed electrochemical reduction of CO2. Sustain. Energy Fuels 4, 5195–5202 (2020).
Hori, Y., Takahashi, R., Yoshinami, Y. & Murata, A. Electrochemical reduction of CO at a copper electrode. J. Phys. Chem. B 101, 7075–7081 (1997).
Wang, L. et al. Selective reduction of CO to acetaldehyde with CuAg electrocatalysts. Proc. Natl Acad. Sci. USA 117, 12572–12575 (2020).
Corral, D. et al. Advanced manufacturing for electrosynthesis of fuels and chemicals from CO2. Energy Environ. Sci. 14, 3064–3074 (2021).
Tan, Y. C., Lee, K. B., Song, H. & Oh, J. Modulating local CO2 concentration as a general strategy for enhancing C−C coupling in CO2 electroreduction. Joule 4, 1104–1120 (2020).
Salvatore, D. A. et al. Electrolysis of gaseous CO2 to CO in a flow cell with a bipolar membrane. ACS Energy Lett. 3, 149–154 (2017).
Chen, Y. et al. A robust, scalable platform for the electrochemical conversion of CO2 to formate: identifying pathways to higher energy efficiencies. ACS Energy Lett. 5, 1825–1833 (2020).
Huang, J. E. et al. CO2 electrolysis to multicarbon products in strong acid. Science 372, 1074–1078 (2021).
McCallum, C. et al. Reducing the crossover of carbonate and liquid products during carbon dioxide electroreduction. Cell Rep. Phys. Sci. 2, 100522 (2021).
Li, T. et al. Electrolytic conversion of bicarbonate into CO in a flow cell. Joule 3, 1487–1497 (2019).
Weng, L.-C., Bell, A. T. & Weber, A. Z. Towards membrane-electrode assembly systems for CO2 reduction: a modeling study. Energy Environ. Sci. 12, 1950–1968 (2019).
Zhang, T. et al. Nickel-nitrogen-carbon molecular catalysts for high rate CO2 electro-reduction to CO: on the role of carbon substrate and reaction chemistry. ACS Appl. Energy Mater. 3, 1617–1626 (2020).
Acknowledgements
This material is based upon work supported by the Office of Fossil Energy and Carbon Management of the US Department of Energy under award number DE-FE0031919 and performed at the Joint Center for Artificial Photosynthesis, a DOE Energy Innovation Hub, supported through the Office of Science of the US Department of Energy under award number DE-SC0004993 and the National Institutes of Health under grant no. S10OD023532. The authors at University of Cincinnati also thank National Science Foundation for financial support (award no. CBET-2033343). J.C.B. acknowledges funding from the National Science Foundation Graduate Research Fellowship under grant no. DGE 1752814. J.C.B. acknowledges funding, in part, by a fellowship award through the National Defense Science and Engineering Graduate (NDSEG) Fellowship Program sponsored by the Army Research Office (ARO). J.C.B. would also like to acknowledge fruitful discussion regarding along-the-channel transport in CO2 electrolysers with E. Lees.
Author information
Authors and Affiliations
Contributions
J.W., A.Z.W. and A.T.B. supervised the project. J.W. and T.Z. designed the experiments. T.Z. prepared the electrodes, performed electrochemical experiments and characterizations with the help of Z.L. J.C.B. performed multiphysics simulation. T.Z., Z.L. and J.C.B. performed data interpretation. T.Z., J.C.B., A.T.B., A.Z.W. and J.W. wrote the manuscript. All authors discussed, commented on and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Edward Anthony and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–31, Notes 1–3, Tables 1–4 and numerical methods.
Source data
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 5
Statistical source data.
Rights and permissions
About this article
Cite this article
Zhang, T., Bui, J.C., Li, Z. et al. Highly selective and productive reduction of carbon dioxide to multicarbon products via in situ CO management using segmented tandem electrodes. Nat Catal 5, 202–211 (2022). https://doi.org/10.1038/s41929-022-00751-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-022-00751-0
This article is cited by
-
Directing CO2 electroreduction pathways for selective C2 product formation using single-site doped copper catalysts
Nature Chemical Engineering (2024)
-
Tandem reactors and reactions for CO2 conversion
Nature Chemical Engineering (2024)
-
Selective and energy-efficient electrosynthesis of ethylene from CO2 by tuning the valence of Cu catalysts through aryl diazonium functionalization
Nature Energy (2024)
-
Atomic Dispersed Hetero-Pairs for Enhanced Electrocatalytic CO2 Reduction
Nano-Micro Letters (2024)
-
Cu-Based Materials for Enhanced C2+ Product Selectivity in Photo-/Electro-Catalytic CO2 Reduction: Challenges and Prospects
Nano-Micro Letters (2024)