Abstract
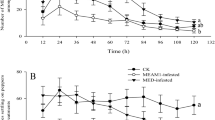
Two cryptic species of the Bemisia tabaci species complex, MEAM1 (Middle East-Asia Minor 1) and MED (Mediterranean), are highly destructive herbivores. Attack by herbivorous insects often induces defense responses in plants, including the accumulation of toxic secondary metabolites, the synthesis of herbivore-induced plant volatiles (HIPVs) and defensive proteins, and the release of volatile organic compounds that attract natural enemies. These defense responses, which often differ depending on the herbivore, may affect the preference and performance of later-arriving con- and heterospecific whiteflies that attack the same plant. In the present study, we found that MEAM1 whiteflies preferred settling and ovipositing on non-infested and MED-infested cabbage over MEAM1-infested cabbage plants, but that MED whiteflies showed no significant difference in settling or oviposition preference among non-infested, MEAM1-infested, and MED-infested plants. MEAM1 infestation increased the contents of total phenols and flavonoids in cabbage, which coincided with the increased expression of the following genes in the phenylpropanoid biosynthesis pathway: PAL2, C4H, and 4CL. Soluble protein contents were also markedly higher in the MEAM1-infested cabbage plants than in the non-infested and MED-infested cabbage plants. Overall, these results demonstrate that previous infestation by MEAM1 and MED whiteflies induced defense responses in cabbage plants that had different effects on the host preference and performance of later-arriving con- and heterospecific whiteflies. Phenolic compounds may be the key factors influencing host choice by MEAM1 and MED whiteflies on cabbage plants.





Similar content being viewed by others
References
Aqueel MA, Collins CM, Raza ABM, Ahmad S, Tariq M, Leather SR (2014) Effect of plant nutrition on aphid size, prey consumption, and life history characteristics of green lacewing. Insect Sci 21:74–82. https://doi.org/10.1111/1744-7917.12019
Awmack CS, Leather SR (2002) Host plant quality and fecundity in herbivorous insects. Ann Rev Entomol 47:817–844. https://doi.org/10.1146/annurev.ento.47.091201.145300
Beckers GJM, Spoel SH (2006) Fine-tuning plant defence signalling: salicylate versus jasmonate. Plant Biol 8:1–10. https://doi.org/10.1055/s-2005-87270
Bernays EA (1999) When host choice is a problem for a generalist herbivore: experiments with the whitefly Bemisia tabaci. Ecol Entomol 24:260–267. https://doi.org/10.1046/j.1365-2311.1999.00193.x
Bernays EA, Minkenberg O (1997) Insect herbivores: different reasons for being a generalist. Ecology 78:1157–1169. https://doi.org/10.1890/0012-9658(1997)078[1157:IHDRFB]2.0.CO;2
Chen MS (2008) Inducible direct plant defense against insect herbivores: a review. Insect Sci 15:101–114. https://doi.org/10.1111/j.1744-7917.2008.00190.x
Close DC, Mcarthur C (2002) Rethinking the role of many plant phenolics: protection from photodamage not herbivores? Oikos 99:166–172. https://doi.org/10.1034/j.1600-0706.2002.990117.x
Crowder DW, Sitvarin MI, Carrière Y (2010) Mate discrimination in invasive whitefly species. J Insect Behav 23:364–380. https://doi.org/10.1007/s10905-010-9219-7
Cui HY, Sun YC, Su JW, Li CY, Ge F (2012) Reduction in the fitness of Bemisia tabaci fed on three previously infested tomato genotypes differing in the jasmonic acid pathway. Environ Entomol 41:1443–1453. https://doi.org/10.1603/EN11264
Cui HY, Guo LT, Wang SL, Xie W, Jiao XG, Wu QJ, Zhang YJ (2017) The ability to manipulate plant glucosinolates and nutrients explains the better performance of Bemisia tabaci Middle East-Asia Minor 1 than Mediterranean on cabbage plants. Ecol Evol 7:6141–6150. https://doi.org/10.1002/ece3.2921
Darshanee HL, Ren H, Ahmed N, Zhang ZF, Liu YH, Liu TX (2017) Volatile-mediated attraction of greenhouse whitefly Trialeurodes vaporariorum to tomato and eggplant. Front Plant Sci 8:1285. https://doi.org/10.3389/fpls.2017.01285
De Vos M, Jander G (2009) Myzus persicae (green peach aphid) salivary components induce defence responses in Arabidopsis thaliana. Plant Cell Environ 32:1548–1560. https://doi.org/10.1111/j.1365-3040.2009.02019.x
De Geyter N, Gholami A, Goormachtig S, Goossens A (2012) Transcriptional machineries in jasmonate-elicited plant secondary metabolism. Trends Plant Sci 17:349–359. https://doi.org/10.1016/j.tplants.2012.03.001
De Moraes CM, Mescher MC, Tumlinson JH (2001) Caterpillar-induced nocturnal plant volatiles repel conspecific females. Nature 410:577–580. https://doi.org/10.1038/35069058
Dinsdale A, Cook L, Riginos C, Buckley YM, De Barro P (2010) Refined global analysis of Bemisia tabaci (Hemiptera: Sternorrhyncha: Aleyrodoidea: Aleyrodidae) mitochondrial cytochrome oxidase 1 to identify species level genetic boundaries. Ann Entomol Soc Am 103:196–208. https://doi.org/10.1603/AN09061
Dixon RA, Achnine L, Kota P, Liu CJ, Reddy MSS, Wang LJ (2002) The phenylpropanoid pathway and plant defence-a genomics perspective. Mol Plant Pathol 3:371–390. https://doi.org/10.1046/j.1364-3703.2002.00131.x
Dudareva N, Klempien A, Muhlemann JK, Kaplan I (2013) Biosynthesis, function and metabolic engineering of plant volatile organic compounds. New Phytol 198:16–32. https://doi.org/10.1111/nph.12145
Elbaz M, Halon E, Malka O, Malitsky S, Blum E, Aharoni A, Morin S (2012) Asymmetric adaptation to indolic and aliphatic glucosinolates in the B and Q sibling species of Bemisia tabaci (Hemiptera: Aleyrodidae). Mol Ecol 21:4533–4546. https://doi.org/10.1111/j.1365-294X.2012.05713.x
Erb M, Robert CAM, Hibbard BE, Turlings TCJ (2011) Sequence of arrival determines plant-mediated interactions between herbivores. J Ecol 99:7–15. https://doi.org/10.1111/j.1365-2745.2010.01757.x
Erb M, Meldau S, Howe GA (2012) Role of phytohormones in insect-specific plant reactions. Trends Plant Sci 17:250–259. https://doi.org/10.1016/j.tplants.2012.01.003
Esterhuizen LL, Mabasa KG, van Heerden SW, Czosnek H, Brown JK, van Heerden H, Rey MEC (2013) Genetic identification of members of the Bemisia tabaci cryptic species complex from South Africa reveals native and introduced haplotypes. J Appl Entomol 137:122–135. https://doi.org/10.1111/j.1439-0418.2012.01720.x
Ferry N, Edwards MG, Gatehouse JA, Gatehouse JA (2004) Plant-insect interactions: molecular approaches to insect resistance. Curr Opin Biotechnol 15:155–161. https://doi.org/10.1016/j.copbio.2004.01.008
Gripenberg S, Mayhew PJ, Parnell M, Roslin T (2010) A meta-analysis of preference-performance relationships in phytophagous insects. Ecol Lett 13:383–393. https://doi.org/10.1111/j.1461-0248.2009.01433.x
Hatano E, Saveer AM, Borrero-Echeverry F, Strauch M, Zakir A, Bengtsson M, Ignell R, Anderson P, Becher PG, Witzgall P, Dekker T (2015) A herbivore-induced plant volatile interferes with host plant and mate location in moths through suppression of olfactory signalling pathways. BMC Biol 13:1–15. https://doi.org/10.1016/j.tplants.2004.09.006
Holopainen JK (2004) Multiple functions of inducible plant volatiles. Trends Plant Sci 9:529–533. https://doi.org/10.1016/j.tplants.2004.09.006
Hopkins RJ, van Dam NM, van Loon JJA (2009) Role of glucosinolates in insect-plant relationships and multitrophic interactions. Annu Rev Entomol 54:57–83. https://doi.org/10.1146/annurev.ento.54.110807.090623
Howe GA, Jander G (2008) Plant immunity to insect herbivores. Annu Rev Plant Biol 59:41–66. https://doi.org/10.1146/annurev.arplant.59.032607.092825
Hu J, Yang JJ, Liu BM, Cui HY, Zhang YJ, Jiao XG (2020) Feeding behavior explains the different effects of cabbage on MEAM1 and MED cryptic species of Bemisia tabaci. Insect Sci 27:1276–1284. https://doi.org/10.1111/1744-7917.12739
Huot B, Yao J, Montgomery BL, He SY (2014) Growth-defense tradeoffs in plants: a balancing act to optimize fitness. Mol Plan 7:1267–1287. https://doi.org/10.1093/mp/ssu049
Iida H, Kitamura T, Honda K (2009) Comparison of egg-hatching rate, survival rate and development time of the immature stage between B- and Q-biotypes of Bemisia tabaci (Gennadius) (Homoptera: Aleyrodidae) on various agricultural crops. Appl Entomol Zool 44:267–273. https://doi.org/10.1303/aez.2009.267
Inbar M, Gerling D (2008) Plant-mediated interactions between whiteflies, herbivores, and natural enemies. Annu Rev Entomol 53:431–448. https://doi.org/10.1146/annurev.ento.53.032107.122456
Inbar M, Doostdar H, Mayer RT (1999) Effects of sessile whitefly nymphs (Homoptera: Aleyrodidae) on leaf-chewing larvae (Lepidoptera: Noctuidae). Environ Entomol 28:353–357. https://doi.org/10.1093/ee/28.3.353
Jiao XG, Xie W, Wang SL, Wu QJ, Zhou L, Pan HP, Liu BB, Zhang YJ (2012) Host preference and nymph performance of B and Q putative species of Bemisia tabaci on three host plants. J Pest Sci 85:423–430. https://doi.org/10.1007/s10340-012-0441-2
Jiao XG, Xie W, Wang SL, Wu QJ, Pan HP, Liu BM, Zhang YJ (2013) Differences in host selection and performance between B and Q putative species of Bemisia tabaci on three host plants. Entomol Exp Appl 147:1–8. https://doi.org/10.1111/eea.12040
Jiao XG, Xie W, Guo LT, Liu BM, Wang SL, Wu QJ, Zhang YJ (2014) Differing effects of cabbage and pepper on B and Q putative species of Bemisia tabaci. J Pest Sci 87:629–637. https://doi.org/10.1007/s10340-014-0594-2
Jiao XG, Xie W, Zeng Y, Wang C, Liu BM, Wang SL, Wu QJ, Zhang YJ (2018) Lack of correlation between host choice and feeding efficiency for the B and Q putative species of Bemisia tabaci on four pepper genotypes. J Pest Sci 91:133–143. https://doi.org/10.1007/s10340-017-0906-4
Karban R, Agrawal AA (2002) Herbivore offense. Annu Rev Ecol Syst 33:641–664. https://doi.org/10.1146/annurev.ecolsys.33.010802.150443
Karban R, Baldwin IT (1997) Induced responses to herbivory. University of Chicago Press, Chicago. https://doi.org/10.7208/9780226424972
Kempema LA, Cui X, Holzer FM, Walling LL (2007) Arabidopsis transcriptome changes in response to phloem-feeding silverleaf whitefly nymphs. Similarities and distinctions in responses to aphids. Plant Physiol 143:849–865. https://doi.org/10.1104/pp.106.090662
Kessler A, Baldwin IT (2002) Plant responses to insect herbivory: the emerging molecular analysis. Ann Rev Plant Biol 53:299–328. https://doi.org/10.1146/annurev.arplant.53.100301.135207
Kessler A, Halitschke R (2007) Specificity and complexity: the impact of herbivore-induced plant responses on arthropod community structure. Curr Opin Plant Biol 10:409–414. https://doi.org/10.1016/j.pbi.2007.06.001
Koiwa H, Bressan RA, Hasegawa PM (1997) Regulation of protease inhibitors and plant defense. Trends Plant Sci 2:379–384. https://doi.org/10.1016/S1360-1385(97)90052-2
Koornneef A, Pieterse CMJ (2008) Cross talk in defense signaling. Plant Physiol 146:839–844. https://doi.org/10.1104/pp.107.112029
Li J, Qian HM, Pan LL, Wang QM, Liu SS (2021) Performance of two species of whiteflies is unaffected by glucosinolate profile in Brassica plants. Pest Manag Sci 77:4313–4320. https://doi.org/10.1002/ps.6460
Liu BM, Yan FM, Chu D, Pan HP, Jiao XG, Xie W, Wu QJ, Wang SL, Xu BY, Zhou XG, Zhang YJ (2012) Difference in feeding behaviors of two invasive whiteflies on host plants with different suitability: implication for competitive displacement. Int J Biol Sci 8:697–706. https://doi.org/10.7150/ijbs.4108
Lu ZX, Yu XP, Heong KL, Hu C (2007) Effect of nitrogen fertilizer on herbivores and its stimulation to major insect pests in rice. Rice Sci 14:56–66. https://doi.org/10.1016/S1672-6308(07)60009-2
Markovich O, Kafle D, Elbaz M, Malitsky S, Aharoni A, Schwarzkopf A, Gershenzon J, Morin S (2013) Arabidopsis thaliana plants with different levels of aliphaticand indolyl-glucosinolates affect host selection and performance of Bemisia tabaci. J Chem Ecol 39:1361–1372. https://doi.org/10.1007/s10886-013-0358-0
Mauch-Mani B, Baccelli I, Luna E, Flors V (2017) Defense priming: an adaptive part of induced resistance. Annu Rev Plant Biol 68:485–512. https://doi.org/10.1146/annurev-arplant-042916-041132
Oliveira MRV, Henneberry TJ, Anderson P (2001) History, current status, and collaborative research projects for Bemisia tabaci. Crop Prot 20:709–723. https://doi.org/10.1016/S0261-2194(01)00108-9
Pan HP, Preisser EL, Chu D, Wang SL, Wu QJ, Carrière Y, Zhou XG, Zhang YJ (2015) Insecticides promote viral outbreaks by altering herbivore competition. Ecol Appl 25:1585–1595. https://doi.org/10.1890/14-0752.1
Pieterse CMJ, Van der Does D, Zamioudis C, Leon-Reyes A, Van Wees SCM (2012) Hormonal modulation of plant immunity. Annu Rev Cell Dev Bi 28:489–521. https://doi.org/10.1146/annurev-cellbio-092910-154055
Poelman EH, Dicke M (2014) Plant-mediated interactions among insects within a community ecological perspective. Annu Plant Rev 47:309–337. https://doi.org/10.1002/9781118829783.ch9
Poelman EH, Broekgaarden C, Van Loon JJA, Dicke M (2008) Early season herbivore differentially affects plant defence responses to subsequently colonizing herbivores and their abundance in the field. Mol Ecol 17:3352–3365. https://doi.org/10.1111/j.1365-294X.2008.03838.x
Rodriguez-Saona C, Crafts-Brandner SJ, Cañas LA (2003) Volatile emissions triggered by multiple herbivore damage: beet armyworm and whitefly feeding on cotton plants. J Chem Ecol 29:2539–2550. https://doi.org/10.1023/A:1026314102866
Sarmento RA, Lemos F, Bleeker PM, Schuurink RC, Pallini A, Oliveira MGA, Lima ER, Kant R, Sabelis MW, Janssen A (2011) A herbivore that manipulates plant defence. Ecol Lett 14:229–236. https://doi.org/10.1111/j.1461-0248.2010.01575.x
Schuman MC, Baldwin IT (2016) The layers of plant responses to insect herbivores. Annu Rev Entomol 61:373–394. https://doi.org/10.1146/annurev-ento-010715-023851
Schwachtje J, Baldwin IT (2008) Why does herbivore attack reconfigure primary metabolism? Plant Physiol 146:845–851. https://doi.org/10.1104/pp.107.112490
Silva DB, Jiménez A, Urbaneja A, Pérez-Hedo M, Bento JM (2021) Changes in plant responses induced by an arthropod influence the colonization behavior of a subsequent herbivore. Pest Manag Sci. https://doi.org/10.1002/ps.6454
Stahl E, Hilfiker O, Reymond P (2018) Plant-arthropod interactions: who is the winner? Plant J 93:703–728. https://doi.org/10.1111/tpj.13773
Stam JM, Kroes A, Li Y, Gols R, van Loon JJA, Poelman EH, Dicke M (2014) Plant interactions with multiple insect herbivores: from community to genes. Annu Rev Plant Biol 65:689–713. https://doi.org/10.1146/annurev-arplant-050213-035937
Su Q, Chen G, Mescher MC, Peng ZK, Xie W, Wang SL, Wu QJ, Liu J, Li CR, Wang WK, Zhang YJ (2018) Whitefly aggregation on tomato is mediated by feeding-induced changes in plant metabolites that influence the behaviour and performance of conspecifics. Funct Ecol 32:1180–1193. https://doi.org/10.1111/1365-2435.13055
Sun DB, Liu YQ, Qin L, Xu J, Li FF, Liu SS (2013) Competitive displacement between two invasive whiteflies: insecticide application and host plant effects. B Entomol Res 103:344–353. https://doi.org/10.1017/S0007485312000788
Visanuvimol L, Bertram SM (2010) Dietary phosphorus availability influences female cricket lifetime reproductive effort. Ecol Entomol 35:386–395. https://doi.org/10.1111/j.1365-2311.2010.01195.x
Viswanathan DV, Lifchits OA, Thaler JS (2007) Consequences of sequential attack for resistance to herbivores when plants have specific induced responses. Oikos 116:1389–1399. https://doi.org/10.1111/j.0030-1299.2007.15882.x
Walling LL (2000) The myriad plant responses to herbivores. J Plant Growth Regul 19:195–216. https://doi.org/10.1007/s003440000026
Walling LL (2008) Avoiding effective defenses: strategies employed by phloem-feeding insects. Plant Physiol 146:859–866. https://doi.org/10.1104/pp.107.113142
Xu HX, Qian LX, Wang XW, Shao RX, Hong Y, Liu SS, Wang XW (2019) A salivary effector enables whitefly to feed on host plants by eliciting salicylic acid-signaling pathway. P Natl Acad Sci USA 116:490–495. https://doi.org/10.1073/pnas.1714990116
Xue M, Wang CX, Bi MJ, Li QL, Liu TX (2010) Induced defense by Bemisia tabaci biotype B (Hemiptera: Aleyrodidae) in tobacco against Myzus persicae (Hemiptera: Aphididae). Environ Entomol 39:883–891. https://doi.org/10.1603/EN09307
Yang CH, Guo JY, Chu D, Ding TB, Wei KK, Cheng DF, Wan FH (2017) Secretory laccase 1 in Bemisia tabaci MED is involved in whitefly-plant interaction. Sci Rep 7:1–9. https://doi.org/10.1038/s41598-017-03765-y
Yang JJ, Xie W, Liu BM, Wang SL, Wu QJ, He YC, Zhang YJ, Jiao XG (2020) Phenolics, rather than glucosinolates, mediate host choice of Bemisia tabaci MEAM1 and MED on five cabbage genotypes. J Appl Entomol 144:287–296. https://doi.org/10.1111/jen.12737
Zarate SI, Kempema LA, Walling LL (2007) Silverleaf whitefly induces salicylic acid defenses and suppresses effectual jasmonic acid defenses. Plant Physiol 143:866–875. https://doi.org/10.1104/pp.106.090035
Zhang PJ, Zheng SJ, van Loon JJA, Boland W, David A, Mumm R, Dicke M (2009) Whiteflies interfere with indirect plant defense against spider mites in Lima bean. P Natl Acad Sci USA 106:21202–21207. https://doi.org/10.1073/pnas.0907890106
Zhang PJ, Li WD, Huang F, Zhang MJ, Xu FC, Lu YB (2013a) Feeding by whiteflies suppresses downstream jasmonic acid signaling by eliciting salicylic acid signaling. J Chem Ecol 39:612–619. https://doi.org/10.1007/s10886-013-0283-2
Zhang PJ, Xu CX, Zhang JM, Lu YB, Wei JN, Liu YQ, David A, Boland W, Turlings TCJ (2013b) Phloem-feeding whiteflies can fool their host plants, but not their parasitoids. Funct Ecol 27:1304–1312. https://doi.org/10.1111/1365-2435.12132
Zhang SZ, Huang H, Shan HW, Zhang F, Wan FH, Liu TX (2013c) Defense against Pieris rapae in cabbage plants induced by Bemisia tabaci biotype B. Entomol Exp Appl 147:293–300. https://doi.org/10.1111/eea.12067
Zhang GF, Lövei GL, Hu M, Wan FH (2014) Asymmetric consequences of host plant occupation on the competition between the whiteflies Bemisia tabaci cryptic species MEAM1 and Trialeurodes vaporariorum (Hemiptera: Aleyrodidae). Pest Manag Sci 70:1797–1807. https://doi.org/10.1002/ps.3713
Zhang X, Sun X, Zhao HP, Xue M, Wang D (2017) Phenolic compounds induced by Bemisia tabaci and Trialeurodes vaporariorum in Nicotiana tabacum L. and their relationship with the salicylic acid signaling pathway. Arthropod-Plant Inte 11:659–667. https://doi.org/10.1007/s11829-017-9508-6
Zhang PJ, Wei JN, Zhao C, Zhang YF, Li CY, Liu SS, Dicke M, Yu XP, Turlings TCJ (2019) Airborne host-plant manipulation by whiteflies via an inducible blend of plant volatiles. P Natl Acad Sci USA 116:7387–7396. https://doi.org/10.1073/pnas.1818599116
Zhao HP, Zhang XY, Xue M, Zhang X (2015) Feeding of whitefly on tobacco decreases aphid performance via increased salicylate signaling. PLoS One 10:e0138584. https://doi.org/10.1371/journal.pone.0138584
Acknowledgements
This study was funded by the National Natural Science Foundation of China (31371941, 31572012).
Funding
Funding was provided by the National Natural Science Foundation of China, 31371941, Xiaoguo Jiao, 31572012, Xiaoguo Jiao.
Author information
Authors and Affiliations
Contributions
XGJ and YJZ designed the experiment. JH, JTL, BML, and PYF performed the experiment. XGJ and NNY analyzed the data. JH, XGJ, and YJZ wrote the paper. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Cesar Rodriguez-Saona.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hu, J., Lu, J., Yang, N. et al. Avoidance of previously infested cabbage by MEAM1 cryptic species of Bemisia tabaci species complex. J Pest Sci 96, 81–92 (2023). https://doi.org/10.1007/s10340-022-01480-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-022-01480-w