Abstract
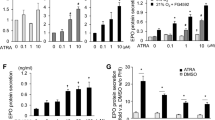
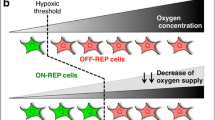
Erythropoietin (EPO), a central protein of erythropoiesis, plays an important role during hypoxia adaptation and is regulated by hypoxia-inducible factor (HIF). However, there is no report on EPO-producing cells and their regulatory mechanisms in yak (Bos grunniens). To understand EPO production and regulation of yak, kidneys from different age of yak were collected and expression of EPO, hypoxia-inducible factor 1 alpha (HIF-1α), and hypoxia-inducible factor 2 alpha (HIF-2α) were detected. Then renal tubule epithelial cells (RTECs) and peritubular interstitial fibroblast-like (RIFs) cells were isolated and cultured to determine their EPO production abilities. Subsequently, the cells were treated with dimethyloxalylglycine (DMOG) and Geldanamycin (GA), which are inhibitors of prolyl-4-hydroxylase domain (PHD) and heat shock protein 90 (HSP90) respectively, and siRNAs of HIF-1α and HIF-2α to explore their effect on EPO production and regulation. The results showed that expressions of EPO, HIF-1α, and HIF-2α were different in the different age groups of yak. High DMOG concentration caused a corresponding increase in the levels of HIF-1α and HIF-2α in RIFs and RTECs, however, EPO levels increased in RIFs only and was not detected at any concentration in RTECs; suggesting that EPO was produced in RIFs. Upon treating RIFs with siRNAs of HIF-1α and HIF-2α, we found that EPO was regulated by PHD through HIF-2α. In addition, increasing GA concentration caused a decrease in expression of HSP90, HIF-1α, HIF-2α, and EPO in RIFs. In conclusion, these findings support our proposition that PHD regulates EPO via HIF-2α in yak RIFs, while HSP90 impelled EPO expression.








Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article. Data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Dane DM, Yilmaz C, Gyawali D et al (2019) Erythropoietin inhalation enhances adult canine alveolar-capillary formation following pneumonectomy. Am J Physiol Lung Cell Mol Physiol 316(5):L936–L945. https://doi.org/10.1152/ajplung.00504.2018
Fernandez-Torres J, Zamudio-Cuevas Y, Martinez-Nava GA et al (2017) Hypoxia-Inducible Factors (HIFs) in the articular cartilage: a systematic review. Eur Rev Med Pharmacol Sci 21(12):2800–2810
Gerl K, Miquerol L, Todorov VT et al (2015) Inducible glomerular erythropoietin production in the adult kidney. Kidney Int 88(6):1345–1355. https://doi.org/10.1038/ki.2015.274
Gradin K, McGuire J, Wenger RH et al (1996) Functional interference between hypoxia and dioxin signal transduction pathways: competition for recruitment of the Arnt transcription factor. Mol Cell Biol 16(10):5221–5231. https://doi.org/10.1128/MCB.16.10.5221
Haase VH (2017a) HIF-prolyl hydroxylases as therapeutic targets in erythropoiesis and iron metabolism. Hemodial Int 21(Suppl 1):S110–S124. https://doi.org/10.1111/hdi.12567
Haase VH (2017b) Oxygen sensors as therapeutic targets in kidney disease. Nephrol Ther 13(Suppl 1):S29–S34. https://doi.org/10.1016/j.nephro.2017.01.015
Hao Y, Gu XH (2014) Effects of heat shock protein 90 expression on pectoralis major oxidation in broilers exposed to acute heat stress. Poult Sci 93(11):2709–2717. https://doi.org/10.3382/ps.2014-03993
He Y, Yu S, Hu J et al (2016) Changes in the anatomic and microscopic structure and the expression of HIF-1alpha and VEGF of the yak heart with aging and hypoxia. PLoS ONE 11(2):e0149947. https://doi.org/10.1371/journal.pone.0149947
Imeri F, Nolan KA, Bapst AM et al (2019) Generation of renal Epo-producing cell lines by conditional gene tagging reveals rapid HIF-2 driven Epo kinetics, cell autonomous feedback regulation, and a telocyte phenotype. Kidney Int 95(2):375–387. https://doi.org/10.1016/j.kint.2018.08.043
Isaacs JS, Jung YJ, Mimnaugh EG et al (2002) Hsp90 regulates a von Hippel Lindau-independent hypoxia-inducible factor-1 alpha-degradative pathway. J Biol Chem 277(33):29936–29944. https://doi.org/10.1074/jbc.M204733200
Ivan M, Kaelin WG Jr (2017) The EGLN-HIF O2-sensing system: multiple inputs and feedbacks. Mol Cell 66(6):772–779. https://doi.org/10.1016/j.molcel.2017.06.002
Jacobson LO, Goldwasser E, Fried W et al (1957) Role of the kidney in erythropoiesis. Nature 179(4560):633–634. https://doi.org/10.1038/179633a0
Jelkmann W (2011) Regulation of erythropoietin production. J Physiol 589(6):1251–1258. https://doi.org/10.1113/jphysiol.2010.195057
Kobayashi A, Mugford JW, Krautzberger AM et al (2014) Identification of a multipotent self-renewing stromal progenitor population during mammalian kidney organogenesis. Stem Cell Rep 3(4):650–662. https://doi.org/10.1016/j.stemcr.2014.08.008
Kobayashi H, Liu Q, Binns TC et al (2016) Distinct subpopulations of FOXD1 stroma-derived cells regulate renal erythropoietin. J Clin Invest 126(5):1926–1938. https://doi.org/10.1172/JCI83551
Koury MJ, Haase VH (2015) Anaemia in kidney disease: harnessing hypoxia responses for therapy. Nat Rev Nephrol 11(7):394–410. https://doi.org/10.1038/nrneph.2015.82
Kurt B, Paliege A, Willam C et al (2013) Deletion of von Hippel-Lindau protein converts renin-producing cells into erythropoietin-producing cells. J Am Soc Nephrol 24(3):433–444. https://doi.org/10.1681/ASN.2012080791
Lee FS, Percy MJ (2011) The HIF pathway and erythrocytosis. Annu Rev Pathol 6:165–192. https://doi.org/10.1146/annurev-pathol-011110-130321
Liu YV, Semenza GL (2007) RACK1 vs. HSP90: competition for HIF-1 alpha degradation vs. stabilization. Cell Cycle 6(6):656–659. https://doi.org/10.4161/cc.6.6.3981
Liu P, Yu S, Cui Y et al (2017) Cloning of HSP90, expression and localization of HSP70/90 in different tissues including lactating/non-lactating yak (Bos grunniens) breast tissue. PLoS ONE 12(7):e0179321. https://doi.org/10.1371/journal.pone.0179321
Masoud GN, Li W (2015) HIF-1alpha pathway: role, regulation and intervention for cancer therapy. Acta Pharm Sin B 5(5):378–389. https://doi.org/10.1016/j.apsb.2015.05.007
McGraw K, List A (2017) Erythropoietin receptor signaling and lipid rafts. Vitam Horm 105:79–100. https://doi.org/10.1016/bs.vh.2017.02.002
Minet E, Mottet D, Michel G et al (1999) Hypoxia-induced activation of HIF-1: role of HIF-1alpha-Hsp90 interaction. FEBS Lett 460(2):251–256. https://doi.org/10.1016/s0014-5793(99)01359-9
Miro-Murillo M, Elorza A, Soro-Arnaiz I et al (2011) Acute Vhl gene inactivation induces cardiac HIF-dependent erythropoietin gene expression. PLoS ONE 6(7):e22589. https://doi.org/10.1371/journal.pone.0022589
Montero D, Lundby C et al (2019) Arterial oxygen content regulates plasma erythropoietin independent of arterial oxygen tension: a blinded crossover study. Kidney Int 95(1):173–177. https://doi.org/10.1016/j.kint.2018.09.015
Obara N, Suzuki N, Kim K et al (2008) Repression via the GATA box is essential for tissue-specific erythropoietin gene expression. Blood 111(10):5223–5232. https://doi.org/10.1182/blood-2007-10-115857
Paliege A, Rosenberger C, Bondke A et al (2010) Hypoxia-inducible factor-2alpha-expressing interstitial fibroblasts are the only renal cells that express erythropoietin under hypoxia-inducible factor stabilization. Kidney Int 77(4):312–318. https://doi.org/10.1038/ki.2009.460
Qi W, Johnson DW, Vesey DA et al (2007) Isolation, propagation and characterization of primary tubule cell culture from human kidney. Nephrology 12(2):155–159. https://doi.org/10.1111/j.1440-1797.2007.00779.x
Rankin EB, Wu C, Khatri R et al (2012) The HIF signaling pathway in osteoblasts directly modulates erythropoiesis through the production of EPO. Cell 149(1):63–74. https://doi.org/10.1016/j.cell.2012.01.051
Ratcliffe P, Koivunen P, Myllyharju J et al (2017) Update on hypoxia-inducible factors and hydroxylases in oxygen regulatory pathways: from physiology to therapeutics. Hypoxia 5:11–20. https://doi.org/10.2147/HP.S127042
Semenza GL (2007) Hypoxia-inducible factor 1 (HIF-1) pathway. Sci STKE. https://doi.org/10.1126/stke.4072007cm8
Semenza GL (2009) Involvement of oxygen-sensing pathways in physiologic and pathologic erythropoiesis. Blood 114(10):2015–2019. https://doi.org/10.1182/blood-2009-05-189985
Semenza GL (2012) Hypoxia-inducible factors in physiology and medicine. Cell 148(3):399–408. https://doi.org/10.1016/j.cell.2012.01.021
Shao B, Long R, Ding Y et al (2010) Morphological adaptations of yak (Bos grunniens) tongue to the foraging environment of the Qinghai-Tibetan Plateau. J Anim Sci 88(8):2594–2603. https://doi.org/10.2527/jas.2009-2398
Sharpe CC, Dockrell ME (2012) Primary culture of human renal proximal tubule epithelial cells and interstitial fibroblasts. Methods Mol Biol 806:175–185. https://doi.org/10.1007/978-1-61779-367-7_12
Singh D, Arora R, Kaur P et al (2017) Overexpression of hypoxia-inducible factor and metabolic pathways: possible targets of cancer. Cell Biosci 7:62. https://doi.org/10.1186/s13578-017-0190-2
Suzuki N, Hirano I, Pan X et al (2013) Erythropoietin production in neuroepithelial and neural crest cells during primitive erythropoiesis. Nat Commun 4:2902. https://doi.org/10.1038/ncomms3902
Takeda K, Aguila HL, Parikh NS et al (2008) Regulation of adult erythropoiesis by prolyl hydroxylase domain proteins. Blood 111(6):3229–3235. https://doi.org/10.1182/blood-2007-09-114561
Tan SJ, Hewitson TD (2016) Propagation and culture of human renal fibroblasts. Methods Mol Biol 1397:11–23. https://doi.org/10.1007/978-1-4939-3353-2_2
Urrutia AA, Afzal A, Nelson J et al (2016) Prolyl-4-hydroxylase 2 and 3 coregulate murine erythropoietin in brain pericytes. Blood 128(21):2550–2560. https://doi.org/10.1182/blood-2016-05-713545
Wenger RH, Hoogewijs D (2010) Regulated oxygen sensing by protein hydroxylation in renal erythropoietin-producing cells. Am J Physiol Renal Physiol 298(6):F1287-1296. https://doi.org/10.1152/ajprenal.00736.2009
Xiong X, Fu M, Lan D et al (2015) Yak response to high-altitude hypoxic stress by altering mRNA expression and DNA methylation of hypoxia-inducible factors. Anim Biotechnol 26(3):222–229. https://doi.org/10.1080/10495398.2014.1002563
Yang X, Cui Y, Yue J et al (2017) The histological characteristics, age-related thickness change of skin, and expression of the HSPs in the skin during hair cycle in yak (Bos grunniens). PLoS ONE 12(5):e0176451. https://doi.org/10.1371/journal.pone.0176451
Funding
This study was funded by the National Natural Science Foundation of China with Grant No. 31772691.
Author information
Authors and Affiliations
Contributions
YC and HL designed the experiment. HL performed the experiment and wrote the manuscript, X-FB analyzed the data. S-JY and SYA revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
All animal procedures performed in this study were approved by the Institutional Animal Care and Use Committee (IACUC) of the College of Veterinary Medicine, Gansu Agriculture University.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cui, Y., Li, H., Yu, SJ. et al. Effects of PHD and HSP90 on erythropoietin production in yak (Bos grunniens) renal interstitial fibroblast-like cells under hypoxia. J Mol Histol 53, 395–411 (2022). https://doi.org/10.1007/s10735-021-10054-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-021-10054-6