New Oral Anticoagulants vs. Vitamin K Antagonists Among Patients With Cardiac Amyloidosis: Prognostic Impact
- 1Department of Cardiology, Rangueil University Hospital, Toulouse, France
- 2Cardiac Imaging Center, Toulouse University Hospital, Toulouse, France
- 3Department of Nuclear Medicine, Toulouse University Hospital, Toulouse, France
- 4Medical School, Toulouse III Paul Sabatier University, Toulouse, France
- 5Department of Nephrology and Referral Center for Rare Diseases, Rangueil University Hospital, Toulouse, France
- 6Department of Hematology, IUC Oncopole, Toulouse, France
- 7Department of Neurology, Purpan University Hospital, Toulouse, France
Background: Atrial arrhythmia (AA) is common among patients with cardiac amyloidosis (CA), who have an increased risk of intracardiac thrombus. The aim of this study was to explore the prognostic impact of vitamin K-antagonists (VKA) and direct oral anticoagulants (DOAC) in patients with CA.
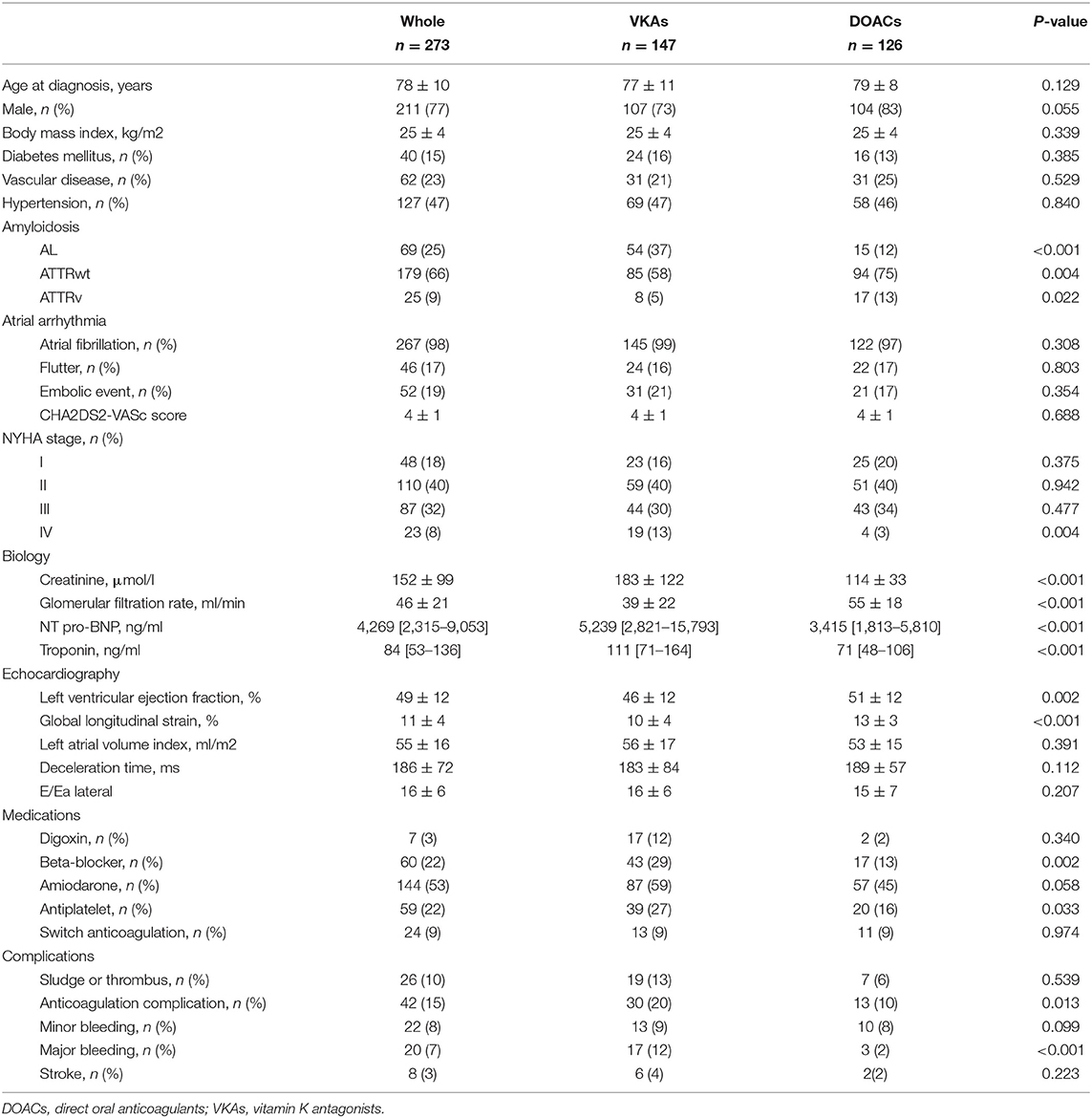
Methods and Results: 273 patients with CA and history of AA with long term anticoagulation−69 (25%) light chain amyloidosis (AL), 179 (66%) wild-type transthyretin amyloidosis (ATTRwt) and 25 (9%) variant transthyretin amyloidosis (ATTRv)–were retrospectively included between January 2012 and July 2020. 147 (54%) and 126 (46%) patients received VKA and DOAC, respectively. Patient receiving VKA were more likely to have AL with renal dysfunction, higher NT-proBNP and troponin levels. Patients with ATTRwt were more likely to receive DOAC therapy. There were more bleeding complications among patients with VKA (20 versus 10%; P = 0.013) but no difference for stroke events (4 vs. 2%; P = 0.223), as compared to patients with DOAC. A total of 124 (45%) patients met the primary endpoint of all-cause mortality: 96 (65%) and 28 (22%) among patients with VKAs and DOACs, respectively (P < 0.001). After multivariate analysis including age and renal function, VKA was no longer associated with all-cause mortality.
Conclusion: Among patients with CA and history of AA receiving oral anticoagulant, DOACs appear to be at least as effective and safe as VKAs.
Introduction
Atrial arrhythmia (AA) is common in patients with cardiac amyloidosis (CA). AA is more frequent in wild-type transthyretin amyloidosis (ATTRwt) than light chain amyloidosis (AL) or variant transthyretin amyloidosis (ATTRv). Patients with CA have an increased risk of intracardiac thrombus, reaching 33% in autopsy series (1), and its more frequent in AL with more fatal embolic events despite normal sinus rhythm and preserved ejection fraction (2). This high prevalence of intracardiac thrombus justify to recommend systematic transesophageal echocardiography before direct-current cardioversion despite correct anticoagulation with international normalized ratio between 2 and 3 more than 3 weeks (3). Furthermore, the CHA2DS2-VASc score is not reliable in cardiac amyloidosis since 67% of patients with thrombus present in pre-cardioversion transesophageal echocardiography had a score of 1 (4). Vitamin K-antagonists (VKAs) were the first anticoagulants used in patients with history of AA requiring anticoagulation, but direct oral anticoagulants (DOACs) have been introduced in previous and current European and US guidelines for the management of atrial fibrillation as suitable alternatives to VKAs for stroke prevention in AA (3, 5). However, efficiency and safety of traditional anticoagulants vs. DOACs in patients with CA have never been explored.
The aim of this study was to explore the prognostic impact of VKAs and DOACs in patients with CA requiring anticoagulation.
Methods
Study Population
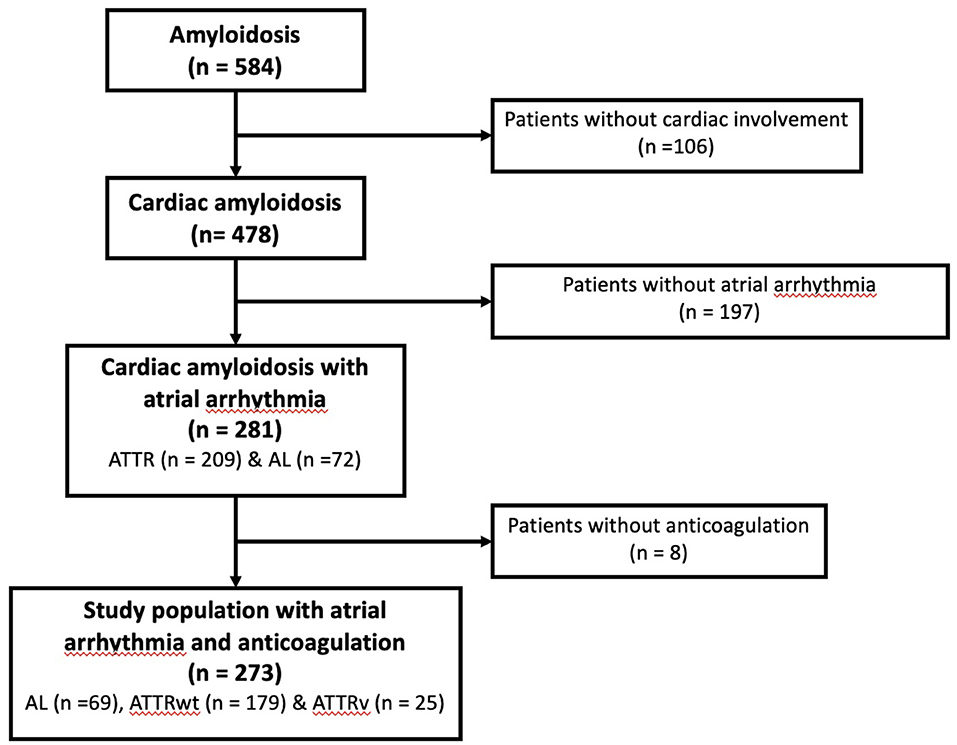
Between January 2012 and July 2020, all consecutive patients with CA diagnosed at the University Hospital of Toulouse, France, were retrospectively enrolled in this cohort. Details of the population selection have previously been described (6) and the flow chart for the population study selection is presented in Figure 1. Briefly, only patients with history of AA receiving oral anticoagulant were included in the final analysis. Patient's medical records were reviewed to collect anticoagulant treatments during management and at the time of follow-up.
AA were defined by sustained atrial fibrillation, common atrial flutter or atrial tachycardia. Oral anticoagulant drugs were represented either by VKA (Coumadine, Fluindione or Acecoumarol) or DOAC (Dabigatran, Rivaroxaban or Apixaban).
The investigation conforms with the principles outlined in the Declaration of Helsinki. All patients were informed at the admission that their clinical data could be used for research purpose and gave their consent. The study was approved by the French Data Protection Authority (Commission Nationale de l'Informatique et des Libertés, #2205212v0).
Clinical Endpoint and Follow-Up
Follow-up was assessed by electronic chart review or by phone interview of patient's general practitioner/cardiologist, patient or family for the clinical endpoint of all-cause and cardiovascular mortality. The secondary outcomes were embolic events (essentially stroke), left atrial appendage thrombus and bleeding event (anticoagulation complication). Major bleeding event was defined by symptomatic bleeding or bleeding causing a fall in hemoglobin level of 2g/dL according to International Society on Thrombosis and Haemostasis bleeding scale (7) and minor bleeding event was defined by all non-major bleedings. Patients without contact up to 6 months were considered as lost to follow-up. Only patients with available demographic data were used for survival analysis.
Statistical Analysis
Continuous variables were tested for normal distribution using the Kolmogorov–Smirnov test and expressed as mean ± standard deviation. Values not normally distributed were presented as medians with interquartile ranges (IQR). Nominal values were expressed as numbers and percentages. Comparisons of numerical variables was compared using the Student's test or the Mann-Whitney rank sum test when appropriate. Nominal variables were investigated by the χ2 test or the Fisher exact as suitable. Univariable and multivariable Cox proportional-hazards regression analysis were performed to test the association of variables associated with all-cause mortality. Univariate variables with a P-value of <0.05 were entered into the multivariate logistic models. All-cause mortality was then summarized using Kaplan-Meier survival curve and log rank test was used for initial comparisons. Patients were censored at the time of death. Differences were considered statistically significant for P-values of <0.05. All analyses were performed using standard statistical software SPSS version 20 (SPSS Inc., Chicago, Illinois).
Results
Baseline Characteristics
Among the 478 patients with CA and complete medical records initially screened, 273 (57%) had a previous history of AA with long-term anticoagulation during the follow-up: 69 (25%) AL, 179 (66%) ATTRwt and 25 (9%) ATTRv (Figure 1).
There were 147 (54%) and 126 (46%) patients receiving VKA and DOAC, respectively. Eigthy-one (55%), 67 (46%) and 2 (1%) received Warfarin, Fluindione and Acenocoumarol, respectively among patients with VKA; and 77 (61%), 35 (28%) and 14 (11%) received Apixaban, Rivaroxaban and Dabigatran, respectively among patients with DOAC. Among patients with DOAC, 55 (44%) received a low dose in accordance with the recommendations for adapting doses according to their age, weight or renal function. The baseline clinical characteristics of the study population are summarized in Table 1. There were more AL patients, renal dysfunction, higher NT-proBNP and troponin levels and associated antiplatelet therapy among patient receiving VKA. Patients with wtATTR were more likely to receive DOAC. Among patients treated with VKA treatment, 66 (45%) reported an under target International Normalized Ratio during the follow-up period.
Impact of Anticoagulant Therapy on Outcomes
There were more bleeding complications among patients with VKA as compared to patients with DOAC, with 30 (20%) vs. 13 (10%) events, respectively, P = 0.013; but no difference for stroke, with 6 (4%) vs. 2 (2%) events, respectively, P = 0.223. Thirty-six patients presented a stroke before the introduction of curative oral anticoagulation.
Survival data were available for all patients, with a median follow-up of 18 months with no significant difference between patients on VKA and DOAC respectively (18 [IQR 8-43] and 18 [IQR 8-32], p = 0.286) A total of 124 (45%) patients met the primary endpoint of all-cause mortality: 96 (65%) and 28 (22%) among patients with VKA and DOAC, respectively (P < 0.001). In univariate analysis, there was an over-risk of all-cause mortality associated with VKA treatment compared DOAC treatment (Figure 2). In multivariate analysis, age, NYHA 3 and 4 class, NT-proBNP level, renal dysfunction, global longitudinal strain level, major bleeding and beta-blocking drugs were still associated with an increased risk of mortality (Table 2). However, type of oral anticoagulation (VKA vs. DOAC) was no more linked to survival after adjustment of these variables.

Figure 2. Kaplan–Meier curves for all-cause mortality according to anticoagulation treatment for the whole population. DOACs, direct oral anticoagulants; VKAs, vitamin K antagonists.
Sub-Population of Patients With ATTRwt
Baseline characteristics of patients with ATTRwt are presented in annex. Patients receiving VKA were more symptomatic, were more likely to have renal dysfunction, lower ejection fraction and lower global longitudinal strain and to receive beta-blocker and antiplatelet therapy.
In the univariate analysis, there was an associated increased risk of VKA treatment over DOAC treatment for the composite endpoint of stroke and bleeding complications (HR 2,76 [1,28–5,97], p = 0.010) that disappeared after multivariate analysis (HR 1,84 [0,77–4,43] p = 0.171).
Regarding outcomes, patients receiving VKA had more major bleeding during the follow-up as compared to patients receiving DOAC (14 vs. 2%, respectively; p < 0.001) but there was no significant difference for stroke events (7 vs. 2%, respectively; p = 0.074). In univariate analysis, there was an over-risk of all-cause mortality associated with VKA treatment compared DOAC treatment (Figure 3). After multivariate analysis, only glomerular filtration rate was associated with an increased risk of mortality (Table 3), but type of oral anticoagulation therapy was no more linked with all-cause mortality.

Figure 3. Kaplan–Meier curves for all-cause mortality according to anticoagulation treatment among patients with ATTRwt. DOACs, direct oral anticoagulants; VKAs, vitamin K antagonists.

Table 3. Cox regression analysis for the occurrence of all-cause mortality among patients with ATTRwt.
Sub-Population of Patients With AL
Baseline characteristics of patients with AL are presented in annex. There was no difference between patients receiving VKA and DOAC except for higher proportion of renal dysfunction among patients receiving VKA. Regarding outcomes, there was no difference between groups for bleeding complications (22 vs. 13% for VKA and DOAC, respectively; P = 0.449) and there was not any stroke event over the follow-up. There was no association between type of anticoagulation therapy and all-cause mortality (Figure 4). After multivariate analysis, only E/Ea ratio on transthoracic echocardiography was associated with increased risk of mortality (Table 4).

Figure 4. Kaplan–Meier curves for all-cause mortality according to anticoagulation treatment among patients with AL. DOACs, direct oral anticoagulants; VKAs, vitamin K antagonists.
Discussion
This retrospective study on 273 patients with cardiac amyloidosis and a history of AA requiring anticoagulation shows that DOACs can be used safely without increasing mortality, embolic events or bleeding complications, which were lower compared to VKA therapy. Conversely, our study shows that there appears to be an over risk of mortality under anticoagulant treatment with VKA, which disappears after adjustment for age and renal function, regardless of the subtype of amyloidosis.
Indeed, we can see that patients with renal insufficiency were more often on VKAs, which can be explained by a reluctance of physicians to prescribe DOACs for these patients, especially at the beginning of the census of our cohort, which coincides with the beginning of the marketing authorization of DOACs.
To our knowledge, this is the first study exploring the impact of the type of anticoagulant on all-cause mortality in a population of CA. The ARISTOTLE trial (8) was the only one to show a decrease in mortality with apixaban as compared to warfarin in patients with atrial fibrillation or atrial flutter but without specific mention of CA. The recent study by Mitrani and co-authors did not find difference between patients with CA received VKAs and DOACs regarding thrombotic events and major bleeds after a 2.4-year follow-up (9). However, they did not report the impact of the type of anticoagulant on all-cause mortality. To date, the use of DOACs in patients with cardiac amyloidosis and supraventricular tachycardia was minor in the published series (10, 11). No cohort study has reported a difference in prognosis according to the type of anticoagulant used. Our study shows that despite an apparent over-risk of VKAs, there is no difference in mortality between anticoagulants and DOACs can be used safely.
In contrast to the Mitrani study (9), our cohort showed a difference in terms of bleeding complications between groups of anticoagulants, which were more frequent among patients receiving VKA. This could be explained by the fact that patients receiving VKA had more impaired renal function and therefore a higher HAS-BLED hemorrhagic score, which would need to be proven by a multivariate analysis regarding bleeding complications (which could not be done due to a lack of events).
Furthermore, the high rate of bleeding complications in this population of CA must be noted and integrated into the benefit-risk balance of such a therapy. Indeed, we know that these are fragile patients with disturbed hemostasis and impaired renal function, which may explain such a high rate of hemorrhagic complications in comparison with the usual population of patients with atrial fibrillation.
Cardiac amyloidosis has been shown to expose to an over-risk of embolic events with an increased prevalence of atrial thrombus at transesophageal echocardiography (12) or surgery (13). However, despite this increased risk confirmed by our study, there is no difference in events between patients treated with VKA and patients treated with DOACs.
These results must be interpreted with caution, as the patient profile is not the same and the excess mortality of patients on VKAs could simply be explained by a selection bias induced by age and renal function that contraindicates the use of DOACs. Indeed, renal function has clearly been shown to be a major prognostic marker in patients with CA, particularly wtATTR (14).
Limitations
This study shares all the limitations and bias associated with a retrospective and single-center study. Although the number of deaths is sufficient to conclude on all-cause mortality, the number of thrombo-embolic and hemorrhagic events remains too small to draw definitive conclusions. Ultimately, true safety of DOACs will only be demonstrated by randomized study.
Conclusion
The choice of therapeutics to be used in CA differs in many ways from other heart failure patients. The thrombotic profile of these patients often requires the use of anticoagulation, the benefit of which may be offset by an increased risk of hemorrhage. The choice between VKA and DOACs can therefore sometimes be difficult. Our study shows that DAOCs can be used without an over-risk of event compared to the standard VKA treatment widely used in clinical practice.
On Behalf of the Toulouse Amyloidosis Research Network Collaborators
Blandine Acket, Department of Neurology, Purpan University Hospital, Toulouse, France; Laurent Alric, Department of Internal Medicine and Digestive Diseases, Purpan University Hospital, Toulouse, France; Christophe Bureau, Department of Hepatology-Gastroenterology, Rangueil University Hospital, Toulouse, France; Dominique Chauveau, Department of Nephrology and Referral Center for Rare Diseases, Rangueil University Hospital, Toulouse, France; Magali Colombat, Department of Pathology, IUCT Oncopôle, Toulouse, France; Audrey Delas, Department of Pathology, IUCT Oncopôle, Toulouse, France; Delphine Dupin-Deguine, Department of Genetic, Toulouse University Hospital, Toulouse, France; Stanislas Faguer, Department of Nephrology and Referral Center for Rare Diseases, Rangueil University Hospital, Toulouse, France; Bénédicte Puissant, Immunology Laboratory, Toulouse University Hospital, Toulouse, France; Grégory Pugnet, Department of Internal Medicine and Digestive Diseases, Purpan University Hospital, Toulouse, France; Grégoire Prévot, Department of Pneumology, Toulouse University Hospital, Toulouse, France; David Ribes, Department of Nephrology and Referral Center for Rare Diseases, Rangueil University Hospital, Toulouse, France; Laurent Sailler, Department of Internal Medicine, Toulouse University Hospital, Toulouse, France.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Author Contributions
OL designed and drove the study. EC analyzed and interpreted the clinical data and wrote the manuscript. KS and KR collected the clinical data. SC, VB, and PF collected the clinical events and the prognosis. YL-B performed the statistical analysis. AH, PC, MR, MG, and DC contributed to the discussion and the reviewing. PM led the study, revised the manuscript, and gave final approval of the version to be published. All authors contributed to the article and approved the submitted version.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fcvm.2021.742428/full#supplementary-material
Abbreviations
AA, Atrial arrhythmia; AL, light chain amyloidosis; ATTR, transthyretin amyloidosis; ATTRv, variant transthyretin amyloidosis; ATTRwt, wild-type transthyretin amyloidosis; CA, cardiac amyloidosis; DOAC, direct oral anticoagulant; VKA, vitamin K-antagonist.
References
1. Feng D, Edwards WD, Oh JK, Chandrasekaran K, Grogan M, Martinez MW, et al. Intracardiac thrombosis and embolism in patients with cardiac amyloidosis. Circulation. (2007) 116:2420–6. doi: 10.1161/CIRCULATIONAHA.107.697763
2. Anderson KP. Cardiac amyloidosis and the risks of cardioversion. J Am Coll Cardiol. (2019) 73:598–601. doi: 10.1016/j.jacc.2018.10.080
3. Kirchhof P, Benussi S, Kotecha D, Arbelo E, Bax JJ, Blomström-Lundqvist C, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur Heart J. (2016) 37:2893–962. doi: 10.1093/eurheartj/ehw210
4. Donnellan E, Elshazly MB, Vakamudi S, Wazni OM, Cohen JA, Kanj M, et al. No association between CHADS-VASc score and left atrial appendage thrombus in patients with transthyretin amyloidosis. JACC Clin Electrophysiol. (2019) 5:1473–4. doi: 10.1016/j.jacep.2019.10.013
5. Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, Blomström-Lundqvist C, et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. (2021) 42:373–498. doi: 10.1093/eurheartj/ehaa612
6. Sanchis K, Cariou E, Colombat M, Ribes D, Huart A, Cintas P, et al. Atrial fibrillation and subtype of atrial fibrillation in cardiac amyloidosis: clinical and echocardiographic features, impact on mortality. Amyloid Int J Exp Clin Investig. (2019) 26:128–38. doi: 10.1080/13506129.2019.1620724
7. Rodeghiero F, Tosetto A, Abshire T, Arnold DM, Coller B, James P, et al. ISTH/SSC bleeding assessment tool: a standardized questionnaire and a proposal for a new bleeding score for inherited bleeding disorders. J Thromb Haemost. (2010) 8:2063–5. doi: 10.1111/j.1538-7836.2010.03975.x
8. Granger CB, Alexander JH, McMurray JJ V, Lopes RD, Hylek EM, Hanna M, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med. (2011) 365:981–92. doi: 10.1056/NEJMoa1107039
9. Mitrani LR, De Los Santos J, Driggin E, Kogan R, Helmke S, Goldsmith J, et al. Anticoagulation with warfarin compared to novel oral anticoagulants for atrial fibrillation in adults with transthyretin cardiac amyloidosis: comparison of thromboembolic events and major bleeding. Amyloid Int J Exp Clin Investig. (2020) 28:30–4. doi: 10.1080/13506129.2020.1810010
10. Mints YY, Doros G, Berk JL, Connors LH, Ruberg FL. Features of atrial fibrillation in wild-type transthyretin cardiac amyloidosis: a systematic review and clinical experience. ESC Hear Fail. (2018) 5:772–9. doi: 10.1002/ehf2.12308
11. Longhi S, Quarta CC, Milandri A, Lorenzini M, Gagliardi C, Manuzzi L, et al. Atrial fibrillation in amyloidotic cardiomyopathy: prevalence, incidence, risk factors and prognostic role. Amyloid Int J Exp Clin Investig. (2015) 22:147–55. doi: 10.3109/13506129.2015.1028616
12. El-Am EA, Dispenzieri A, Melduni RM, Ammash NM, White RD, Hodge DO, et al. Direct current cardioversion of atrial arrhythmias in adults with cardiac amyloidosis. J Am Coll Cardiol. (2019) 73:589–97. doi: 10.1016/j.jacc.2018.10.079
13. Fayyaz AU, Bois MC, Dasari S, Padmanabhan D, Vrana JA, Stulak JM, et al. Amyloidosis in surgically resected atrial appendages: a study of 345 consecutive cases with clinical implications. Mod Pathol. (2020) 33:764–74. doi: 10.1038/s41379-019-0407-5
Keywords: cardiac amyloidosis, direct oral anticoagulants, vitamin K-antagonists (VKAs), prognosis, atrial arrhythmia
Citation: Cariou E, Sanchis K, Rguez K, Blanchard V, Cazalbou S, Fournier P, Huart A, Roussel M, Cintas P, Galinier M, Carrié D, Maury P, Lavie-Badie Y and Lairez O (2021) New Oral Anticoagulants vs. Vitamin K Antagonists Among Patients With Cardiac Amyloidosis: Prognostic Impact. Front. Cardiovasc. Med. 8:742428. doi: 10.3389/fcvm.2021.742428
Received: 16 July 2021; Accepted: 20 September 2021;
Published: 30 November 2021.
Edited by:
William Moody, Queen Elizabeth Hospital Birmingham, United KingdomReviewed by:
Joseph Bono, Queen Elizabeth Hospital Birmingham, United KingdomAndre Rodrigues Duraes, Federal University of Bahia, Brazil
Copyright © 2021 Cariou, Sanchis, Rguez, Blanchard, Cazalbou, Fournier, Huart, Roussel, Cintas, Galinier, Carrié, Maury, Lavie-Badie and Lairez. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Eve Cariou, cariou.e@chu-toulouse.fr
 Eve Cariou1,2*
Eve Cariou1,2*  Khailène Rguez
Khailène Rguez Olivier Lairez
Olivier Lairez