Abstract
Purpose
The purpose of this study was to compare the differences of arrhythmogenic substrate using high-density mapping in ventricular tachycardia (VT) patients with ischemic (ICM) vs non-ischemic cardiomyopathy (NICM).
Methods
Data from patients presenting for VT ablation from December 2016 to December 2020 at Westmead Hospital were reviewed.
Results
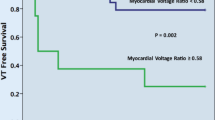
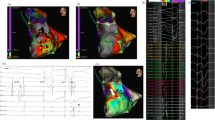
Sixty consecutive patients with structural heart disease (ICM 57%, NICM 43%, mean age 66 years) having catheter ablation of scar-related VT with pre-dominant left ventricular involvement were included. ICM was associated with larger proportion of dense scar area (bipolar; 19 [12–29]% vs 6 [3–10]%, P < 0.001, unipolar; 20 [12–32]% vs 11 [7–19]%, P = 0.01) compared with NICM. However, the scar ratio (unipolar dense scar [%]/bipolar dense scar [%]) was significantly higher in NICM patients (1.2 [0.8–1.7] vs 1.7 [1.3–2.3], P = 0.003). Larger scar area in ICM was paralleled by higher proportion of complex electrograms (6 [2–13] % vs 3 [1–5] %, P = 0.01), longer and wider voltage based conducting channels, higher incidence of late potential-based conducting channels, longer VT cycle-length (399 ± 80 ms vs 359 ± 68 ms, P = 0.04) and greater maximal stimulation-QRS interval among sites with good pace-map correlation (75 [51–99]ms vs 48 [31–73]ms, P = 0.02). Ventricular arrhythmia (VA) storm was more highly prevalent in ICM than NICM (50% vs 23%, P = 0.03). During the follow-up period, NICM had a significantly higher cumulative incidence for the VA recurrence than ICM (P = 0.03).
Conclusions
High-density multi-electrode catheter mapping of left ventricular arrhythmogenic substrate of NICM tends to show smaller dense scar area and higher scar ratio, compared with ICM, suggestive the extent of epicardial/intramural substrate, with paucity of substrate targets for ablation, which results in the worse outcomes with ablation.




Similar content being viewed by others
Availability of data and material
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
Change history
18 February 2022
A Correction to this paper has been published: https://doi.org/10.1007/s10840-022-01129-9
Abbreviations
- AAD:
-
Anti-arrhythmic drug
- ATP:
-
Anti-tachycardia pacing
- CF:
-
Contact force
- CL:
-
Cycle length
- CMR:
-
Cardiac magnetic resonance
- CS:
-
Coronary sinus
- EP:
-
Electrophysiology
- HCM:
-
Hypertrophic cardiomyopathy
- ICD:
-
Implantable cardioverter defibrillator
- ICM:
-
Ischemic cardiomyopathy
- IQR:
-
Interquartile range
- LAVA:
-
Local abnormal ventricular activation
- LV:
-
Left ventricle
- mA:
-
Milliamps
- mV:
-
Millivolts
- NICM:
-
Non-ischemic cardiomyopathy
- PES:
-
Programmed electrical stimulation
- RV:
-
Right ventricle
- SD:
-
Standard deviation
- SHD:
-
Structural heart disease
- VA:
-
Ventricular arrhythmia
- VT:
-
Ventricular tachycardia
References
Della Bella P, Baratto F, Tsiachris D, Trevisi N, Vergara P, Bisceglia C, et al. Management of ventricular tachycardia in the setting of a dedicated unit for the treatment of complex ventricular arrhythmias: long-term outcome after ablation. Circulation. 2013;127(13):1359–68. https://doi.org/10.1161/circulationaha.112.000872.
Gokoglan Y, Mohanty S, Gianni C, Santangeli P, Trivedi C, Gunes MF, et al. Scar homogenization versus limited-substrate ablation in patients with nonischemic cardiomyopathy and ventricular tachycardia. J Am Coll Cardiol. 2016;68(18):1990–8. https://doi.org/10.1016/j.jacc.2016.08.033.
Kumar S, Baldinger SH, Romero J, Fujii A, Mahida SN, Tedrow UB, et al. Substrate-based ablation versus ablation guided by activation and entrainment mapping for ventricular tachycardia: a systematic review and meta-analysis. J Cardiovasc Electrophysiol. 2016;27(12):1437–47. https://doi.org/10.1111/jce.13088.
Marchlinski F, Garcia F, Siadatan A, Sauer W, Beldner S, Zado E, et al. Ventricular tachycardia/ventricular fibrillation ablation in the setting of ischemic heart disease. J Cardiovasc Electrophysiol. 2005;16(Suppl 1):S59-70. https://doi.org/10.1111/j.1540-8167.2005.50210.x.
Marchlinski FE, Callans DJ, Gottlieb CD, Zado E. Linear ablation lesions for control of unmappable ventricular tachycardia in patients with ischemic and nonischemic cardiomyopathy. Circulation. 2000;101(11):1288–96. https://doi.org/10.1161/01.cir.101.11.1288.
Berte B, Relan J, Sacher F, Pillois X, Appetiti A, Yamashita S, et al. Impact of electrode type on mapping of scar-related VT. J Cardiovasc Electrophysiol. 2015;26(11):1213–23. https://doi.org/10.1111/jce.12761.
De Silva K, Virk S, Nalliah CJ, Campbell T, Kumar S. Multielectrode mapping versus point-by-point mapping for catheter ablation of ventricular tachycardia: a systematic review and meta-analysis. JACC Clinical electrophysiology. 2020;6(7):876–8. https://doi.org/10.1016/j.jacep.2020.04.007.
Nakahara S, Tung R, Ramirez RJ, Michowitz Y, Vaseghi M, Buch E, et al. Characterization of the arrhythmogenic substrate in ischemic and nonischemic cardiomyopathy implications for catheter ablation of hemodynamically unstable ventricular tachycardia. J Am Coll Cardiol. 2010;55(21):2355–65. https://doi.org/10.1016/j.jacc.2010.01.041.
Elliott P, Andersson B, Arbustini E, Bilinska Z, Cecchi F, Charron P, et al. Classification of the cardiomyopathies: a position statement from the European Society Of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2008;29(2):270–6. https://doi.org/10.1093/eurheartj/ehm342.
Fujii A, Nagashima K, Kumar S, Tanigawa S, Baldinger SH, Michaud GF, et al. Significance of inducible nonsustained ventricular tachycardias after catheter ablation for ventricular tachycardia in ischemic cardiomyopathy. Circ Arrhythm Electrophysiol. 2017;10(12):1–10. https://doi.org/10.1161/CIRCEP.117.005005.
Campbell T, Trivic I, Bennett RG, Anderson RD, Turnbull S, Pham T, et al. Catheter ablation of ventricular arrhythmia guided by a high-density grid catheter. J Cardiovasc Electrophysiol. 2020;31(2):474–84. https://doi.org/10.1111/jce.14351.
Anderson RD, Lee G, Trivic I, Campbell T, Pham T, Nalliah C, et al. Focal ventricular tachycardias in structural heart disease: prevalence, characteristics, and clinical outcomes after catheter ablation. JACC Clinical electrophysiology. 2020;6(1):56–69. https://doi.org/10.1016/j.jacep.2019.09.013.
Jais P, Maury P, Khairy P, Sacher F, Nault I, Komatsu Y, et al. Elimination of local abnormal ventricular activities: a new end point for substrate modification in patients with scar-related ventricular tachycardia. Circulation. 2012;125(18):2184–96. https://doi.org/10.1161/circulationaha.111.043216.
Hutchinson MD, Gerstenfeld EP, Desjardins B, Bala R, Riley MP, Garcia FC, et al. Endocardial unipolar voltage mapping to detect epicardial ventricular tachycardia substrate in patients with nonischemic left ventricular cardiomyopathy. Circ Arrhythm Electrophysiol. 2011;4(1):49–55. https://doi.org/10.1161/circep.110.959957.
Mountantonakis SE, Park RE, Frankel DS, Hutchinson MD, Dixit S, Cooper J, et al. Relationship between voltage map “channels” and the location of critical isthmus sites in patients with post-infarction cardiomyopathy and ventricular tachycardia. J Am Coll Cardiol. 2013;61(20):2088–95. https://doi.org/10.1016/j.jacc.2013.02.031.
Berruezo A, Acosta J, Fernandez-Armenta J, Pedrote A, Barrera A, Arana-Rueda E, et al. Safety, long-term outcomes and predictors of recurrence after first-line combined endoepicardial ventricular tachycardia substrate ablation in arrhythmogenic cardiomyopathy. Impact of arrhythmic substrate distribution pattern. A prospective multicentre study. Europace : European pacing, arrhythmias, and cardiac electrophysiology : journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology. 2017;19(4):607–16. doi: https://doi.org/10.1093/europace/euw212
Looi KL, Tang A, Agarwal S. Ventricular arrhythmia storm in the era of implantable cardioverter-defibrillator. Postgrad Med J. 2015;91(1079):519–26. https://doi.org/10.1136/postgradmedj-2015-133550.
Cronin EM, Bogun FM, Maury P, Peichl P, Chen M, Namboodiri N, et al. 2019 HRS/EHRA/APHRS/LAHRS expert consensus statement on catheter ablation of ventricular arrhythmias. Europace : European pacing, arrhythmias, and cardiac electrophysiology : journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology. 2019. https://doi.org/10.1093/europace/euz132
Yalin K, Golcuk E, Bilge AK, Aksu T, Buyukbayrak H, Tiryakioglu SK, et al. Combined analysis of unipolar and bipolar voltage mapping identifies recurrences after unmappable scar-related ventricular tachycardia ablation. Europace : European pacing, arrhythmias, and cardiac electrophysiology : journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology. 2015;17(10):1580-6. https://doi.org/10.1093/europace/euv013
Komatsu Y, Daly M, Sacher F, Derval N, Pascale P, Roten L, et al. Electrophysiologic characterization of local abnormal ventricular activities in postinfarction ventricular tachycardia with respect to their anatomic location. Heart Rhythm. 2013;10(11):1630–7. https://doi.org/10.1016/j.hrthm.2013.08.031.
Baldinger SH, Nagashima K, Kumar S, Barbhaiya CR, Choi EK, Epstein LM, et al. Electrogram analysis and pacing are complimentary for recognition of abnormal conduction and far-field potentials during substrate mapping of infarct-related ventricular tachycardia. Circ Arrhythm Electrophysiol. 2015;8(4):874–81. https://doi.org/10.1161/circep.114.002714.
Josephson ME, Anter E. Substrate mapping for ventricular tachycardia: assumptions and misconceptions. JACC Clinical Electrophysiology. 2015;1(5):341–52. https://doi.org/10.1016/j.jacep.2015.09.001.
Campos B, Jauregui ME, Marchlinski FE, Dixit S, Gerstenfeld EP. Use of a novel fragmentation map to identify the substrate for ventricular tachycardia in postinfarction cardiomyopathy. Heart Rhythm. 2015;12(1):95–103. https://doi.org/10.1016/j.hrthm.2014.10.002.
Ciaccio EJ, Ashikaga H, Coromilas J, Hopenfeld B, Cervantes DO, Wit AL, et al. Model of bipolar electrogram fractionation and conduction block associated with activation wavefront direction at infarct border zone lateral isthmus boundaries. Circ Arrhythm Electrophysiol. 2014;7(1):152–63. https://doi.org/10.1161/circep.113.000840.
Kumar S, Fujii A, Kapur S, Romero J, Mehta NK, Tanigawa S, et al. Beyond the storm: comparison of clinical factors, arrhythmogenic substrate, and catheter ablation outcomes in structural heart disease patients with versus those without a history of ventricular tachycardia storm. J Cardiovasc Electrophysiol. 2017;28(1):56–67. https://doi.org/10.1111/jce.13117.
Scott PA, Rosengarten JA, Murday DC, Peebles CR, Harden SP, Curzen NP, et al. Left ventricular scar burden specifies the potential for ventricular arrhythmogenesis: an LGE-CMR study. J Cardiovasc Electrophysiol. 2013;24(4):430–6. https://doi.org/10.1111/jce.12035.
Di Biase L, Santangeli P, Burkhardt DJ, Bai R, Mohanty P, Carbucicchio C, et al. Endo-epicardial homogenization of the scar versus limited substrate ablation for the treatment of electrical storms in patients with ischemic cardiomyopathy. J Am Coll Cardiol. 2012;60(2):132–41. https://doi.org/10.1016/j.jacc.2012.03.044.
Tung R, Mathuria NS, Nagel R, Mandapati R, Buch EF, Bradfield JS, et al. Impact of local ablation on interconnected channels within ventricular scar: mechanistic implications for substrate modification. Circ Arrhythm Electrophysiol. 2013;6(6):1131–8. https://doi.org/10.1161/circep.113.000867.
Wolf M, Sacher F, Cochet H, Kitamura T, Takigawa M, Yamashita S, et al. Long-term outcome of substrate modification in ablation of post-myocardial infarction ventricular tachycardia. Circ Arrhythm Electrophysiol. 2018;11(2):e005635. https://doi.org/10.1161/circep.117.005635.
Tung R, Josephson ME, Bradfield JS, Shivkumar K. Directional influences of ventricular activation on myocardial scar characterization: voltage mapping with multiple wavefronts during ventricular tachycardia ablation. Circ Arrhythm Electrophysiol. 2016;9(8):1–11. https://doi.org/10.1161/circep.116.004155.
Funding
Associate Professor Saurabh Kumar is supported by the NSW Early-mid Career Fellowship.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Yasuhito Kotake, Chrishan J. Nalliah, Timothy Campbell, Richard G. Bennett, Samual Turnbull and Saurabh Kumar. The first draft of the manuscript was written by Chrishan J. Nalliah and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the Western Sydney Local Health District Human Research Ethics Committee and complies with the Declaration of Helsinki.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
Dr. Kumar has received research grants from Abbott Medical and Biotronik; he has received honoraria from Biosense Webster, Abbott Medical, Biotronik and Sanofi Aventis. Mr Campbell has received speakers Honoria from Biosense Webster. Dr. Kotake is the recipient of Nihon Koden/Abbott arrhythmia Fellowship from the Japan Heart Rhythm Society. All other authors have reported no other relationships relevant to the contents of this paper to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yasuhito Kotake and Chrishan J. Nalliah are equal co-first authors.
The original online version of this article was revised: In Fig. 1 of this article "F: Pace map" should have been "H: Pace map".
Rights and permissions
About this article
Cite this article
Kotake, Y., Nalliah, C.J., Campbell, T. et al. Comparison of the arrhythmogenic substrate for ventricular tachycardia in patients with ischemic vs non-ischemic cardiomyopathy — insights from high-density, multi-electrode catheter mapping. J Interv Card Electrophysiol 66, 5–14 (2023). https://doi.org/10.1007/s10840-021-01088-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-021-01088-7