Abstract
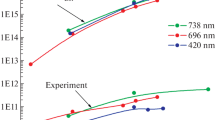
In this paper, the features of the process of argon radiation mixed with molecular and atomic impurities in condensing supersonic jets are studied experimentally. The mixture particles were activated by a well-focused electron beam. The dependence of the radiation intensity of individual argon lines on the gas-dynamic parameters in the jet is studied by changing the total density of the gas mixture due to changes in the stagnation pressure in the gas source chamber. The anomalous increase in the radiation intensity in a certain pressure range, which is different for different compositions of mixtures, was recorded on individual lines of atomic argon (Ar-I). At the same time, a similar effect was not detected in the spectrum of argon ions (Ar-II). It is established that anomaly was caused by the highly efficient molecular cluster mechanism of selective excitation of individual levels of argon atoms. It is absent in non-condensing jets and weakens at the stage of formation of large clusters. The main channels of energy transmission are discussed. The empirical model of the excitation-radiation process based on the obtained data was proposed.











Similar content being viewed by others
References
Khmel SY, Sharafutdinov RG (2000) Abstracts of invited lectures and contributed papers of 15th ESCAMPIG. Hungary 24A:384
Sharafutdinov RG, Khmel SY (2003) Plasma Chem Plasma Process 23(3):463–488. https://doi.org/10.1023/A:1023230931546
Golomb D, Good RE (1986) J Chem Phys 49:4176–4180. https://doi.org/10.1063/1.1670732
Madirbaev VZh, Zarvin AE, Korobeishchikov NG, Sharafutdinov RG (2002) Phys Solid State 44(3):515–517. https://doi.org/10.1134/1.1462691
Madirbaev VGh, Zarvin AE, Korobeishchikov NG (2013) In: XXXI International Conference on Phenomena in Ionized Gases (ICPIG XXXI Granada), PS2–085
Zarvin AE, Madirbaev VZh, Korobeishchikov NG (2014) In: XXI International Conference on Chemical Reactors (CHEMREACTOR‐21)
Dubrovin KA, Zarvin AE, Kalyada VV, Yaskin AS (2020) Tech Phys Lett 46(4):337–340. https://doi.org/10.1134/S1063785020040057
Zarvin AE, Khudozhitkov VE, Dubrovin KA, Kalyada VV, Yaskin AS (2020). J Phys Conf Ser. https://doi.org/10.1088/1742-6596/1683/3/032008
Zarvin AE, Kalyada VV, Korobeishchikov NG, Khodakov MD, Shmakov AA (2016) Instrum Exp Tech 59(2):294–301. https://doi.org/10.1134/S0020441216010176
Zarvin AE, Kalyada VV, Madirbaev VZh, Korobeishchikov NG, Khodakov MD, Yaskin AS, Khudozhitkov VE, Gimelshein SF (2017) IEEE Transact Pl Sci 45(5):819–827. https://doi.org/10.1109/TPS.2017.2682901
Korobeishchikov NG, Zarvin AE, Madirbaev VZh (2004) Tech Phys 49(8):973–981
Kazakov VV, Kazakov VG, Kovalev VS, Meshkov OI, Yatsenko AS (2017) Phys Scr 92:105002. https://doi.org/10.1088/1402-4896/aa822e
Bier K, Hagena O (1966) In: Rarefied Gas Dynamics. Adv Appl Mech: Proc. 4-th Internat. Symp., N.Y.-London, Acad. Press. p 2:260–278
Campargue R (1984) J Phys Chem 88(20):4466–4474
Biordi C (1977) Prog Energy Combust Sci 3(3):151–173
Crunelle B, Surdyk D, Pauwels JF, Sochet LR (1997) J Chem Phys 94:433–459
Even U (2014) Adv Chem 2014:636042. https://doi.org/10.1155/2014/636042
Smith JA, Driscoll JF (1975) J Fluid Mech 72(4):695–719
Zarvin AE, Khudozhitkov VE, Kalyada VV (2018) In: IOP Conf. Ser.: Mater. Sci. Eng. 387:012086. https://doi.org/10.1088/1757-899X/387/1/012086
Khudozhitkov VE, Zarvin AE, Kalyada VV (2018) J Phys: Conf Ser 1105:012112. https://doi.org/10.1088/1742-6596/1105/1/012112
Korobeishchikov NG, Zarvin AE, Madirbaev VZh, Sharafutdinov RG (2005) Pl Chem Pl Proc 25(4):319–349. https://doi.org/10.1007/s11090-004-3132-9
Hagena OF, Obert W (1972) J Chem Phys 56(5):1793–1802. https://doi.org/10.1063/1.1677455
Zarvin AE, Madirbaev VZh, Korobeishchikov NG, Gartvich GG, Sharafutdinov RG (2005) Tech Phys 50(11):1444–1450
Korobeishchikov NG, Zarvin AE, Kalyada VV, Schmakov AA (2012) Adv Mater Phys Chem 2:31–34. https://doi.org/10.4236/ampc.2012.24B009
Golomb D, Good RE, Bailey AB, Busby MR, Dawbarn R (1972) J Chem Phys 57(9):3844–3852. https://doi.org/10.1063/1.1678854
Smirnov BM (2003) Physics – uspekhi 46(6):589–628. https://doi.org/10.1070/PU2003v046n06ABEH001381
Zarvin AE, Kalyada VV, Khudozhitkov VE (2017) Thermophys Aeromech 24(5):671–681. https://doi.org/10.1134/S0869864317050031
Bird GA (1976) Phys Fluids 19:1486–1491. https://doi.org/10.1063/1.861351
Zarvin AE, Sharafutdinov RG (1979) Rarefied Gas Dynamics. Ed. R. Campargue. Paris, France. In: Proceedings of 11-th International Symposium, V. II, p 991–1000
Verheijen MJ, Beijerinck HCW, Renes WA, Verster NF (1984) Chem Phys 85(1):63–71. https://doi.org/10.1016/S0301-0104(84)85173-3
Schütte S, Buck U (2002) Intern J Mass Spectrom 220(2):183–192
NIST Chemistry WebBook, https://webbook.nist.gov/chemistry/ doi: https://doi.org/10.18434/T4D303
Hagena OF (1981) Surf Sci 106(1–3):101–116. https://doi.org/10.1016/0039-6028(81)90187-4
Buck U, Krohne R (1996) J Chem Phys 105(13):5408–5415. https://doi.org/10.1063/1.472406
Striganov AR, Sventitskii NS (1968) Plenum, N.Y. https://doi.org/10.1016/0584-8547(69)80033-9
Berry RS, Smirnov BM (2007) Physics – Uspekhi 48(4):345–388. https://doi.org/10.1070/PU2005v048n04ABEH002022
Zhukhovitskii DI (1995) J Chem Phys 103:9401–9407. https://doi.org/10.1063/1.470000
Bernstein ER (1995) Annu Rev Phys Chem 46:197–222
Baletto F, Ferrando R (2005) Rev Mod Phys 77(1):371–423
Lundwall M, Lindblad A, Bergersen H, Rander T, Öhrwall G, Tchaplyguine M, Peredkov S, Svensson S, Björneholm O (2006) J Phys B: At Mol Opt Phys 39:3321–3333. https://doi.org/10.1088/0953-4075/39/16/015
Lundwall M, Fink RF, Tchaplyguine M, Lindblad A, Öhrwall G, Bergersen H, Peredkov S, Rander T, Svensson S, Björneholm O (2006) J Phys B: At Mol Opt Phys 39:5225–5235. https://doi.org/10.1088/0953-4075/39/24/018
Malins A, Williams SR, Eggers J, Tanaka H, Royall CP (2011) J Non-Cryst Solids 357(2):760–766. https://doi.org/10.1016/j.jnoncrysol.2010.08.021
Richter C, Hollas D, Saak CM, Förstel M, Miteva T, Mucke M, Björneholm O, Sisourat N, Slavíček P, Hergenhahn U (2018) Nat Commun 9:4988. https://doi.org/10.1038/s41467-018-07501-6
Aladi M, Bolla R, Rácz P, Földes IB (2016) Nucl Instr Meth. In: Phys Res B, 369:68–71. https://doi.org/10.1016/j.nimb.2015.10.061
Acknowledgements
The study was conducted using the shared equipment at the Applied Physics Centre at NSU’s Physics Department with the financial support of the RFBR (grant no. 20-01-00332) and the Ministry of science and higher education of the Russian Federation, (project number FSUS-2020-0039).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zarvin, A.E., Madirbaev, V.Z., Dubrovin, K.A. et al. On the Mechanism of Ionic-Cluster Excitation of Argon Levels in Molecular Gas Mixtures. Plasma Chem Plasma Process 42, 247–265 (2022). https://doi.org/10.1007/s11090-021-10214-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-021-10214-2