Abstract
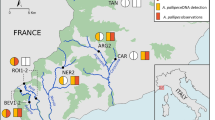
Invasive lionfish are considered to be one of the worst marine invaders primarily due to their threat as predators to native species. The distribution of lionfish across their invasive range has been fairly well characterized, but their presence within estuaries is largely unknown. While removal of invasive lionfish is the primary mode of managing the invasion, early detection in new locations could be valuable for documenting range expansion. Environmental DNA (eDNA) has become a useful tool to identify the presence of invasive species prior to establishment and may be especially valuable for systems that are challenging to survey. In this study, an environmental DNA assay was developed and successfully identified the presence of lionfish DNA in four riverine estuaries in the northern Gulf of Mexico. Detections were significantly higher in July but did not vary by location. Occupancy modeling shows that the assay is not exceptionally sensitive and may be underreporting the presence of lionfish, possibly due to the effect of salinity. The presence of invasive lionfish within estuaries could put native fish and invertebrates at risk, especially species that use these systems as nurseries. Ongoing monitoring using molecular methods may be useful in the management of invasive lionfish by providing early detection of fish moving into systems that neighbor known invaded regions.


Similar content being viewed by others
Data availability
Data will be made available upon request.
Code availability
Not applicable.
References
Albins MA, Hixon MA (2008) Invasive Indo-Pacific lionfish Pterois volitans reduce recruitment of Atlantic coral-reef fishes. Mar Ecol Prog Ser 367:233–238. https://doi.org/10.3354/meps07620
Alvaro LE, Lehrter JC, Fung M (2019) Spatial patterns of particulate organic carbon concentrations and isotopic signatures across a salinity gradient in a river dominated Estuary. Gulf Caribb Res 30:SC47–SC51. https://doi.org/10.18785/gcr.3001.18
Andruszkiewicz Allen E, Zhang WG, Lavery AC, Govindarajan AF (2021) Environmental DNA shedding and decay rates from diverse animal forms and thermal regimes. eDNA 3:492-514
Anton A, Simpson MS, Vu I (2014) Environmental and biotic correlates to lionfish invasion success in Bahamian coral reefs. PLoS ONE 9:1–10. https://doi.org/10.1371/journal.pone.0106229
Bajer PG, Ghosal R, Maselko M et al (2019) Biological control of invasive fish and aquatic invertebrates: a brief review with case studies. Manag Biol Invas 10:1. https://doi.org/10.3391/mbi.2019.10.2.02
Barbour AB, Montgomery ML, Adamson AA et al (2010) Mangrove use by the invasive lionfish Pterois volitans. Mar Ecol Prog Ser 401:291–294. https://doi.org/10.3354/meps08373
Barker BD, Horodysky AZ, Kerstetter DW (2018) Hot or not? Comparative behavioral thermoregulation, critical temperature regimes, and thermal tolerances of the invasive lionfish Pterois sp. versus native western North Atlantic reef fishes. Biol Invasions 20:45–58. https://doi.org/10.1007/s10530-017-1511-4
Barnes MA, Turner CR, Jerde CL et al (2014) Environmental conditions influence eDNA persistence in aquatic systems. Environ Sci Technol 48:1819–1827. https://doi.org/10.1021/es404734p
Buchheister A, Bonzek CF, Gartland J, Latour RJ (2013) Patterns and drivers of the demersal fish community of Chesapeake Bay. Mar Ecol Prog Ser 481:161–180. https://doi.org/10.3354/meps10253
Buxton AS, Groombridge JJ, Zakaria NB, Griffiths RA (2017) Seasonal variation in environmental DNA in relation to population size and environmental factors. Sci Rep 7:1–9. https://doi.org/10.1038/srep46294
Bylemans J, Furlan EM, Pearce L et al (2016) Improving the containment of a freshwater invader using environmental DNA (eDNA) based monitoring. Biol Invasions 18:3081–3089. https://doi.org/10.1007/s10530-016-1203-5
Carassou L, Dzwonkowski B, Hernandez FJ et al (2011) Environmental influences on juvenile fish abundances in a river-dominated coastal system. Mar Coast Fish 3:411–427. https://doi.org/10.1080/19425120.2011.642492
Côté IM, Smith NS (2018) The lionfish Pterois sp. invasion: has the worst-case scenario come to pass? J Fish Biol 92:660–689. https://doi.org/10.1111/jfb.13544
Costanza R, d’Arge R, de Groot R et al (1997) The value of the world’s ecosystem services and natural capital. Nature 387:253–260. https://doi.org/10.1038/387253a0
Dabruzzi TF, Bennett WA, Fangue NA (2017) Thermal ecology of red lionfish Pterois volitans from Southeast Sulawesi, Indonesia, with comparisons to other Scorpaenidae. Aquat Biol 26:1–14. https://doi.org/10.3354/ab00668
Dahl KA, Edwards MA, Patterson WF (2019) Density-dependent condition and growth of invasive lionfish in the northern Gulf of Mexico. Mar Ecol Prog Ser 623:145–159. https://doi.org/10.3354/meps13028
Dahl KA, Patterson WF, Robertson A, Ortmann AC (2017) DNA barcoding significantly improves resolution of invasive lionfish diet in the Northern Gulf of Mexico. Biol Invasions 19:1917–1933. https://doi.org/10.1007/s10530-017-1407-3
Darling JA, Mahon AR (2011) From molecules to management: adopting DNA-based methods for monitoring biological invasions in aquatic environments. Environ Res 111:978–988. https://doi.org/10.1016/j.envres.2011.02.001
Deiner K, Altermatt F (2014) Transport distance of invertebrate environmental DNA in a natural river. PLoS ONE 9:e88786. https://doi.org/10.1371/journal.pone.0088786
Dejean T, Valentini A, Duparc A et al (2011) Persistence of environmental DNA in freshwater ecosystems. PLoS ONE 6:8–11. https://doi.org/10.1371/journal.pone.0023398
DeRoy E, Scott R, Hussey N, MacIsaac H (2020) High predatory efficiency and abundance drive expected ecological impacts of a marine invasive fish. Mar Ecol Prog Ser 637:195–208. https://doi.org/10.3354/meps13251
Devkota J, Fang X (2014) Age of water and salt exchange in the Perdido and Wolf Bay System. World Environ Water Resour Congr 2014 Water Without Borders - Proc 2014 World Environ Water Resour Congr 2:1292–1301. https://doi.org/10.1061/9780784413548.130
Duke EM, Burton RS (2020) Efficacy of metabarcoding for identification of fish eggs evaluated with mock communities. Ecol Evol 10:3463–3476. https://doi.org/10.1002/ece3.6144
Farrington HL, Lance RF (2014) Development of genetic markers for environmental DNA (eDNA) monitoring of sturgeon. Ecosyst Manag Restor Res Progr 12. ERDC TN-EMRRP-RQ-02
Ficetola GF, Miaud C, Pompanon F, Taberlet P (2008) Species detection using environmental DNA from water samples. Biol Lett 4:423–425
Finnoff D, Shogren JF, Leung B, Lodge D (2007) Take a risk: preferring prevention over control of biological invaders. Ecol Econ 62:216–222. https://doi.org/10.1016/j.ecolecon.2006.03.025
Fishelson L (1997) Experiments and observations on food consumption, growth and starvation in Dendrochirus brachypterus and Pterois volitans (Pteroinae, Scorpaenidae). Environ Biol Fishes 50:391–403. https://doi.org/10.1023/A:1007331304122
Goldberg CS, Turner CR, Deiner K et al (2016) Critical considerations for the application of environmental DNA methods to detect aquatic species. Methods Ecol Evol 7:1299–1307. https://doi.org/10.1111/2041-210X.12595
Green SJ, Akins JL, CôtÉ IM (2011) Foraging behaviour and prey consumption in the Indo-Pacific lionfish on Bahamian coral reefs. Mar Ecol Prog Ser 433:159–167. https://doi.org/10.3354/meps09208
Harms-Tuohy CA, Schizas NV, Appeldoorn RS (2016) Use of DNA metabarcoding for stomach content analysis in the invasive lionfish Pterois volitans in Puerto Rico. Mar Ecol Prog Ser 558:181–191. https://doi.org/10.3354/meps11738
Harris HE, Fogg AQ, Allen MS et al (2020) Precipitous declines in northern Gulf of Mexico invasive lionfish populations following the emergence of an ulcerative skin disease. Sci Rep 10:1–17. https://doi.org/10.1038/s41598-020-58886-8
Harvey RG, Mazzotti FJ (2015) Public awareness and attitudes toward invasive lionfish: preliminary results from baseline survey interim report to the Florida Fish and Wildlife Conservation Commission. Interim Report to the Florida Fish and Wildlife Conservation Commission. March 2015, p 38
Janosik AM, Whitacker JM, VanTassel NM, Rider SJ (2021) Improved environmental DNA sampling scheme for Alabama sturgeon provides new insight into a species once presumed extinct. J of Appl Ichthyol 00:1–8. https://doi.org/10.1111/jai.14183
Jerde CL, Chadderton WL, Mahon AR, Renshaw MA, Corush J, Budny ML, Mysorekar S, Lodge DM (2013) Detection of Asian carp DNA as part of a Great Lakes basin-wide surveillance program. Can J Fish Aquat 70:522–526
Jud ZR, Layman CA, Lee JA, Arrington DA (2011) Recent invasion of a Florida (USA) estuarine system by lionfish Pterois volitans/p. miles. Aquat Biol 13:21–26. https://doi.org/10.3354/ab00351
Jud ZR, Nichols PK, Layman CA (2015) Broad salinity tolerance in the invasive lionfish Pterois spp. may facilitate estuarine colonization. Environ Biol Fishes 98:135–143. https://doi.org/10.1007/s10641-014-0242-y
Kearse M, Moir R, Wilson A et al (2012) Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28:1647–1649. https://doi.org/10.1093/bioinformatics/bts199
Kimball ME, Miller JM, Whitfield PE, Hare JA (2004) Thermal tolerance and potential distribution of invasive lionfish (Pterois volitans/miles complex) on the east coast of the United States. Mar Ecol Prog Ser 283:269–278. https://doi.org/10.3354/meps283269
Kindinger TL, Albins MA (2017) Consumptive and non-consumptive effects of an invasive marine predator on native coral-reef herbivores. Biol Invasions 19:131–146. https://doi.org/10.1007/s10530-016-1268-1
Klingström T, Bongcam-Rudloff E, Pettersson, OV (2018) A comprehensive model of DNA fragmentation for the preservation of high molecular weight DNA. BioRxiv, p.254276.
Klymus KE, Richter CA, Chapman DC, Paukert C (2015) Quantification of eDNA shedding rates from invasive bighead carp Hypophthalmichthys nobilis and silver carp Hypophthalmichthys molitrix. Biol Conserv 183:77–84. https://doi.org/10.1016/j.biocon.2014.11.020
Lee J, Webb BM, Dzwonkowski B et al (2019) Characteristics of exchange flow in a multiple inlet diurnal estuary: Mobile Bay, Alabama. J Mar Syst 191:38–50. https://doi.org/10.1016/j.jmarsys.2018.12.004
Lesser MP, Slattery M (2011) Phase shift to algal dominated communities at mesophotic depths associated with lionfish (Pterois volitans) invasion on a Bahamian coral reef. Biol Invasions 13:1855–1868. https://doi.org/10.1007/s10530-011-0005-z
Leung B, Lodge DM, Finnoff D et al (2002) An ounce of prevention or a pound of cure: bioeconomic risk analysis of invasive species. Proc R Soc B Biol Sci 269:2407–2413. https://doi.org/10.1098/rspb.2002.2179
Leung B, Mandrak NE (2007) The risk of establishment of aquatic invasive species: joining invasibility and propagule pressure. Proc R Soc B Biol Sci 274:2603–2609. https://doi.org/10.1098/rspb.2007.0841
Madenjian CP, Stapanian MA, Witzel LD et al (2011) Evidence for predatory control of the invasive round goby. Biol Invasions 13:987–1002. https://doi.org/10.1007/s10530-010-9884-7
Mauvisseau Q, Coignet A, Delaunay C et al (2018) Environmental DNA as an efficient tool for detecting invasive crayfishes in freshwater ponds. Hydrobiologia 805:163–175. https://doi.org/10.1007/s10750-017-3288-y
McDonald RB, Moody RM, Heck KL, Cebrian J (2016) Fish, macroinvertebrate and epifaunal communities in shallow coastal lagoons with varying seagrass cover of the Northern Gulf of Mexico. Estuaries Coasts 39:718–730. https://doi.org/10.1007/s12237-015-0031-7
Miralles L, Dopico E, Devlo-Delva F, Garcia-Vazquez E (2016) Controlling populations of invasive pygmy mussel (Xenostrobus securis) through citizen science and environmental DNA. Mar Pollut Bull 110:127–132. https://doi.org/10.1016/j.marpolbul.2016.06.072
Morris JA, Akins JL (2009) Feeding ecology of invasive lionfish (Pterois volitans) in the Bahamian archipelago. Environ Biol Fishes 86:389–398. https://doi.org/10.1007/s10641-009-9538-8
Morris JA, Whitfield PE (2009) Biology, ecology, control and management of the invasive Indo-Pacific lionfish: an updated integrated assessment. Integr Assess NOS NCCOS:57
Muñoz RC, Currin CA, Whitfield PE (2011) Diet of invasive lionfish on hard bottom reefs of the Southeast USA: insights from stomach contents and stable isotopes. Mar Ecol Prog Ser 432:181–193. https://doi.org/10.3354/meps09154
Nelson JA, Stallings CD, Landing WM, Chanton J (2013) Biomass transfer subsidizes nitrogen to offshore food webs. Ecosystems 16:1130–1138. https://doi.org/10.1007/s10021-013-9672-1
Peake J, Bogdanoff AK, Layman CA, et al (2018) Feeding ecology of invasive lionfish (Pterois volitans and Pterois miles) in the temperate and tropical western Atlantic
Peterson BJ (1999) Stable isotopes as tracers of organic matter input and transfer in benthic food webs: a review. Acta Oecologica 20:479–487
Pfleger MO, Rider SJ, Johnston CE, Janosik AM (2016) Saving the doomed: using eDNA to aid in detection of rare sturgeon for conservation (Acipenseridae). Glob Ecol Conserv 8:99–107. https://doi.org/10.1016/j.gecco.2016.08.008
Piaggio AJ, Engeman RM, Hopken MW et al (2014) Detecting an elusive invasive species: a diagnostic PCR to detect Burmese python in Florida waters and an assessment of persistence of environmental DNA. Mol Ecol Resour 14:374–380. https://doi.org/10.1111/1755-0998.12180
Ramos S, Amorim E, Elliott M, Cabral H, Bordalo AA (2012) Early life stages of fishes as indicators of estuarine ecosystem health. Ecol Ind 19:172–183
Rojas-Vélez S, Tavera J, Acero A (2019) Unraveling lionfish invasion: is Pterois volitans truly a morphologically novel predator in the Caribbean? Biol Invasions 21:1921–1931. https://doi.org/10.1007/s10530-019-01946-6
Secondi J, Dejean T, Valentini A et al (2016) Detection of a global aquatic invasive amphibian, Xenopus laevis, using environmental DNA. Amphib Reptil 37:131–136. https://doi.org/10.1163/15685381-00003036
Sepulveda AJ, Schabacker J, Smith S et al (2019) Improved detection of rare, endangered and invasive trout in using a new large-volume sampling method for eDNA capture. Environ DNA 1:227–237. https://doi.org/10.1002/edn3.23
Simmons M, Tucker A, Chadderton WL et al (2015) Active and passive environmental DNA surveillance of aquatic invasive species. Can J Fish Aquat Sci 73:76–83. https://doi.org/10.1139/cjfas-2015-0262
Smith NS, Green SJ, Akins JL et al (2017) Density-dependent colonization and natural disturbance limit the effectiveness of invasive lionfish culling efforts. Biol Invasions 19:2385–2399. https://doi.org/10.1007/s10530-017-1449-6
Stoeckle BC, Beggel S, Cerwenka AF, Motivans E, Kuehn R, Geist J (2017) A systematic approach to evaluate the influence of environmental conditions on eDNA detection success in aquatic ecosystems. PLoS ONE 12:e0189119. https://doi.org/10.1371/journal.pone.0189119
Strickler KM, Fremier AK, Goldberg CS (2015) Quantifying effects of UV-B, temperature, and pH on eDNA degradation in aquatic microcosms. Biol Conserv 183:85–92. https://doi.org/10.1016/j.biocon.2014.11.038
Thomsen PF, Kielgast J, Iversen LL et al (2012) Detection of a diverse marine fish fauna using environmental DNA from seawater samples. PLoS ONE 7:1–9. https://doi.org/10.1371/journal.pone.0041732
Tréguier A, Paillisson JM, Dejean T et al (2014) Environmental DNA surveillance for invertebrate species: advantages and technical limitations to detect invasive crayfish Procambarus clarkii in freshwater ponds. J Appl Ecol 51:871–879. https://doi.org/10.1111/1365-2664.12262
Wilcox CL, Motomura H, Matsunuma M, Bowen BW (2018) Phylogeography of lionfishes (Pterois) indicate taxonomic over splitting and hybrid origin of the invasive Pterois volitans. J Hered 109:162–175. https://doi.org/10.1093/jhered/esx056
Wimbush J, Frischer ME, Zarzynski JW, Nierzwicki-Bauer SA (2009) Eradication of colonizing populations of zebra mussels (Dreissena polymorpha) by early detection and SCUBA removal: Lake George, NY. Aquat Conserv Mar Freshw Ecosyst 19:703–713. https://doi.org/10.1002/aqc
Xia Z, Zhan A, Gao Y et al (2018) Early detection of a highly invasive bivalve based on environmental DNA (eDNA). Biol Invasions 20:437–447. https://doi.org/10.1007/s10530-017-1545-7
Acknowledgements
We thank Lauren Doubek, Sara Ousley, Patience Mosely, Kendra Babcock, Erika Headrick, Sean Gordon, Ashlie Johnson, Jessica Valek, Rachel Richardson, Jacob Dombrowski, Melissa Hebert, Alisha Honer, Jack Prior, Kerri Foote, and Katie Vaccaro-Garska for help with boat work for collection of samples. Thanks go to Dave Armstrong and Alabama Division of Wildlife and Freshwater Fisheries for boat ramp access for collection of eDNA samples. We thank Victoria Bogantes for GIS help with the map. We also thank Marine Services Center of the University of West Florida for boat support.
Author information
Authors and Affiliations
Contributions
JM Whitaker made substantial contributions to the design of the laboratory work, analysis and interpretation of data, and drafted the manuscript. AL Brower contributed to the conception and design of the work and performed laboratory work and initial analyses. AM Janosik made substantial contributions to the conception and design of the project, analysis and interpretation of data, and revised it critically for important intellectual content. All authors approved of the version to be published.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Whitaker, J.M., Brower, A.L. & Janosik, A.M. Invasive lionfish detected in estuaries in the northern Gulf of Mexico using environmental DNA. Environ Biol Fish 104, 1475–1485 (2021). https://doi.org/10.1007/s10641-021-01177-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-021-01177-6