Abstract
Purpose
Xijiao Dihuang Tang (XJDHT) is a classical formula of traditional Chinese medicine constituted of Cornu Bubali, Rehmannia glutinosa (Gaertn.) DC., Paeonia lactiflora Pall., and Paeonia suffruticosa Andrews. It was first mentioned in the medical classic “Beiji Qianjin Yaofang” written by Simiao Sun in Tang Dynasty. It shows very strong antipyretic and anticoagulant effects and has been clinically applied to treat various type of blood loss, purple and black spots, heat stroke, and glossitis. Kawasaki disease (KD) is considered as a kind of acute febrile illness in children with systemic vasculitis as the main lesions. The aim of this research is to clarify whether XJDHT can play a protective role in KD.
Methods
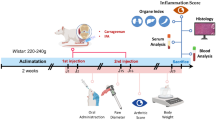
A mouse model of Candida albicans water-soluble fraction (CAWS)–induced coronary arteritis and a KD cell model with tumor necrosis factor (TNF)-α induction were employed to investigate the potential effect and mechanism of XJDHT on coronary artery injury in KD.
Results
Data showed that XJDHT remarkably alleviated the coronary artery injury of KD mice, as evidenced by reduced inflammation and downregulated expression of pro-inflammatory cytokines interleukin (IL)-1β and TNF-α. In vitro investigation showed that XJDHT could promote cell proliferation, inhibit cell apoptosis, and improve mitochondrial functions. Subsequent studies demonstrated that XJDHT rescued endothelial cell injury by PI3K/Akt-NFκB signaling pathway. Component analysis of XJDHT detected thirty-eight chemically active ingredients, including paeoniflorin, albiflorin, and paeoniflorigenone, which in in vitro experiments exhibited significant rescue effects on TNF-α–mediated endothelial cell injury.
Conclusion
Our findings demonstrated that XJDHT mitigated coronary artery injury of KD through suppressing endothelial cell damage via PI3K/Akt-NFκB signaling.






Similar content being viewed by others
Availability of Data and Materials
The datasets obtained in the present study are available from the corresponding author on reasonable request.
Abbreviations
- KD:
-
Kawasaki disease
- XJDHT:
-
Xijiao Dihuang Tang
- CAWS:
-
Candida albicans Water-soluble fraction
- TNF-α:
-
Tumor necrosis factor-α
- IL-1β:
-
Interleukin-1β
References
Singh S, Vignesh P, Burgner D. The epidemiology of Kawasaki disease: a global update. Arch Dis Child. 2015;100:1084–8.
Mccrindle BW, Rowley AH, Newburger JW, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a scientific statement for health professionals from the American Heart Association. Circulation. 2017;135:e927–99.
Liu R, Wang M, Duan JA. Antipyretic and antioxidant activities of the aqueous extract of Cornu bubali (water buffalo horn). Am J Chin Med. 2010;38:293–306.
Liu J, Pei T, Mu J, et al. Systems pharmacology uncovers the multiple mechanisms of Xijiao Dihuang decoction for the treatment of viral hemorrhagic fever. Evid Based Complement Alternat Med. 2016;2016:9025036.
Lu J, Yan J, Yan J, et al. Network pharmacology based research into the effect and mechanism of Xijiao Dihuang decoction against sepsis. Biomed Pharmacother. 2020;122: 109777.
He M, Chen Z, Martin M, et al. MiR-483 Targeting of CTGF suppresses endothelial-to-mesenchymal transition. Circ Res. 2017;120:354–65.
Jia C, Zhang J, Chen H, et al. Endothelial cell pyroptosis plays an important role in Kawasaki disease via HMGB1/RAGE/cathepsin B signaling pathway and NLRP3 inflammasome activation. Cell Death Dis. 2019;10:1–16.
Ueno K, Ninomiya Y, Hazeki D, Masuda K, Nomura Y, Kawano Y. Disruption of endothelial cell homeostasis plays a key role in the early pathogenesis of coronary artery abnormalities in Kawasaki disease. Sci Rep. 2017;7:43719.
Huiyuen JS, Duong TT, Yeung RSM. TNF-α is necessary for induction of coronary artery inflammation and aneurysm formation in an animal model of Kawasaki disease. J Immunol. 2006;176:6294–301.
Jiang C, Fang X, Jiang Y, et al. TNF-α induces vascular endothelial cells apoptosis through overexpressing pregnancy induced noncoding RNA in Kawasaki disease model. Int J Biochem Cell B. 2016;72:118–24.
Ohno N. Murine model of Kawasaki disease induced by mannoprotein-beta-glucan complex, CAWS, obtained from Candida albicans. Jpn J Infect Dis. 2004;57:9–10.
Stock AT, Hansen J, Sleeman MA, Mckenzie BS, Wicks IP. GM-CSF primes cardiac inflammation in a mouse model of Kawasaki disease. J Exp Med. 2016;213:1983–98.
Yang Y, Wang C, Zhuge Y, et al. Photodynamic antifungal activity of hypocrellin A against Candida albicans. Front Microbiol. 2019;10:1810.
Wang L, Fan H, He J, Wang L, Tian Z, Wang C. Protective effects of omega-3 fatty acids against Alzheimer’s disease in rat brain endothelial cells. Brain Behav. 2018;8: e01037.
Wu Y, Wang Y, Gong S, et al. Ruscogenin alleviates LPS-induced pulmonary endothelial cell apoptosis by suppressing TLR4 signaling. Biomed Pharmacother. 2020;125: 109868.
Ni W, Watts SW, Ng M. Elimination of vitamin D receptor in vascular endothelial cells alters vascular function. Hypertension. 2014;64:1290–8.
Gao L, Laude K, Cai H. Mitochondrial pathophysiology, reactive oxygen species, and cardiovascular diseases. Vet Clin N Am-Small. 2008;38:137–55.
Allan LA, Clarke PR. Apoptosis and autophagy: regulation of caspase-9 by phosphorylation. FEBS J. 2009;276:6063–73.
Ke F, Wang ZL, Song X, et al. Cryptotanshinone induces cell cycle arrest and apoptosis through the JAK2/STAT3 and PI3K/Akt/NFκB pathways in cholangiocarcinoma cells. Drug Des Dev Ther. 2017;11:1753–66.
Cao RJ, Li K, Xing WY, et al. Disabled-1 is down-regulated in clinical breast cancer and regulates cell apoptosis through NF-κB/Bcl-2/caspase-9. J Cell Mol Med. 2019;23:1622–7.
Wu R, Shen D, Sohun H, et al. MiR-186, a serum microRNA, induces endothelial cell apoptosis by targeting SMAD6 in Kawasaki disease. Int J Mol Med. 2018;41:1899–908.
Ichiyama T, Yoshitomi T, Nishikawa M, et al. NF-κB activation in peripheral blood monocytes/macrophages and T cells during acute Kawasaki disease. Clin Immunol. 2001;99:373–7.
Tian J, An X, Niu L. Correlation between NF-κB signal pathway-mediated caspase-4 activation and Kawasaki disease. Exp Ther Med. 2017;13:3333–6.
Kuhnel F, Zender L, Paul Y, et al. NFκB mediates apoptosis through transcriptional activation of Fas (CD95) in adenoviral hepatitis. J Biol Chem. 2000;275:6421–7.
Dhingra S, Sharma AK, Arora RC, Slezak J, Singal PK. IL-10 attenuates TNF-α-induced NFκB pathway activation and cardiomyocyte apoptosis. Cardiovasc Res. 2009;82:59–66.
Babaei MA, Huri HZ, Kamalidehghan B, Yeap SK, Ahmadipour F. Apoptotic induction and inhibition of NF-κB signaling pathway in human prostatic cancer PC3 cells by natural compound 2,2’-oxybis (4-allyl-1-methoxybenzene), biseugenol B, from Litsea costalis: an in vitro study. OncoTargets Ther. 2017;10:277–94.
An X, Lv H, Tian J, He X, Ling N. Role of the PTEN/PI3K/VEGF pathway in the development of Kawasaki disease. Exp Ther Med. 2016;11:1318–22.
Xie L, Xie H, Chen C, Tao Z, Zhang C, Cai L. Inhibiting the PI3K/AKT/NF-κB signal pathway with nobiletin for attenuating the development of osteoarthritis: in vitro and in vivo studies. Food & Funct. 2019;10:2161–75.
Boyd JH, Mathur S, Wang Y, Bateman RM, Walley KR. Toll-like receptor stimulation in cardiomyocytes decreases contractility and initiates an NF-κB dependent inflammatory response. Cardiovasc Res. 2006;72:384–93.
Schabbauer G, Tencati M, Pedersen B, Pawlinski R, Mackman N. PI3K-Akt pathway suppresses coagulation and inflammation in endotoxemic mice. Arterioscl Throm Vas. 2004;24:1963–9.
Chen T, Guo ZP, Jiao XY, et al. Peoniflorin suppresses tumor necrosis factor-α induced chemokine production in human dermal microvascular endothelial cells by blocking nuclear factor-κB and ERK pathway. Arch Dermatol Res. 2011;303:351–60.
Xu H, Song J, Gao X, et al. Paeoniflorin attenuates lipopolysaccharide-induced permeability of endothelial cells: involvements of F-actin expression and phosphorylations of PI3K/Akt and PKC. Inflammation. 2013;36:216–25.
Xu YJ, Mei Y, Shi XQ, et al. Albiflorin ameliorates memory deficits in APP/PS1 transgenic mice via ameliorating mitochondrial dysfunction. Brain Res. 2019;1719:113–23.
Huang Y, Ohno O, Suenaga K, Miyamoto K. Apoptosis-inducing activity and antiproliferative effect of paeoniflorigenone from moutan cortex. Biosci Biotechnol Biochem. 2017;81:1106–13.
Noval Rivas M, Arditi M. Kawasaki disease: pathophysiology and insights from mouse models. Nat Rev Rheumatol. 2020;16:391–405.
Funding
This research was supported by Zhejiang Provincial Science and Technology Project of Traditional Chinese Medicine (No. 2018ZZ019), the Natural Science Foundation of China (No. 82000469, No. 81970435, No. 81770502, and No. 81900240), Zhejiang Provincial Natural Science Foundation of China (No. LY18H020012), Wenzhou Science & Technology Bureau Foundation (No. Y20190064), and Zhejiang Provincial Medicine and Health Technology Project (No. 2021KY801).
Author information
Authors and Affiliations
Contributions
ZJ, ZGYZ, and RX contributed equally to this paper and conducted the experiments, data acquisition, and manuscript writing. NC, NC, WZW, and LHZ performed partial animal experiments and data analysis. CMP and JC supervised all the experiments and revised the manuscript.
Corresponding authors
Ethics declarations
Ethics Approval and Consent to Participate
All the necessary experiments were performed according to the National Institutes of Health Guide for the Care and Use of Laboratory Animals, and the protocols were approved by the Animal Care and Use Committee of Wenzhou Medical University (wydw 2017–0046).
Consent for Publication
Not applicable.
Research Involving Human Participants and/or Animals
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, J., Zhuge, Y., Rong, X. et al. Protective Roles of Xijiao Dihuang Tang on Coronary Artery Injury in Kawasaki Disease. Cardiovasc Drugs Ther 37, 257–270 (2023). https://doi.org/10.1007/s10557-021-07277-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-021-07277-w