Abstract
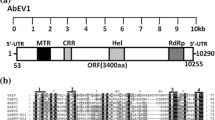
A double-stranded RNA (dsRNA) of approximately 16 kbp was isolated from symptomless common buckwheat (Fagopyrum esculentum) plants. The size of the dsRNA suggested that it was the replicative form of an endornavirus. The dsRNA was sequenced, and it consisted of 15,677 nt, containing a single open reading frame that potentially encoded a polyprotein of 5190 aa. The polyprotein contained conserved domains for a viral methyltransferase, viral RNA helicase 1, MSCRAMM family adhesion SdrC, UDP-glycosyltransferase, and viral RNA-dependent RNA polymerase 2. A site-specific nick in the plus strand was detected near the 5' end of the dsRNA. BLASTp analysis showed that the polyprotein shared the highest identity with the polyprotein of winged bean endornavirus 1. Results of phylogenetic analysis supported placing this novel virus from common buckwheat, which was provisionally named "Fagopyrum esculentum endornavirus 1", in the genus Alphaendornavirus of the family Endornaviridae.


Similar content being viewed by others
References
Bae T, Schneewind O (2003) The YSIRK-G/S motif of staphylococcal protein A and its role in efficiency of signal peptide processing. J Bacteriol 185:2910–2919. https://doi.org/10.1128/JB.185.9.2910-2919.2003
Fukuhara T (2019) Endornaviruses: persistent dsRNA viruses with symbiotic properties in diverse eukaryotes. Virus Genes 55:165–173. https://doi.org/10.1007/s11262-019-01635-5
Fukuhara T, Koga R, Aoki N, Yuki C, Yamamoto N, Oyama N, Undagawa T, Horiuchi H, Miyazaki S, Higashi Y, Takeshita M, Ikeda K, Arakawa M, Matsumoto N, Moriyama H (2006) The wide distribution of endornaviruses. Arch Virol 151:995–1002. https://doi.org/10.1007/s00705-005-0688-5
Hacker CV, Brasier CM, Buck KW (2005) A double-stranded RNA from a Phytophthora species is related to the plant endornaviruses and contains a putative UDP glycosyltransferase gene. J Gen Virol 86:1561–1570. https://doi.org/10.1099/vir.0.80808-0
Hershlag R, Escalante C, Rodrigues de Souto E, Kankhum S, Okada R, Valverde RA (2019) Occurrence of putative endornaviruses in non-cultivated plant species in south Louisiana. Arch Virol 164:1863–1868. https://doi.org/10.1007/s00705-019-04270-5
Khankhum S, Escalante C, Rodrigues de Souto E, Valverde RA (2017) Extraction and electrophoretic analysis of large dsRNAs from desiccated plant tissues infected with plant viruses and biotrophic fungi. Eur J Plant Pathol 147:431–441. https://doi.org/10.1007/s10658-016-1014-7
Kreft M (2016) Buckwheat phenolic metabolites in health and disease. Nutr Res Rev 29:30–39. https://doi.org/10.1017/S0954422415000190
Lefebvre A, Scalla R, Pfeiffer P (1990) The double-stranded RNA associated with the “447” cytoplasmic male sterility in Vicia faba is packaged together with its replicase in cytoplasmic membranous vesicles. Plant Mol Biol 14:477–490. https://doi.org/10.1007/BF00027494
Le SQ, Gascuel O (2008) An improved general amino acid replacement matrix. Mol Biol Evol 25:1307–1320. https://doi.org/10.1093/molbev/msn067
Lim S, Kim KH, Zhao F, Yoo RH, Igori D, Lee SH, Moon JS (2015) Complete genome sequence of a novel endornavirus isolated from hot pepper. Arch Virol 160:3153–3156. https://doi.org/10.1007/s00705-015-2616-7
Okada R, Ichinose S, Takeshita K, Urayama SI, Fukuhara T, Komatsu K, Arie T, Ishihara A, Egusa M, Kodama M, Moriyama H (2018) Molecular characterization of a novel mycovirus in Alternaria alternata manifesting two-sided effects: down-regulation of host growth and up-regulation of host plant pathogenicity. Virology 519:23–32. https://doi.org/10.1016/j.virol.2018.03.027
Okada R, Kiyota E, Moriyama H, Fukuhara T, Natsuaki T (2015) A simple and rapid method to purify viral dsRNA from plant and fungal tissue. J Gen Plant Pathol 81:103–107. https://doi.org/10.1007/s10327-014-0575-6
Okada R, Kiyota E, Moriyama H, Fukuhara T, Valverde RA (2017) Molecular and biological properties of an endornavirus infecting winged bean (Psophocarpus tetragonolobus). Virus Genes 53:141–145. https://doi.org/10.1007/s11262-016-1398-7
Okada R, Kiyota E, Moriyama H, Toshiyuki F, Valverde RA (2014) A new endornavirus species infecting Malabar spinach (Basella alba L.). Arch Virol 159:807–809. https://doi.org/10.1007/s00705-013-1875-4
Okada R, Kiyota E, Sabanadzovic S, Moriyama H, Fukuhara T, Saha P, Roossinck MJ, Severin A, Valverde RA (2011) Bell pepper endornavirus: molecular and biological properties, and occurrence in the genus Capsicum. J Gen Virol 92:2664–2673. https://doi.org/10.1099/vir.0.034686-0
Okada R, Yong CK, Valverde RA, Sabanadzovic S, Aoki N, Hotate S, Kiyota E, Moriyama H, Fukuhara T (2013) Molecular characterization of two evolutionarily distinct endornaviruses co-infecting common bean (Phaseolus vulgaris). J Gen Virol 94:220–229. https://doi.org/10.1099/vir.0.044487-0
Otulak-Koziel K, Koziel E, Escalante C, Valverde RA (2020) Ultrastructural analysis of cells from bell pepper (Capsicum annuum) infected with bell pepper endornavirus. Front Plant Sci 11:491. https://doi.org/10.3389/fpls.2020.00491
Sytar O, Brestic M, Zivcak M, Phan Tran LS (2016) The contribution of buckwheat genetic resources to health and dietary diversity. Curr Genomics 17:193–206. https://doi.org/10.2174/1389202917666160202215425
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729. https://doi.org/10.1093/molbev/mst197
Thompson JD, Higgins DG, Gibson TJ (1994) Clustal W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, positions-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680. https://doi.org/10.1093/nar/22.22.4673
Tuomivirta TT, Kaitera J, Hantula J (2009) A novel putative virus of Gremmeniella abietina type B (Ascomycota: Helotiaceae) has a composite genome with endornavirus affinities. J Gen Virol 90:2299–2305. https://doi.org/10.1099/vir.0.011973-0
Uchida K, Sakuta K, Ito A, Takahashi Y, Katayama Y, Omatsu T, Mizutani T, Arie T, Komatsu K, Fukuhara T, Uematsu S, Okada R, Moriyama H (2021) Two novel endornaviruses co-infecting a phytophthora pathogen of Asparagus officinalis modulate the developmental stages and fungicide sensitivities of the host oomycete. Front Microbiol 12:633502. https://doi.org/10.3389/fmicb.2021.633502
Valverde RA, Khalifa MA, Okada R, Fukuhara T, Sabanadzovic S, ICTV Report Consortium (2019) ICTV virus taxonomy profile: Endornaviridae. J Gen Virol 100:1204–1205. https://doi.org/10.1099/jgv.0.001277
Valverde RA, Sabanadzovic S, Rush MC (2011) Identification of Oryza sativa endornavirus in rice genotypes from breeding programmes in the United States. Plant Breed 130:271–274. https://doi.org/10.1111/j.1439-0523.2010.01778.x
Acknowledgments
This investigation was partially supported by a subsidy based on the three kinds of electric power laws for science and technology promotion of power plants and other nuclear energy facilities siting prefecture from Ministry of Education, Culture, Sports, Science and Technology. We wish to thank the United States Department of Agriculture, National Institute of Food and Agriculture, for partial support for this investigation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Robert H. A. Coutts.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Okada, R., Moriyama, H., Ogawara, T. et al. Complete nucleotide sequence of an alphaendornavirus isolated from common buckwheat (Fagopyrum esculentum). Arch Virol 166, 3483–3486 (2021). https://doi.org/10.1007/s00705-021-05264-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-021-05264-y