Abstract
Background
The rate of saphenous vein graft (SVG) occlusion within the first year of bypass graft surgery is 15%. The CHA2DS2-VASc score is used to predict the risk of thromboembolic events in patients with nonvalvular atrial fibrillation. We aimed to evaluate the predictive role of the CHA2DS2-VASc score in the estimation of intracoronary thrombus burden in patients with acute myocardial infarction (AMI) who underwent SVG-PCI.
Methods
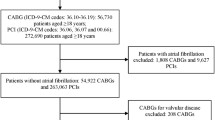
We retrospectively evaluated 221 patients who were admitted with AMI and underwent PCI of SVGs at the Department of Cardiology in the Turkiye Yuksek Ihtisas Education and Research Hospital between 2012 and 2018. The study population was divided into two groups according to their Thrombolysis in Myocardial Infarction (TIMI) thrombus grade: low thrombus burden (LTB; TIMI 0–3) and high thrombus burden (HTB; TIMI 4 and 5).
Results
The study included 221 patients with a mean age of 63.3 ± 6.7 years. The patients with HTB had significantly higher CHA2DS2-VASc scores (p < 0.001) compared with LTB patients. Univariate and multivariate regression analysis demonstrated that both CHA2DS2-VASc score (OR: 1.573, 95% CI: 1.153–2.147, p = 0.004) as a continuous variable and a binary cut-off level of the CHA2DS2-VASc score > 3 (OR: 3.876, 95% CI: 1.705–8.808, p = 0.001) were significantly associated with HTB. The ability of the CHA2DS2-VASc score to predict HTB burden was evaluated by receiver-operating characteristics analysis curve analysis. The optimum cut-off value of the CHA2DS2-VASc score for predicting HTB was 3 (with a sensitivity of 67.9% and a specificity of 69.3%) according to the Youden index.
Conclusion
The CHA2DS2-VASc score can be used as an easy practical tool to predict HTB in AMI patients undergoing SVG-PCI.
Zusammenfassung
Hintergrund
Die Verschlussrate für V.-saphena-Transplantate (SVG) innerhalb des ersten Jahres nach der Bypass-Operation liegt bei 15%. Zur Vorhersage des Risikos thromboembolischer Ereignisse bei Patienten mit nichtvalvulärem Vorhofflimmern wird der CHA2DS2-VASc-Score verwendet. Ziel der vorliegenden Arbeit war es, die prädiktive Bedeutung des CHA2DS2-VASc-Scores bei der Einschätzung der intrakoronaren Thrombuslast für Patienten mit akutem Myokardinfarkt (AMI) zu ermitteln, bei denen eine perkutane koronare Intervention (PCI) an einem SVG erfolgte.
Methoden
Retrospektiv wurden 221 Patienten untersucht, die wegen AMI stationär aufgenommen wurden und bei denen zwischen 2012 und 2018 am Department of Cardiology im Turkiye Yuksek Ihtisas Education and Research Hospital eine SVG-PCI durchgeführt wurde. Die Studienpopulation wurde entsprechend ihrem Thrombusgrad gemäß Thrombolysis in Myocardial Infarction (TIMI) in 2 Gruppen unterteilt: niedrige Thrombuslast (LTB; TIMI 0–3) und hohe Thrombuslast (HTB; TIMI 4 und 5).
Ergebnisse
In die Studie wurden 221 Patienten mit einem Durchschnittsalter von 63,3 ± 6,7 Jahren aufgenommen. Patienten mit HTB wiesen signifikant höhere CHA2DS2-VASc-Scores auf (p < 0,001) als LTB-Patienten. In der univariaten und multivariaten Regressionsanalyse zeigte sich, dass der CHA2DS2-VASc-Score (Odds Ratio, OR: 1,573; 95 %-Konfidenzintervall, 95 %-KI: 1,153–2,147; p = 0,004) als kontinuierliche Variable und auch ein binärer Grenzwert mit einem CHA2DS2-VASc-Score > 3 (OR: 3,876; 95 %-KI: 1,705–8,808; p = 0,001) in signifikanter Weise mit HTB assoziiert waren. Die Eignung des CHA2DS2-VASc-Scores zur Vorhersage der HTB wurde durch eine Kurvenanalyse mit Receiver-Operating-Characteristics-Analyse beurteilt. Der optimale Grenzwert für den CHA2DS2-VASc-Score zur Vorhersage von HTB betrug 3 (mit einer Sensitivität von 67,9 % und einer Spezifität von 69,3 %) gemäß Youden-Index.
Schlussfolgerung
Der CHA2DS2-VASc-Score kann als einfaches praktisches Instrument zur Vorhersage einer HTB bei AMI-Patienten, die mittels SVG-PCI behandelt werden, eingesetzt werden.

Similar content being viewed by others
References
Lopes RD, Hafley GE, Allen KB, Ferguson TB, Peterson ED, Harrington RA et al (2009) Endoscopic versus open vein-graft harvesting in coronary-artery bypass surgery. N Engl J Med 361:235–244. https://doi.org/10.1056/NEJMoa0900708
Neumann FJ, Sousa-Uva M, Ahlsson A, Alfonso F, Banning AP, Benedetto U et al (2019) 2018 ESC/EACTS guidelines on myocardial revascularization. Eur Heart J 40:87–165. https://doi.org/10.1093/eurheartj/ehy394
Lee MS, Park SJ, Kandzari DE, Kirtane AJ, Fearon WF, Brilakis ES et al (2011) Saphenous vein graft intervention. JACC Cardiovasc Interv 4:831–843. https://doi.org/10.1016/j.jcin.2011.05.014
Owens CD (2010) Adaptive changes in autogenous vein grafts for arterial reconstruction: clinical implications. J Vasc Surg 51:736–746. https://doi.org/10.1016/j.jvs.2009.07.102
Parang P, Arora R (2009) Coronary vein graft disease: pathogenesis and prevention. Can J Cardiol 25:e57–62. https://doi.org/10.1016/s0828-282x(09)70486-6
Campeau L, Enjalbert M, Lespérance J, Bourassa MG, Kwiterovich P Jr., Wacholder S et al (1984) The relation of risk factors to the development of atherosclerosis in saphenous-vein bypass grafts and the progression of disease in the native circulation. A study 10 years after aortocoronary bypass surgery. N Engl J Med 311:1329–1332. https://doi.org/10.1056/nejm198411223112101
Motwani JG, Topol EJ (1998) Aortocoronary saphenous vein graft disease: pathogenesis, predisposition, and prevention. Circulation 97:916–931. https://doi.org/10.1161/01.cir.97.9.916
Brilakis ES, Rao SV, Banerjee S, Goldman S, Shunk KA, Holmes DR Jr. et al (2011) Percutaneous coronary intervention in native arteries versus bypass grafts in prior coronary artery bypass grafting patients: a report from the national cardiovascular data registry. JACC Cardiovasc Interv 4:844–850. https://doi.org/10.1016/j.jcin.2011.03.018
Dash D (2014) An update on coronary bypass graft intervention. Heart Asia 6:41–45. https://doi.org/10.1136/heartasia-2013-010478
Cameron A, Kemp HG Jr., Green GE (1988) Reoperation for coronary artery disease. 10 years of clinical follow-up. Circulation 78:I158–162
O’Connor GT, Malenka DJ, Quinton H, Robb JF, Kellett MA Jr., Shubrooks S et al (1999) Multivariate prediction of in-hospital mortality after percutaneous coronary interventions in 1994–1996. Northern New England cardiovascular disease study group. J Am Coll Cardiol 34:681–691. https://doi.org/10.1016/s0735-1097(99)00267-3
Kirtane AJ, Heyman ER, Metzger C, Breall JA, Carrozza JP Jr. (2008) Correlates of adverse events during saphenous vein graft intervention with distal embolic protection: a PRIDE substudy. JACC Cardiovasc Interv 1:186–191. https://doi.org/10.1016/j.jcin.2008.01.002
January CT, Wann LS, Calkins H, Chen LY, Cigarroa JE, Cleveland JC Jr. et al (2019) 2019 AHA/ACC/HRS focused update of the 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American college of cardiology/American heart association task force on clinical practice guidelines and the heart rhythm society. Heart Rhythm 16:e66–e93. https://doi.org/10.1016/j.hrthm.2019.01.024
Chua SK, Lo HM, Chiu CZ, Shyu KG (2014) Use of CHADS2 and CHA2DS2-VASc scores to predict subsequent myocardial infarction, stroke, and death in patients with acute coronary syndrome: data from Taiwan acute coronary syndrome full spectrum registry. PLoS One 9:e111167. https://doi.org/10.1371/journal.pone.0111167
Satılmış S, Durmuş G (2020) Predictive accuracy of CHA(2)DS(2)-VASc score in determining the high thrombus burden in patients with non-ST-elevation myocardial infarction. Acta Cardiol. https://doi.org/10.1080/00015385.2019.1707934
Ipek G, Onuk T, Karatas MB, Gungor B, Osken A, Keskin M et al (2016) CHA2DS2-VASc score is a predictor of no-reflow in patients with ST-segment elevation myocardial infarction who underwent primary percutaneous intervention. Angiology 67:840–845. https://doi.org/10.1177/0003319715622844
Keskin K, Sezai Yıldız S, Çetinkal G, Aksan G, Kilci H, Çetin Ş et al (2017) The value of CHA(2)DS(2)VASC score in predicting all-cause mortality in patients with ST-segment elevation myocardial infarction who have undergone primary Percutaneous coronary intervention. Acta Cardiol Sin 33:598–604. https://doi.org/10.6515/acs20170723a
Thygesen K, Alpert JS, Jaffe AS, Chaitman BR, Bax JJ, Morrow DA et al (2018) Fourth universal definition of myocardial infarction. J Am Coll Cardiol 72:2231–2264. https://doi.org/10.1016/j.jacc.2018.08.1038
Camm AJ, Kirchhof P, Lip GY, Schotten U, Savelieva I, Ernst S et al (2010) Guidelines for the management of atrial fibrillation: the task force for the management of atrial fibrillation of the European society of cardiology (ESC). Eur Heart J 31:2369–2429. https://doi.org/10.1093/eurheartj/ehq278
Lang RM, Bierig M, Devereux RB, Flachskampf FA, Foster E, Pellikka PA et al (2005) Recommendations for chamber quantification: a report from the American society of echocardiography’s guidelines and standards committee and the chamber Quantification writing group, developed in conjunction with the European association of echocardiography, a branch of the European society of cardiology. J Am Soc Echocardiogr 18:1440–1463. https://doi.org/10.1016/j.echo.2005.10.005
Roffi M, Patrono C, Collet JP, Mueller C, Valgimigli M, Andreotti F et al (2016) 2015 ESC guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: task force for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation of the European society of cardiology (ESC). Eur Heart J 37:267–315. https://doi.org/10.1093/eurheartj/ehv320
Gibson CM, de Lemos JA, Murphy SA, Marble SJ, McCabe CH, Cannon CP et al (2001) Combination therapy with abciximab reduces angiographically evident thrombus in acute myocardial infarction: a TIMI 14 substudy. Circulation 103:2550–2554. https://doi.org/10.1161/01.cir.103.21.2550
Niccoli G, Burzotta F, Galiuto L, Crea F (2009) Myocardial no-reflow in humans. J Am Coll Cardiol 54:281–292. https://doi.org/10.1016/j.jacc.2009.03.054
Falk E, Thuesen L (2003) Pathology of coronary microembolisation and no reflow. Heart 89:983–985. https://doi.org/10.1136/heart.89.9.983
Ellis SG, Lincoff AM, Miller D, Tcheng JE, Kleiman NS, Kereiakes D et al (1998) Reduction in complications of angioplasty with abciximab occurs largely independently of baseline lesion morphology. EPIC and EPILOG investigators. Evaluation of 7E3 for the prevention of Ischemic complications. Evaluation of PTCA to improve long-term outcome with abciximab GPIIb/IIIa receptor blockade. J Am Coll Cardiol 32:1619–1623. https://doi.org/10.1016/s0735-1097(98)00403-3
Fernández-Ortiz A, Badimon JJ, Falk E, Fuster V, Meyer B, Mailhac A et al (1994) Characterization of the relative thrombogenicity of atherosclerotic plaque components: implications for consequences of plaque rupture. J Am Coll Cardiol 23:1562–1569. https://doi.org/10.1016/0735-1097(94)90657-2
Tanboga IH, Topcu S, Aksakal E, Kalkan K, Sevimli S, Acikel M (2014) Determinants of angiographic thrombus burden in patients with ST-segment elevation myocardial infarction. Clin Appl Thromb Hemost 20:716–722. https://doi.org/10.1177/1076029613483169
Baim DS, Wahr D, George B, Leon MB, Greenberg J, Cutlip DE et al (2002) Randomized trial of a distal embolic protection device during percutaneous intervention of saphenous vein aorto-coronary bypass grafts. Circulation 105:1285–1290
Wang X, Pei C, Bai Y, Dai Q, Deng X, Liu Y et al (2019) Predictive value of CHA(2)DS(2)-VASc score for ischemic events in patients undergoing percutaneous coronary intervention. Angiology 70:878–886. https://doi.org/10.1177/0003319718804661
Huang FY, Huang BT, Pu XB, Yang Y, Chen SJ, Xia TL et al (2017) CHADS(2), CHA(2)DS(2)-VASc and R(2)CHADS(2) scores predict mortality in patients with coronary artery disease. Intern Emerg Med 12:479–486. https://doi.org/10.1007/s11739-017-1608-x
Biancari F, Asim Mahar MA, Kangasniemi OP (2013) CHADS2 and CHA2DS2-VASc scores for prediction of immediate and late stroke after coronary artery bypass graft surgery. J Stroke Cerebrovasc Dis 22:1304–1311. https://doi.org/10.1016/j.jstrokecerebrovasdis.2012.11.004
Mirbolouk F, Gholipour M, Salari A, Shakiba M, Kheyrkhah J, Nikseresht V et al (2018) CHA2DS2-VASc score predict no-reflow phenomenon in primary percutaneous coronary intervention in primary percutaneous coronary intervention. J Cardiovasc Thorac Res 10:46–52. https://doi.org/10.15171/jcvtr.2018.08
Oikonomou E, Mourouzis K, Vogiatzi G, Siasos G, Deftereos S, Papaioannou S et al (2018) Coronary microcirculation and the no-reflow phenomenon. CPD 24:2934–2942. https://doi.org/10.2174/1381612824666180911122230
Durante A, Camici PG (2015) Novel insights into an “old” phenomenon: the no reflow. Int J Cardiol 187:273–280. https://doi.org/10.1016/j.ijcard.2015.03.359
Di Carli MF, Janisse J, Grunberger G, Ager J (2003) Role of chronic hyperglycemia in the pathogenesis of coronary microvascular dysfunction in diabetes. J Am Coll Cardiol 41:1387–1393. https://doi.org/10.1016/s0735-1097(03)00166-9
Ashoori A, Pourhosseini H, Ghodsi S, Salarifar M, Nematipour E, Alidoosti M et al (2019) CHA2DS2-VASc score as an independent predictor of suboptimal reperfusion and short-term mortality after primary PCI in patients with acute ST segment elevation myocardial infarction. Medicina. https://doi.org/10.3390/medicina55020035
Duman H, Çinier G, Bakırcı EM, Duman H, Şimşek Z, Hamur H et al (2019) Relationship between C‑reactive protein to albumin ratio and thrombus burden in patients with acute coronary syndrome. Clin Appl Thromb Hemost 25:1076029618824418. https://doi.org/10.1177/1076029618824418
Eid-Lidt G, Gaspar J, Adames AE, Damas de Los Santos F, Valdez RI, Ramírez-Gutiérrez AE et al (2010) Long-term outcomes of saphenous vein graft stenting compared with native coronary artery stenting in patients with previous coronary artery bypass graft surgery. Arch Cardiol Mex 80:3–9
Sdringola S, Assali AR, Ghani M, Moustapha A, Achour H, Yusuf SW et al (2001) Risk assessment of slow or no-reflow phenomenon in aortocoronary vein graft percutaneous intervention. Catheter Cardiovasc Interv 54:318–324. https://doi.org/10.1002/ccd.1290
Morishima I, Sone T, Mokuno S, Taga S, Shimauchi A, Oki Y et al (1995) Clinical significance of no-reflow phenomenon observed on angiography after successful treatment of acute myocardial infarction with percutaneous transluminal coronary angioplasty. Am Heart J 130:239–243. https://doi.org/10.1016/0002-8703(95)90434-4
Bayramoğlu A, Taşolar H, Kaya A, Tanboğa İH, Yaman M, Bektaş O et al (2018) Prediction of no-reflow and major adverse cardiovascular events with a new scoring system in STEMI patients. J Interven Cardiol 31:144–149. https://doi.org/10.1111/joic.12463
Gürbak İ, Panç C, Şahin AA, Derviş E, Yıldız İ, Güler A et al (2020) CHA2DS2-VASc score as a predictor of no-reflow phenomenon after saphenous vein graft percutaneous coronary intervention in patients with non-ST-segment elevation acute coronary syndromes. Kardiol Pol 78:1129–1136. https://doi.org/10.33963/kp.15603
Yip HK, Chen MC, Chang HW, Hang CL, Hsieh YK, Fang CY et al (2002) Angiographic morphologic features of infarct-related arteries and timely reperfusion in acute myocardial infarction: predictors of slow-flow and no-reflow phenomenon. Chest 122:1322–1332. https://doi.org/10.1378/chest.122.4.1322
Avci E, Yildirim T, Aydin G, Kiris T, Dolapoglu A, Kadi H et al (2018) Combining clinical predictors to better predict for the no-reflow phenomenon. Eur Rev Med Pharmacol Sci 22:4987–4994. https://doi.org/10.26355/eurrev_201808_15639
Hashemi-Jazi M, Hosseini SM, Gholamrezaei A (2017) Factors associated with the no-reflow phenomenon following percutaneous intervention of saphenous vein coronary bypass grafts. ARYA Atheroscler 13:221–229
Kojima S, Sakamoto T, Ishihara M, Kimura K, Miyazaki S, Tei C et al (2004) The white blood cell count is an independent predictor of no-reflow and mortality following acute myocardial infarction in the coronary interventional era. Ann Med 36:153–160. https://doi.org/10.1080/07853890310021553
Hong YJ, Jeong MH, Choi YH, Ko JS, Lee MG, Kang WY et al (2009) Predictors of no-reflow after percutaneous coronary intervention for culprit lesion with plaque rupture in infarct-related artery in patients with acute myocardial infarction. J Cardiol 54:36–44. https://doi.org/10.1016/j.jjcc.2009.03.003
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
O. Maden, Ö. Çakmak Karaaslan, Y. Kanal, İ. Yakut, N. M. Yaman, H. C. Könte, K. G. Balcı, M. T. Selçuk and H. Selçuk declare that they have no competing interests.
For this article no studies with human participants or animals were performed by any of the authors. All studies performed were in accordance with the ethical standards indicated in each case.
Rights and permissions
About this article
Cite this article
Maden, O., Çakmak Karaaslan, Ö., Kanal, Y. et al. Association of CHA2DS2-VASc score with thrombus burden in patients with acute myocardial infarction undergoing SVG-PCI. Herz 47, 456–464 (2022). https://doi.org/10.1007/s00059-021-05070-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00059-021-05070-x