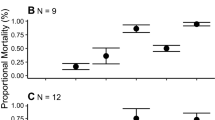
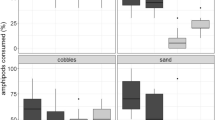
Abstract
Prey fish species can respond to the presence of predators using a variety of antipredatory behaviors to reduce their risk of being consumed. As such behaviors may also imply fitness costs, the ability of prey to perceive and appropriately respond to the predation threat is crucial to determine the persistence of its populations. Little is known whether antipredatory behaviors adopted by prey fish species are predator-specific or a general strategy employed regardless of predator species. Here, we used two phylogenetically close piscivorous fish species (Hoplias sp. 2 and Hoplerythrinus unitaeniatus) with similar morphology and foraging strategy (ambushing) to test whether prey (Moenkhausia sanctaefilomenae and Astyanax lacustris) behavioral responses were related to predator species identity. We video recorded and evaluated prey refuge use, habitat segregation, shoal formation, and mobility in microcosms before and after the addition of each predator. We found that M. sanctaefilomenae responded to the addition of Hoplias sp. 2 by reducing its mobility and keeping as far as possible from the predator location in a vigilant state, while shoal formation was employed regardless of predator species. On the other hand, A. lacustris had a consistent response to the presence of predators with no adjustment for predator species. We conclude that antipredatory responses may vary according to predator characteristics not obviously related to their morphology and foraging behavior, such as predator activity level and habitat use.



Similar content being viewed by others
Data availability
The authors declare that data are available upon request.
Code availability
The authors declare that code is available upon request.
References
Abozaid A, Tsang B, Gerlai R (2020) The effects of small but abrupt change in temperature on the behavior of larval zebrafish. Physiol Behav 227:113–169. https://doi.org/10.1016/j.physbeh.2020.113169
Abrahams MV, Pink M, Klassen C (2009) Predator avoidance. In: Encyclopedia of life sciences (ELS). Wiley, Chichester
Agostinho AA, Thomaz SM, Gomes LC, Baltar SLSMA (2007) Influence of the macrophyte Eichhornia azurea on fish assemblage of the Upper Paraná River floodplain (Brazil). Aquatic Ecol 41:611–619. https://doi.org/10.1007/s10452-007-9122-2
Arthur AD, Pech RP, Dickman CR (2004) Habitat structure mediates the non-lethal effects of predation on enclosed populations of house mice. J Anim Ecol 73:867–877. https://doi.org/10.1111/j.0021-8790.2004.00864.x
Bean CW, Winfield IJ (1995) Habitat use and activity patterns of roach (Rutilus rutilus (L.)), rudd (Scardinius erythrophthalmus (L.)), perch (Perca fluviatilis L.) and pike (Esox lucius L.) in the laboratory: the role of predation threat and structural complexity. Ecol Freshw Fish 4:37–46. https://doi.org/10.1111/j.1600-0633.1995.tb00025.x
Bifi AG (2013) Revisão taxonômica das espécies do grupo Hoplias malabaricus (Bloch, 1794) (Characiformes: Erythrinidae) da bacia do rio do Prata. (Master thesis, Universidade Estadual de Maringá, Brazil). http://nou-rau.uem.br/nou-rau/document/?code=vtls000205331
Blomberg SP, Garland JRT, Ives AR (2003) Testing for phylogenetic signal in comparative data: behavioral traits are more labile. Evolution 57:717–745. https://doi.org/10.1111/j.0014-3820.2003.tb00285.x
Borner KK, Krause S, Mehner T et al (2015) Turbidity affects social dynamics in Trinidadian guppies. Behav Ecol Sociobiol 69:645–651. https://doi.org/10.1007/s00265-015-1875-3
Brown C, Warburton K (1997) Predator recognition and anti-predator responses in the rainbowfish Melanotaenia eachamensis. Behav Ecol Sociobiol 41:61–68. https://doi.org/10.1007/s002650050364
Chelini MC, Willemart RH, Hebets EA (2009) Costs and benefits of freezing behaviour in the harvestman Eumesosoma roeweri (Arachnida, Opiliones). Behav Process 82:153–159. https://doi.org/10.1016/j.beproc.2009.06.001
Croft DP, Arrowsmith BJ, Bielby J, Skinner K, White E, Couzin ID, Magurran AE, Ramnarine I, Krause J (2003) Mechanisms underlying shoal composition in the Trinidadian guppy, Poecilia reticulata. Oikos 100:429–438. https://doi.org/10.1034/j.1600-0706.2003.12023.x
Davis S, Lukeman R, Schaerf TM, Ward AJW (2017) Familiarity affects collective motion in shoals of guppies (Poecilia reticulata). R Soc Open Sci 4(9):170312. https://doi.org/10.1098/rsos.170312
de Almeida VLL, Hahn NS, Vazzoler AEAM (1997) Feeding patterns in five predatory fishes of the high Parana River floodplain (PR, Brazil). Ecol Freshw Fish 6:123–133. https://doi.org/10.1111/j.1600-0633.1997.tb00154.x
deRoos AM, McCauley E, Wilson WG (1991) Mobility versus density-limited predator-prey dynamics on different spatial scales. Proc R Soc B 246:117–122. https://doi.org/10.1098/rspb.1991.0132
Dias MLGG, Eiras JC, Machado MH, Souza GTR, Pavanelli GC (2003) The life cycle of Clinostomum complanatum Rudolphi, 1814 (Digenea, Clinostomidae) on the floodplain of the high Paraná river, Brazil. Parasitol Res 89:506–508. https://doi.org/10.1007/s00436-002-0796-z
Dias RM, Silva JCB, Gomes LC, Agostinho AA (2017) Effects of macrophyte complexity and hydrometric level on fish assemblages in a Neotropical floodplain. Env Biol Fish 100:703–716. https://doi.org/10.1007/s10641-017-0597-y
Diehl S (1988) Foraging efficiency of three freshwater fishes: effects of structural complexity and light. Oikos 53:207–214. https://doi.org/10.2307/3566064
Dill LM, Heithaus MR, Walters CJ (2003) Behaviorally mediated indirect interactions in marine communities and their conservation implications. Ecology 84:1151–1157. https://doi.org/10.1890/0012-9658(2003)084[1151:BMIIIM]2.0.CO;2
DiRienzo N, Pruitt JN, Hedrick AV (2013) The combined behavioural tendencies of predator and prey mediate the outcome of their interaction. Anim Behav 86:317–322. https://doi.org/10.1016/j.anbehav.2013.05.020
Fernandes MN, Moron SE (1996) Respiratory organs in erythrinid fishes. In: Val AL, Randall DJ, MacKinley D (eds) Proceedings of Physiology of Tropical Fishes. American Fisheries Society, San Francisco, pp 93–110
Ferrari MCO, Sih A, Chivers DP (2009) The paradox of risk allocation: a review and prospectus. Anim Behav 78:579–585. https://doi.org/10.1016/j.anbehav.2009.05.034
Figueiredo BRS, Mormul RP, Thomaz SM (2015) Swimming and hiding regardless of the habitat: prey fish do not choose between a native and a non-native macrophyte species as a refuge. Hydrobiologia 746:285–290. https://doi.org/10.1007/s10750-014-2096-x
Figueiredo BRS, Fiori LF, Keppeler FW, Mormul RP, Benedito E (2018) Non-lethal effects of a native and a non-native piscivorous fish on the interaction between a mesopredator and benthic and pelagic invertebrates. Aquat Invasions 13:553–563. https://doi.org/10.3391/ai.2018.13.4.12
Figueiredo BRS, Granzotti RV, Fiori LF, Nieman CL, Benedito E (2020) Cascading effects of predation risk under high organic and inorganic turbidity: impacts on individuals and shoals of a mesopredator fish. Aquat Ecol 54:855–868. https://doi.org/10.1007/s10452-020-09782-w
Fox J, Weisberg S (2011) An R companion to applied regression. Sage, Thousand Oaks
Gomes LC, Bulla CK, Agostinho AA, Vasconcelos LP, Miranda LE (2012) Fish assemblage dynamics in a Neotropical floodplain relative to aquatic macrophytes and the homogenizing effect of a flood pulse. Hydrobiologia 685:97–107. https://doi.org/10.1007/s10750-011-0870-6
Granzotti RV, Muniz CM, Gomes LC (2018) Habitat complexity does not influence prey consumption in an experimental three-level trophic chain. Iheringia Sér Zool 108:1–8. https://doi.org/10.1590/1678-4766e2018028
Guariento DR, Luttberg B, Mehner T, Esteves FA (2014) The effect of predation pressure and predator adaptive foraging on the relative importance of consumptive and non-consumptive predator net effects in a freshwater model system. Oikos 123:705–713. https://doi.org/10.1111/j.1600-0706.2013.01201.x
Hahn NS, Fugi R, Andrian IF (2004) Trophic ecology of the fish assemblages. In: Thomaz SM, Agostinho AA, Hahn NS (eds) The Upper Parana River and its floodplain. Backhuys Publishers, Leiden, pp 247–270
Hamilton WD (1971) Geometry for the selfish herd. J Theor Biol 31:295–311
Hettyey A, Thonhauser KE, Bókony V, Penn DJ, Hoi H, Griggio M (2016) Naive tadpoles do not recognize recent invasive predatory fishes as dangerous. Ecology 97:2975–2985. https://doi.org/10.1002/ecy.1532
Huey RB, Pianka ER (1981) Ecological consequences of foraging mode. Ecology 62:991–999
Jiao J, Pilyugin SS, Osenberg CW (2016) Random movement of predators can eliminate trophic cascades in marine protected areas. Ecosphere 7:e01421. https://doi.org/10.1002/ecs2.1421
Júlio HF Jr, Tós DC, Agostinho AA, Pavanelli CS (2009) A massive invasion of fish species after eliminating a natural barrier in the upper Rio Paraná basin. Neotrop Ichthyol 7:709–719. https://doi.org/10.1590/S1679-62252009000400021
Klecka J, Boukal DS (2013) Foraging and vulnerability traits modify predator-prey body mass allometry: freshwater macroinvertebrates as a case study. J Anim Ecol 82:1031–1041. https://doi.org/10.1111/1365-2656.12078
Kovalenko KE, Dibble ED, Agostinho AA, Pelicice FM (2010) Recognition of non-native peacock bass, Cichla kelberi by native prey: testing the naivete hypothesis. Biol Invasions 12:3071–3080. https://doi.org/10.1007/s10530-010-9698-7
Krause J, Ruxton GD (2002) Living in groups. Oxford University Press, New York
Lautala T, Hirvonen H (2008) Antipredator behaviour of naive Arctic charr young in the presence of predator odours and conspecific alarm cues. Ecol Freshw Fish 17:78–85. https://doi.org/10.1111/j.1600-0633.2007.00261.x
Lima SL (1998) Nonlethal effects in the ecology of predator-prey interactions. Bioscience 48:25–34. https://doi.org/10.2307/1313225
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation: a review and prospectus. Can J Zool 68:619–640
Lopes TM, Cunha ER, Silva JCB, Behrend RDL, Gomes LC (2015) Dense macrophytes influence the horizontal distribution of fish in floodplain lakes. Environ Biol Fish 98:1741–1755. https://doi.org/10.1007/s10641-015-0394-4
Luz-Agostinho KDG, Agostinho AA, Gomes LC, Júlio HF Jr (2008) Influence of flood pulses on diet composition and trophic relationships among piscivorous fish in the upper Parana River floodplain. Hydrobiologia 607:187–198. https://doi.org/10.1007/s10750-008-9390-4
Meerhoff M, Iglesias C, Teixeira-de Mello F, Clemente JM, Jensen E, Lauridsen TL, Jeppesen E (2007) Effects of habitat complexity on community structure and predator avoidance behaviour of littoral zooplankton in temperate versus subtropical shallow lakes. Freshw Biol 52:1009–1021. https://doi.org/10.1111/j.1365-2427.2007.01748.x
Menezes NA, Weitzman SH, Oyakawa OT, Lima FCT, Castro RMC, Weitzman MJ (2007) Peixes de Água doce da mata Atlântica. MZUSP, São Paulo
Miller JRB, Ament JM, Schmitz OJ (2014) Fear on the move: predator hunting mode predicts variation in prey mortality and plasticity in prey spatial response. J Anim Ecol 83:214–222. https://doi.org/10.1111/1365-2656.12111
Moron SE, Andrade CA, Fernandes MN (2009) Response of mucous cells of the gills of traíra (Hoplias malabaricus) and jeju (Hoplerythrinus unitaeniatus) (Teleostei: Erythrinidae) to hypo- and hyper-osmotic ion stress. Neotrop Ichthyol 7:491–498. https://doi.org/10.1590/S1679-62252009000300017
Nannini MA, Belk MC (2006) Antipredator responses of two native stream fishes to an introduced predator: does similarity in morphology predict similarity in behavioural response? Ecol Freshw Fish 15:453–463. https://doi.org/10.1111/j.1600-0633.2006.00177.x
Olsson J, Svanbäck R, Eklöv P (2007) Effects of resource level and habitat type on behavioral and morphological plasticity in Eurasian perch. Oecologia 152:48–56. https://doi.org/10.1007/s00442-006-0588-8
Ota RR, Deprá GC, Graça WJ, Pavanelli CS (2018) Peixes da planície de inundação do alto rio Paraná e áreas adjacentes: revised, annotated and updated. Neotrop Ichthyol 16:e170094. https://doi.org/10.1590/1982-0224-20170094
Oufiero CE, Whitlow KR (2016) The evolution of phenotypic plasticity in fish swimming. Curr Zool 62:475–488. https://doi.org/10.1093/cz/zow084
Pavlov DS, Kasumyan AO (2000) Patterns and mechanisms of schooling behavior in fish: a review. J Ichthyol 40:63–231
Person L, Eklov P (1995) Prey refuges affecting interactions between piscivorous perch and juvenile perch and roach. Ecology 76:70–81
Petry AC, Agostinho AA, Piana PA, Gomes LC (2007) Effects of temperature on prey consumption and growth in mass of juvenile trahira Hoplias aff. malabaricus (Bloch, 1794). J Fish Biol 70:1855–1864. https://doi.org/10.1111/j.1095-8649.2007.01461.x
Preisser EL, Bolnick DI (2008) The many faces of fear: comparing the pathways and impacts of nonconsumptive predator effects on prey populations. PLoS ONE 3:e2465. https://doi.org/10.1371/journal.pone.0002465
Preisser EL, Orrock JL, Schmitz OJ (2007) Predator hunting mode and habitat domain alter nonconsumptive effects in predator–prey interactions. Ecology 88:2744–2751. https://doi.org/10.1890/07-0260.1
R Core Team (2018). R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Sansom A, Lind J, Cresswell W (2009) Individual behavior and survival: the roles of predator avoidance, foraging success, and vigilance. Behav Ecol 20(6):1168–1174. https://doi.org/10.1093/beheco/arp110
Savino J, Stein R (1982) Predator-prey interaction between largemouth bass and bluegills as influenced by simulated, submersed vegetation. Trans Am Fish Soc 111:255–266
Schmitz OJ (2005) Behavior of predators and prey and links with population-level processes. In: Barbosa P, Castellanos I (eds) Ecology of predator–prey interactions. Oxford University Press, Oxford, pp 256–278
Schmitz OJ (2008) Effects of predator hunting mode on grassland ecosystem function. Science 319:952–954. https://doi.org/10.1126/science.1152355
Schmitz OJ (2017) Predator and prey functional traits: understanding the adaptive machinery driving predator–prey interactions. F1000Research 6:1767. https://doi.org/10.12688/f1000research.11813.1
Schmitz OJ, Suttle KB (2001) Effects of top predator species on direct and indirect interactions in a food web. Ecology 82:2072–2081. https://doi.org/10.1890/0012-9658(2001)082[2072:EOTPSO]2.0.CO;2
Schmitz OJ, Kriva V, Ovadia O (2004) Trophic cascades: the primacy of trait-mediated indirect interactions. Ecol Lett 7:153–163. https://doi.org/10.1111/j.1461-0248.2003.00560.x
Sih A (1987) Predators and prey lifestyles: an evolutionary and ecological overview. In: McKerfoot WC, Sih A (eds) Predation: direct and indirect impacts on aquatic communities. University Press New England, Hanover, pp 203–224
Strauss SY, Lau JA, Carroll SP (2006) Evolutionary responses of natives to introduced species: what do introductions tell us about natural communities? Ecol Lett 9:354–371
Stuart-Smith RD, White RWG, Barmuta LA (2008) A shift in the habitat use pattern of a lentic galaxiid fish: an acute behavioural response to an introduced predator. Environ Biol Fish 82:93–100
Sweeney K, Cusack B, Armagost F, O’Brien T, Keiser CN, Pruitt JN (2013) Predator and prey activity levels jointly influence the outcome of long-term foraging bouts. Behav Ecol 24:1205–1210. https://doi.org/10.1093/beheco/art052
Venables WN, Ripley BD (2002) Modern applied statistics with S. Springer, New York
Volterra V (1926) Fluctuations in the abundance of a species considered mathematically. Nature 118:558–560
Ward AJW, Hart PJB (2003) The effects of kin and familiarity on interactions between fish. Fish Fish 4:348–358. https://doi.org/10.1046/j.1467-2979.2003.00135.x
Werner EE, Peacor SD (2003) A review of trait-mediated indirect interactions in ecological communities. Ecology 84:1083–1100. https://doi.org/10.1890/0012-9658(2003)084[1083:AROTII]2.0.CO;2
Werner EE, Gilliam JF, Hall DJ, Mittelbach GG (1983) An experimental test of the effects of predation risk on habitat use in fish. Ecology 64:1540–1548
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer, New York
Yofukuji KY, Gonino GMR, Alves GHZ, Lopes TM, Figueiredo BRS (2021) Acute ecotoxicity of exposure to sugarcane ashes on the behaviour of predator and prey fish species. Water Air Soil Pollut 232:312. https://doi.org/10.1007/s11270-021-05256-3
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer, New York
Acknowledgements
We thank K. Cogliati and anonymous reviewers for comments made to improve this manuscript. We are grateful to Valmir A. Capatti, Igor P. Affonso, Ragna W. Tavares, Herick Santana, Valmir A. Teixeira, and Sebastião Rodrigues for field assistance and Angelo A. Agostinho, Pitágoras Piana, Norma Hahn, and Lisiane Hahn for insightful comments on the initial manuscript version. This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES/PROEX)—Finance Code 001 (scholarships awarded to TML, RVG, AGO and MTB and funding to research) and Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (logistical support).
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES/PROEX).
Author information
Authors and Affiliations
Contributions
Conceived and designed the investigation: TML and LCG. Performed field and/or laboratory work: TML, RVG, AGO, and MTB. Analyzed the data: TML, RVG, AGO, MTB, and BRSF. All authors contributed to writing and reviewing the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
This experimental research was approved by the Instituto Chico Mendes de Conservação da Biodiversidade (SISBIO/ ICMBIO; License no. 22442–1) and the Ethical Board for the use of experimental animals from the Universidade Estadual de Maringá (CEUA; Technical Advice no. 123/2010).
Consent to participate
N/A.
Consent to publication
N/A.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lopes, T.M., Granzotti, R.V., Oliveira, A.G. et al. Phylogenetically and morphologically close ambush piscivores can elicit different antipredatory responses in characid prey. Environ Biol Fish 104, 1279–1292 (2021). https://doi.org/10.1007/s10641-021-01154-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-021-01154-z