Abstract
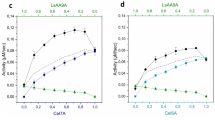
This paper studies the characteristics of the kinetic interaction of lytic polysaccharide monooxygenases (LPMOs) from Thielavia terrestris (TtLPMO), Penicillium verruculosum (PvLPMO), and Trichoderma reesei (TrLPMO) with purified cellobiohydrolases (CBH I and CBH II) and endoglucanase II (EG II) of P. verruculosum during the enzymatic destruction of microcrystalline (Avicel) and amorphous cellulose. TtLPMO belongs to the C1-type of LPMOs, while PvLPMO and TrLPMO are of the mixed C1/C4-type, according to the generally accepted classification of this class of oxidases. Under the action of any of the three LPMO together with CBH II or EG II on Avicel or amorphous cellulose, respectively, the enzymes displayed synergism, which was manifested in an increase in the yield of reducing sugars (RS). The synergism was expressed to the greatest extent in the initial reaction period. The mixtures of PvLPMO and TrLPMO with CBH I also demonstrated synergism on Avicel as a substrate, while TtLPMO, on the contrary, had an inhibitory effect on RS formation from cellulose with CBH I. The observed synergism and antagonism between LPMOs and CBH II/CBH I can be explained in terms of a simple kinetic scheme, in which the first enzyme forms additional ends of polysaccharide molecules that serve as a substrate or inhibitor for the second enzyme.


Similar content being viewed by others
REFERENCES
Lynd, L.R., Weimer, P.J., van Zyl, W.H., and Pretorius, I.S., Microbiol. Mol. Biol. Rev., 2002, vol. 66, no. 3, pp. 506–577. https://doi.org/10.1128/MMBR.66.3.506-577.2002
Tiwari, R., Nain, L., Labrou, N.E., and Shukla, P., Crit. Rev. Microbiol., 2018, vol. 44, no. 2, pp. 244–257. https://doi.org/10.1080/1040841X.2017.1337713
Vaaje-Kolstad, G., Westereng, B., Horn, S.J., Liu, Z., Zgai, H., Sorlie, M., and Eijsink, V.G.H., Science, 2010, vol. 330, pp. 219–222. https://doi.org/10.1126/science.1192231
Quinlan, R.J., Sweeney, M.D., Leggio, L.L., Otten, H., Poulsen, J.-C.N., Johansen, K.S., Krogh, K.B.R.M., Jorgensen, C.I., Tovborg, M., Anthonsen, A., Tryfona, T., Walter, C.P., Dupree, P., Xu, F., Davies, G.J., and Walton, P.H., Proc. Natl. Acad. Sci. U. S. A., 2011, vol. 108, no. 37, pp. 15079–15084. https://doi.org/10.1073/pnas.1105776108
Horn, S.J., Vaaje-Kolstad, G., Westereng, B., and Eijsink, V.G.H., Biotechnol. Biofuels, 2012, vol. 5. Article no. 45. https://doi.org/10.1186/1754-6834-5-45
Pollegioni, L., Tonin, F., and Rosini, E., FEBS J., 2015, vol. 282, no. 7, pp. 1190–1213. https://doi.org/10.1111/febs.13224
Levasseur, A., Drula, E., Lombard, V., Coutinho, P.M., and Henrissat, B., Biotechnol. Biofuels, 2013, vol. 6. Article no. 41. https://doi.org/10.1186/1754-6834-6-41
Agger, J.W., Isaksen, T., Varnai, A., Vidal-Melgosa, S., Willats, W.G.T., Ludwig, R., Horn, S.J., Eijsink, V.G.H., and Westereng, B., Proc. Natl. Acad. Sci. U. S. A., 2014, vol. 111, no. 17, pp. 6287–6292. https://doi.org/10.1073/pnas.1323629111
Frommhagen, M., Sforza, S., Westphal, A.H., Visser, J., Hinz, S.W.A., Koetsier, M.J., van Berkel, W.J.H., Gruppen, H., and Kabel, M.A., Biotechnol. Biofuels, 2015, vol. 8. Article no. 101. https://doi.org/10.1186/s13068-015-0284-1
Jensen, M.S., Klinkenberg, G., Bissaro, B., Chylenski, P., Vaaje-Kolstad, G., Kvitvang, H.F., Nærdal, G.K., Sletta, H., Forsberg, Z., and Eijsink, V.G.H., J. Biol. Chem., 2019, vol. 294, no. 50, pp. 19349–19364.https://doi.org/10.1074/jbc.RA119.010056
Phillips, C.M., Beeson, W.T., Cate, J.H., and Marletta, M.A., ACS Chem. Biol., 2011, vol. 6, no. 12, pp. 1399–1406. https://doi.org/10.1021/cb200351y
Li, X., Beeson, W.T., Phillips, C.M., Marletta, M.A., and Cate, J.H.D., Structure, 2012, vol. 20, no. 6, pp. 1051–1061. https://doi.org/10.1016/j.str.2012.04.002
Hemsworth, G.R., Davies, G.J., and Walton, P.H., Curr. Opin. Struct. Biol., 2013, vol. 23, no. 5, pp. 660–668. https://doi.org/10.1016/j.sbi.2013.05.006
Müller, G., Várnai, A., Johansen, K.S., Eijsink, V.G.H., and Horn, S.J., Biotechnol. Biofuels, 2015, vol. 8. Article no. 187. https://doi.org/10.1186/s13068-015-0376-y
Kim, I.J., Nam, K.H., Yun, E.J., Kim, S., Youn, H.J., Lee, H.J., Choi, I.-G., and Kim, K.H., Appl. Microbiol. Biotechnol., 2015, vol. 99, pp. 8537–8547. https://doi.org/10.1007/s00253-015-6592-3
Sun, F.F., Hong, J., Hu, J., Saddler, J.N., Fang, X., Zhang, Z., and Shen, S., Enzyme Microb. Technol., 2015, vol. 79-80, pp. 42–48. https://doi.org/10.1016/j.enzmictec.2015.06.020
Ghatge, S.S., Telke, A.A., Waghmode, T.R., Lee, Y., Lee, K.-W., Oh, D.-B., Shin, H.-D., and Kim, S.-W., Appl. Microbiol. Biotechnol., 2015, vol. 99, pp. 3041–3055. https://doi.org/10.1007/s00253-014-6116-6
Hu, J., Chandra, R., Arantes, V., Gourlay, K., van Dyk, J.S., and Saddler, J., Biores. Technol., 2015, vol. 186, pp. 149–153. https://doi.org/10.1016/j.biortech.2015.03.055
Bulakhov, A.G., Gusakov, A.V., Chekushina, A.V., Satrutdinov, A.D., Koshelev, A.V., Matys, V.Yu., and Sinitsyn, A.P., Biochemistry (Moscow), 2016, vol. 81, no. 5, pp. 530–537.
Kim, I.J., Seo, N., An, H.J., Kim, J.-H., Harris, P.V., and Kim, K.H., Biotechnol. Biofuels, 2017, vol. 10. Article no. 46. https://doi.org/10.1186/s13068-017-0721-4
Long, L., Yang, H., Ren, H., Liu, R., Sun, F.F., Xiao, Z., Hu, J., and Xu, Z., ACS Sustainable Chem. Eng., 2020, vol. 8, no. 32, pp. 11986–11993. https://doi.org/10.1021/acssuschemeng.0c02564
Guo, X., Sang, J., Chai, C., An, Y., Wei, Z., Zhang, H., Ma, L., Dai, Y., Lu, F., and Liu, F., Biochem. Eng. J., 2020, vol. 162. Article no. 107712. https://doi.org/10.1016/j.bej.2020.107712
Eibinger, M., Ganner, T., Bubner, P., Rosker, S., Kracher, D., Haltrich, D., Ludwig, R., Plank, H., and Nidetzky, B., J. Biol. Chem., 2014, vol. 289, no. 52, pp. 35929–35938. https://doi.org/10.1074/jbc.M114.602227
Karnaouri, A., Muraleedharan, M.N., Dimarogona, M., Topakas, E., Rova, U., Sandren, M., and Christakopoulos, P., Biotechnol. Biofuels, 2017, vol. 10. Article no. 126. https://doi.org/10.1186/s13068-017-0813-1
Zhou, H., Li, T., Yu, Z., Ju, J., Zhang, H., Tan, H., Li, K., and Yin, H., Int. J. Biol. Macromol., 2019, vol. 139, pp. 570–576. https://doi.org/10.1016/j.ijbiomac.2019.08.004
Garrido, M.M., Landoni, M., Sabbadin, F., Valacco, M.P., Couto, A., Bruce, N.C., Wirth, S.A., and Campos, E., Appl. Microbiol. Biotechnol., 2020, vol. 104, pp. 9631–9643. https://doi.org/10.1007/s00253-020-10911-6
Keller, M.B., Badino, S.F., Blossom, B.M., McBrayer, B., Borch, K., and Westh, P., ACS Sustainable Chem. Eng, 2020, vol. 8, no. 37, pp. 14117–14126. https://doi.org/10.1021/acssuschemeng.0c04779
Tokin, R., Ipsen, J.O., Westh, P., and Johansen, K.S., Biotechnol. Lett., 2020, vol. 42, pp. 1975–1984. https://doi.org/10.1007/s10529-020-02922-0
Pierce, B.C., Agger, J.W., Wichmann, J., and Meyer, A.S., Enzyme Microb. Technol., 2017, vol. 98, pp. 58–66. https://doi.org/10.1016/j.enzmictec.2016.12.007
Semenova, M.V., Gusakov, A.V., Telitsin, V.D., Rozhkova, A.M., Kondratyeva, E.G., and Sinitsyn, A.P., Biochim. Biophys. Acta—Proteins Proteomics, 2020, vol. 1868, no. 1. Article no. 140297. https://doi.org/10.1016/j.bbapap.2019.140297
Morozova, V.V., Gusakov, A.V., Andrianov, R.M., Pravilnikov, A.G., Osipov, D.O., and Sinitsyn, A.P., Biotechnol. J., 2010, vol. 5, no. 8, pp. 871–880. https://doi.org/10.1002/biot.201000050
Dotsenko, A.S., Rozhkova, A.M., and Gusakov, A.V., Moscow Univ. Chem. Bull., 2015, vol. 70, no. 6, pp. 283–286. https://doi.org/10.3103/S0027131415060024
Korotkova, O.G., Semenova, M.V., Morozova, V.V., Zorov, I.N., Sokolova, L.M., Bubnova, T.M., Okunev, O.N., and Sinitsyn, A.P., Biochemistry (Moscow), 2009, vol. 74, no. 5, pp. 569–577. https://doi.org/10.1134/S0006297909050137
Wood, T.M., Methods Enzymol., 1988, vol. 160, pp. 19–25. https://doi.org/10.1016/0076-6879(88)60103-0
Nelson, N., J. Biol. Chem., 1944, vol. 153, pp. 375–380.
Peterson, G.L., Anal. Biochem., 1979, vol. 100, no. 2, pp. 201–220. https://doi.org/10.1016/0003-2697(79)90222-7
Westereng, B., Agger, J.W., Horn, S.J., Vaaje-Kolstad, G., Aachmann, F.L., Stenstrom, Y.H., and Eijsink, V.G.H., J. Chromatogr., A, 2013, vol. 1271, no. 1, pp. 144–152. https://doi.org/10.1016/j.chroma.2012.11.048
Hildebrand, A., Addison, J.B., Kasugo, T., and Fan, Z., Biochem. Eng. J., 2016, vol. 109, pp. 236–242. https://doi.org/10.1016/j.bej.2016.01.024
Villares, A., Moreau, C., Bennati-Granier, C., Garajova, S., Foucat, L., Falourd, X., Saake, B., Berrin, J.-G., and Cathala, B., Sci. Rep., 2017, vol. 7. Article no. 40262. https://doi.org/10.1038/srep40262
Song, B., Li, B., Wang, X., Shen, W., Park, S., Collings, C., Feng, A., Smith, S.J., Walton, J.D., and Ding, S.-Y., Biotechnol. Biofuels, 2018, vol. 11. Article no. 41. https://doi.org/10.1186/s13068-018-1023-1
Forsberg, Z., Sørlie, M., Petrović, D., Courtade, G., Aachmann, F.L., Vaaje-Kolstad, G., Bissaro, B., Rohr, A.K., and Eijsink, V.G.H., Curr. Opin. Struct. Biol., 2019, vol. 59, pp. 54–64. https://doi.org/10.1016/j.sbi.2019.02.015
Funding
The study was carried out within the framework of the research work “Molecular design, structural-functional analysis, and regulation of enzyme systems, cell structures, bionanomaterials: fundamental bases and applications in technology, medicine, and environmental protection,” state registration number AAAA-A21-121011290089-4.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest. This article does not describe any research involving humans or animals as subjects.
Additional information
Translated by L. Solovyova
Rights and permissions
About this article
Cite this article
Semenova, M.V., Gusakov, A.V., Telitsin, V.D. et al. Enzymatic Destruction of Cellulose: Characteristics of the Kinetic Interaction of Lytic Polysaccharide Monooxygenases and Individual Cellulases. Appl Biochem Microbiol 57, 618–625 (2021). https://doi.org/10.1134/S0003683821050136
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0003683821050136