Abstract
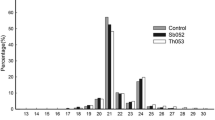
Peach is one of the most widely grown fruit crops in China, nectarine [Prunus persica (L.) Batsch var. nucipersica (Suckow) C.K. Schneid.] is an important commercial variety of peach. MicroRNAs (miRNAs) play important functions in post-transcriptional gene regulation in plants. In this study, miRNAs and their target genes were identified and predicted by sequencing four small RNA libraries prepared from nectarine leaf and fruit tissues infected with different virus/viroid combinations using next-generation sequencing technology (NGS). A total of 116 known peach miRNAs were identified, and 303 putative novel miRNAs were predicted. Some of these miRNAs were found to be highly conserved. Most of the novel miRNAs were 24-nt in length and many of these miRNAs came from MIR5271 and MIR7717 families. All differently expressed miRNAs had 2785 putative target genes. Comparative profiling revealed that 45 miRNAs exhibited significant differential expression between leaf and fruit tissue. Moreover, expression levels of these miRNAs were significantly affected in the two tissues despite the fact that the trees were infected by different virus/viroid combinations. We also found that there were more differentially expressed miRNAs in leaves tissue than in fruits, and a few miRNAs were identified that play important roles in the response to virus/viroid infection. Our results show that the expression of some miRNAs appears to be tissue-specific, and virus/viroid infection had a greater effect on the miRNA expression profiles in leaves than in fruits of nectarine. We hypothesize that virus/viroid infection induces the production of many novel miRNAs and also affects their lengths.










Similar content being viewed by others
Abbreviations
- ACLSV:
-
Apple chlorotic leaf spot virus
- ASGV:
-
Apple stem grooving virus
- ASPV:
-
Apple stem pitting virus
- ATP:
-
Adenosine triphosphate
- bp:
-
Base pair
- CGMMV:
-
Cucumber green mottle mosaic virus
- FC:
-
Fold change
- FDR:
-
False discovery rate
- GO:
-
Gene Ontology
- IPGI:
-
International Peach Genome Initiative
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- min:
-
Minute
- miRNAs:
-
MicroRNAs
- NGS:
-
Next-generation sequencing
- nt:
-
Nucleotide(s)
- PLMVd:
-
Peach latent mosaic viroid
- RISC:
-
RNA-induced silencing complexes
- RNA:
-
Ribonucleic Acid
- rpm:
-
Revolutions per minute
- rRNA:
-
Ribosomal RNA
- RSV:
-
Rice stripe virus
- RT-qPCR:
-
Real-time quantitative PCR
- sec:
-
Second
- snoRNA:
-
Small nucleolar RNA
- snRNA:
-
Small nuclear RNA
- sRNA:
-
Small RNA
- TEF-2:
-
Translation enlongation factor 2
- tRNA:
-
Transfer RNA
References
Allen E, Xie Z, Gustafson AM, Carrington JC (2005) microRNA-directed phasing during trans-acting siRNA biogenesis in plants. Cell 121:207–221. https://doi.org/10.1016/j.cell.2005.04.004
Apweiler R, Bairoch A, Wu CH, Barker WC, Boeckmann B et al (2004) UniProt: the Universal Protein Knowledgebase. Nucleic Acids Res 32:115–119. https://doi.org/10.1093/nar/gkh131
Barakat A, Sriram A, Park J, Zhebentyayeva T, Main D et al (2012) Genome wide identification of chilling responsive microRNAs in Prunus persica. BMC Genomics 13:481. https://doi.org/10.1186/1471-2164-13-481
Bartel DP (2009) MicroRNAs: target recognition and regulatory functions. Cell 136:215–233. https://doi.org/10.1016/j.cell.2009.01.002
Bazzini AA, Hopp HE, Beachy RN, Asurmendi S (2007) Infection and coaccumulation of tobacco mosaic virus proteins alter microRNA levels, correlating with symptom and plant development. Proc Natl Acad Sci U S A 104:12157–12162. https://doi.org/10.1073/pnas.0705114104
Byrne DH (2002) Peach breeding trends: a worldwide perspective. Acta Hortic 592:49–59. https://doi.org/10.17660/ActaHortic.2002.592.5
Deng YY, Li JQ, Wu SF, Zhu YP, Chen YW et al (2006) Integrated NR database in protein annotation system and its localization. Comput Eng 32:71–74
Dong XM, Zhang Y, Liu W (2016) Research progress of peach virus disease and its detection technology. Northern Horticulture 3:199–203
Du P, Wu JG, Zhang JY, Zhao SQ, Zheng H et al (2011) Viral infection induces expression of novel phased microRNAs from conserved cellular microRNA precursors. PLoS Pathog 7:e1002176. https://doi.org/10.1371/journal.ppat.1002176
Eddy SR (1998) Profile hidden Markov models. Bioinformatics 14:755–763. https://doi.org/10.1093/bioinformatics/14.9.755
Eldem V, Akçay UC, Ozhuner E, Bakır Y, Zheng USH et al (2012) Genome-wide identification of miRNAs responsive to drought in peach (Prunus persica) by high-throughput deep sequencing. PLoS One 7:e50298. https://doi.org/10.1371/journal.pone.0050298
Fahlgren N, Howell MD, Kasschau KD, Chapman EJ, Sullivan CM et al (2007) High-throughput sequencing of Arabidopsis microRNAs: evidence for frequent birth and death of miRNA genes. PLoS One 2:e219. https://doi.org/10.1371/journal.pone.0000219
Friedlander MR, Mackowiak SD, Li N, Chen W, Rajewsky N (2012) miRDeep2 accurately identifies known and hundreds of novel microRNA genes in seven animal clades. Nucleic Acids Res 40:37–52. https://doi.org/10.1093/nar/gkr688
Gao ZH, Shi T, Luo XY, Zhang Z, Zhuang WB et al (2012) High-throughput sequencing of small RNAs and analysis of differentially expressed microRNAs associated with pistil development in Japanese apricot. BMC Genom 13:371. https://doi.org/10.1186/1471-2164-13-371
Han R (2014) Identification of miRNA and tasiRNA in different organs/developmental stages of wheat and analysis of their temporal and spatial expression characteristics. Northwest A&F University, Xi An
Hernandez C, Flores R (1992) Plus and minus RNAs of peach latent mosaic viroid self-cleave in vitro via hammerhead structures. Proc Natl Acad Sci U S A 89:3711–3715. https://doi.org/10.1073/pnas.89.9.3711
Jo Y, Lian S, Chu H, Cho JK, Yoo SH et al (2018) Peach RNA viromes in six different peach cultivars. Sci Rep 8:52–43. https://doi.org/10.1038/s41598-018-20256-w
Kanehisa M, Goto S, Kawashima S, Yasushi O, Masahiro H (2004) The KEGG resource for deciphering the genome. Nucleic Acids Res 32:277–280. https://doi.org/10.1093/nar/gkh063
Koonin EV, Fedorova ND, Jackson JD, Jacobs AR, Krylov DM et al (2004) A comprehensive evolutionary classification of proteins encoded in complete eukaryotic genomes. Genome Biol 5:R7. https://doi.org/10.1186/gb-2004-5-2-r7
Langmead B, Trapnell C, Pop M, Salzberg S (2009) Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol 10:r25. https://doi.org/10.1186/gb-2009-10-3-r25
Li SX, Shao ZR, Fu XL, Xiao W, Li L et al (2017) Identification and characterization of Prunus persica miRNAs in response to UVB radiation in greenhouse through high-throughput sequencing. BMC Genom 18:938. https://doi.org/10.1186/s12864-017-4347-5
Liang CQ, Liu HW, Luo LX, Li JQ (2016) Analysis of miRNA expression characteristics related to cucumber green mottle mosaic virus infection in cucumber. Acta Phytotaxon Sin 46:56–62. https://doi.org/10.13926/j.cnki.apps.2016.01.007
Liu DM (2014) Study on the effect of CYP450 protein on corn infection by SCMV. China Agricultural University, Beijing
Liu YL (2015) Mining and analysis of miRNA and target genes involved in citrus fruit development. Huazhong Agricultural University, Wu Han
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCt method. Methods 25:402–408. https://doi.org/10.1006/meth.2001
Lu XY, Huang XL (2008) Plant miRNAs and abiotic stress responses. Biochem Bioph Res Co 368:458–462. https://doi.org/10.1016/j.bbrc.2008.02.007
Luo XY, Gao ZH, Shi T, Cheng ZM, Zhang Z et al (2013) Identification of miRNAs and their target genes in peach (Prunus persica L.) Using High-Throughput Sequencing and Degradome Analysis. PLoS One 8:e79090. https://doi.org/10.1371/journal.pone.0079090
Reinhart BJ, Weinstein EG, Rhoades MW, Bartel DP (2002) MicroRNAs in plants. Genes Dev 16:1616–1626. https://doi.org/10.1101/gad.1004402
Reyes JL, Chua N (2007) ABA induction of miR159 controls transcript levels of two MYB factors during Arabidopsis seed germination. Plant J 49:592–606. https://doi.org/10.1111/j.1365-313X.2006.02980.x
Roman LT, Michael YG, Darren AN, Eugene VK (2000) The COG database: a tool for genome scale analysis of protein functions and evolution. Nucleic Acids Res 28:33–36. https://doi.org/10.1093/nar/28.1.33
Romualdi C, Bortoluzzi S, D’Alessi F, Danieli GA (2003) IDEG6: a web tool for detection of differentially expressed genes in multiple tag sampling experiments. Physiol Genomics 12:159–162. https://doi.org/10.1152/physiolgenomics.00096.2002
Shang FD, Zhang X, Zhao YX, Liu JF (2018) Recombinant expression and purification of maize disease resistance protein PSiP NB-ARC domain. Proc 2018 Annual Conf Chin Soc Plant Pathol
Shi L, Guo YB, Shen Y (2019) miRNA regulates the growth, development and secondary metabolism of medicinal plants. Chin J Biochem Mol Bio 35:361–370
Shi MY, Hu X, Wei Y, Hou X, Yuan X et al (2017) Degradome Revealed Conserved Regulations of miRNAs on Auxin-Responsive Genes during Fruit Enlargement in Peaches. Int J Mol Sci 18:2599. https://doi.org/10.3390/ijms18122599
Shulaev V, Korban SS, Sosinski B, Abbott AG, Aldwinckle HS et al (2008) Multiple models for Rosaceae genomics. Plant Physiol 147:985–1003. https://doi.org/10.1104/PP.107.115618
Singh A, Roy S, Singh S, Das SS, Gautam V et al (2017) Phytohormonal crosstalk modulates the expression of miR166/165s, target Class III HD-ZIPs, and KANADI genes during root growth in Arabidopsis thaliana. Sci Rep 7:E311–E357. https://doi.org/10.1038/s41598-017-03632-w
Storey JD (2002) A direct approach to false discovery rates. J R Stat Soc B 64:479–498. https://doi.org/10.1111/1467-9868.00346
Tong ZG, Gao ZH, Wang F, Zhou J, Zhang Z (2009) Selection of reliable reference genes for gene expression studies in peach using real-time PCR. BMC Mol Biol 10:71. https://doi.org/10.1186/1471-2199-10-71
Wang CM, Li QZ (2007) Small molecule microRNA and its biological characteristics. J Northeast Agric Univ 4:548–553
Wu JG, Yang RX, Yang ZR, Yao SZ, Zhao SS et al (2017) ROS accumulation and antiviral defence control by microRNA528 in rice. Nat Plants 3:16203. https://doi.org/10.1038/nplants.2016.203
Wu LT, Ruan Y, Peng Q, Zhang Y, Liu CL (2006) Research progress of miRNA. Crop Res 572–576. https://doi.org/10.16848/j.cnki.issn.1001-5280.2006.05.046
Wu L, Qi YJ (2010) Research Progress of plant small RNA. Life Sci 22:682–687. https://doi.org/10.13376/j.cbls/2010.07.004
Xu Y,Li S,Na C,Yang L,Lu M (2019) Analyses of virus/viroid communities in nectarine trees by next-generation sequencing and insight into viral synergisms implication in host disease symptoms.Sci Rep 9: 12261. https://doi.org/10.1038/s41598-019-48714-z
Yang LJ, Li SF, Lu MG (2020) Research progress of miRNA in plant pathogen regulation. Biotechnol Bull 36:101–109. https://doi.org/10.13560/j.cnki.biotech.bull.1985.2019-1045
Yang SS, Chen GL, Yao JC (2019) Research progress of miRNA in plant stress resistance. Seed Technol 37:154
Yin L, Fang H, Huang Y, Lu J, Qu JJ (2017) Research progress on the domains of plant TIR-NB-LRR type disease resistance genes. Guangxi Plants 37:186–190
Zhang CH, Zhang BB, Ma RJ, Yu ML, Guo SL et al (2016a) Identification of known and novel micrornas and their targets in peach (prunus persica) fruit by high-throughput sequencing. PLoS One 11:e0159253. https://doi.org/10.1371/journal.pone.0159253
Zhang C, Ding ZM, Wu KC, Yang L, Li Y et al (2016b) Suppression of Jasmonic Acid-Mediated Defense by Viral-Inducible microRNA319 Facilitates Virus Infection in Rice. Mol Plant 9:1302–1314. https://doi.org/10.1016/j.molp.2016.06.014
Zhang F (2017) Construction of Infectious Clones of Chinese Wheat Mosaic Virus and Its Interaction with Host HSP70. Zhejiang Normal University, Jin Hua
Zhang XY (2006) miRNA: A new gene regulatory element. Journal of Leshan Normal University 21:51–53
Zhu H, Xia R, Zhao BY, An YQ, Dardick CD et al (2012) Unique expression, processing regulation, and regulatory network of peach (Prunus persica) miRNAs. BMC Plant Biol 12:149. https://doi.org/10.1186/1471-2229-12-149
Acknowledgements
This work was supported by the earmarked fund of the China Agriculture Research System (grant number CARS-30-3-01), the Key Inter-Governmental Projects of the National Key Research and Development Program [grant number 2017YFE0110900].
Author information
Authors and Affiliations
Contributions
Lu Meiguang contributed to the study conception and design. Data collection and analysis were performed by Yang Lijuan and Lu Meiguang. The first draft of the manuscript was written and revised by Yang Lijuan and Lu Meiguang. Material preparation and Investigation by Li Shifang and Zhang Zimeng. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Additional information
Communicated by: Yuan Qin
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, L., Li, S., Zhang, Z. et al. Genome-Wide Identification of MicroRNAs that are Responsive to Virus/Viroid Infection in Nectarine Trees Through High-Throughput Sequencing. Tropical Plant Biol. 15, 78–92 (2022). https://doi.org/10.1007/s12042-021-09300-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12042-021-09300-8