Abstract
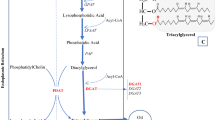
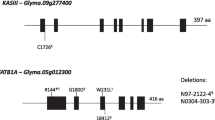
Soybean oil is the second most-produced vegetable oil worldwide. To enhance the nutritional quality and oxidative stability of soybean oil, many soybean breeding programs are trying to increase oleic acid content and reduce linoleic and linolenic acid contents. The fatty acid profile of soybean oil is controlled by many genes, including those which code for omega-3 and omega-6 desaturases. Mutations in GmFAD2-1 and GmFAD3 genes are widely studied and their combinations can produce soybean oil with high oleic and low linoleic and linolenic content. However, few studies evaluate the effect of these mutations on gene expression. Therefore, the present study sought to identify reference genes, evaluate the expression of GmFAD2-1 and GmFAD3 seed desaturase genes in thirteen wild-type and mutated soybean accessions, and associate the expression patterns with fatty acid composition and with the GmFAD2-1 and GmFAD3 genotypes. GmCONS7 and GmUKN2 were identified as the best reference genes for combined use to normalize data. The GmFAD2-1A mutation of PI603452 accession was associated with a decrease in gene expression of GmFAD2-1A; however, downregulation may not be due to the truncated enzyme structure alone. These results suggested that there are factors other than GmFAD2-1A and GmFAD2-1B that have a considerable effect on oleic content, at least in soybeans with mutations in these two genes.




Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
References
Allen JF (1992) Protein phosphorylation in regulation of photosynthesis. Biochim Biophys Acta 1098(3):275–335. https://doi.org/10.1016/S0005-2728(09)91014-3
Alt JL, Fehr WR, Welke GA, Shannon JG (2005) Transgressive segregation for oleate content in three soybean populations. Crop Sci 45:2005–2007. https://doi.org/10.2135/cropsci2004.0693
ANP – Agência Nacional de Petróleo (2019) Matérias-primas utilizadas na produção de biodiesel (B100) no Brasil— 2009–2018. Tabela 4.13. Available from: http://www.anp.gov.br/publicacoes/anuario-estatistico/5237-anuario-estatistico-2019
Bachlava E, Dewey RE, Burton JW, Cardinal AJ (2009) Mapping and comparison of quantitative trait loci for oleic acid seed content in two segregating soybean populations. Crop Sci 49(2):433–442. https://doi.org/10.2135/cropsci2008.06.0324
Bilyeu K, Palavalli L, Sleper D, Beuselinck P (2005) Mutations in soybean microsomal omega-3 fatty acid desaturase genes reduce linolenic acid concentration in soybean seeds. Crop Sci 45(5):1830–1836. https://doi.org/10.2135/cropsci2004.0632
Borges A, Tsai SM, Caldas DGG (2012) Validation of reference genes for RT-qPCR normalization in common bean during biotic and abiotic stresses. Plant Cell Rep 31(5):827–838. https://doi.org/10.1007/s00299-011-1204-x
Burkey KO, Booker FL, Pursley WA, Heagle AS (2007) Elevated carbon dioxide and ozone effects on peanut: II. Seed yield and quality. Crop Sci 47(4):1488–1497. https://doi.org/10.2135/cropsci2006.08.0538
Burton JW, Wilson RF, Brim CA (1983) Recurrent selection in soybeans. IV. Selection for increased oleic acid percentage in seed oil 1. Crop Sci 23(4):744–747. https://doi.org/10.2135/cropsci1983.0011183X002300040034x
Combs R, Bilyeu K (2019) Novel alleles of FAD2-1A induce high levels of oleic acid in soybean oil. Mol Breed 39(6):79. https://doi.org/10.1007/s11032-019-0972-9
CONAB (2021) Acompanhamento Da Safra Brasileira de Grãos. Safra. 2019/2020 7. Available from: https://www.conab.gov.br/info-agro/safras/graos/boletim-da-safra-de-graos.
Fehr WR (2007) Breeding for modified fatty acid composition in soybean. Crop Sci 47:72–87
Gao M, Liu Y, Ma X, Shuai Q et al (2017) Evaluation of reference genes for normalization of gene expression using quantitative RT-PCR under aluminum, cadmium, and heat stresses in soybean. PLoS ONE 12(1):e0168965. https://doi.org/10.1371/journal.pone.0168965
Gesteira ADS, Schuster I, José IC, Piovesan ND et al (2003) Biometrical analyses of linolenic acid content of soybean seeds. Genet Mol Biol 26(1):65–68. https://doi.org/10.1590/S1415-47572003000100011
Goettel W, Xia E, Upchurch R, Wang ML et al (2014) Identification and characterization of transcript polymorphisms in soybean lines varying in oil composition and content. BMC Genomics 15:299. https://doi.org/10.1186/1471-2164-15-299
Goodstein DM, Shu S, Howson R, Neupane R et al (2012) Phytozome: a comparative platform for green plant genomics. Nucleic Acids Res 40(D1):D1178–D1186. https://doi.org/10.1093/nar/gkr944
Hellemans J, Mortier G, De Paepe A, Speleman F, Vandesompele J (2007) qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data. Genome Biol 8(2):R19. https://doi.org/10.1186/gb-2007-8-2-r19
Heppard EP, Kinney AJ, Stecca KL, Miao GH (1996) Developmental and growth temperature regulation of two different microsomal [omega]-6 desaturase genes in soybeans. Plant Physiol 110(1):311–319. https://doi.org/10.1104/pp.110.1.311
Hirschburger D, Müller M, Voegele RT, Link T (2015) Reference genes in the pathosystem Phakopsora pachyrhizi/soybean suitable for normalization in transcript profiling. Int J Mol Sci 16(9):23057–23075. https://doi.org/10.3390/ijms160923057
Hu FB, Stampfer MJ, Manson JE, Rimm E et al (1997) Dietary fat intake and the risk of coronary heart disease in women. N Engl J Med 337(21):1491–1499. https://doi.org/10.1056/NEJM199711203372102
Hu R, Fan C, Li H, Zhang Q, Fu YF (2009) Evaluation of putative reference genes for gene expression normalization in soybean by quantitative real-time RT-PCR. BMC Mol Biol 10(1):93. https://doi.org/10.1186/1471-2199-10-93
Irsigler AS, Costa MD, Zhang P, Reis PA et al (2007) Expression profiling on soybean leaves reveals integration of ER-and osmotic-stress pathways. BMC Genomics 8(1):431. https://doi.org/10.1186/1471-2164-8-431
Juhász ACP, Ciabotti S, Teixeira LCAA (2017) Breeding for nutritional quality. In: Silva F, Borém A, Sediyama T, Ludke WH (Org) Soybean breeding, 1st edn., vol 1. Springer International Publishing, Cham, pp 375–393. https://doi.org/10.1007/978-3-319-57433-2_20
Le DT, Aldrich DL, Valliyodan B, Watanabe Y et al (2012) Evaluation of candidate reference genes for normalization of quantitative RT-PCR in soybean tissues under various abiotic stress conditions. PLoS ONE 7(9):e46487. https://doi.org/10.1371/journal.pone.0046487
Lee S, Kim JH, Sundaramoorthy J, Park GT et al (2018) Identification of GmSALT3 haplotypes and development of molecular markers based on their diversity associated with salt tolerance in soybean. Mol Breed 38(7):86. https://doi.org/10.1007/s11032-018-0845-7
Leefel RC (1992) Registration of high-protein soybean germplasm lines BARC-6, BARC-7, BARC-8, and BARC-9. Crop Sci 32(2):502–502. https://doi.org/10.2135/cropsci1992.0011183X003200020054x
Li Q, Fan CM, Zhang XM, Fu YF (2012) Validation of reference genes for real-time quantitative PCR normalization in soybean developmental and germinating seeds. Plant Cell Rep 31(10):1789–1798. https://doi.org/10.1007/s00299-012-1326-9
Libault M, Thibivilliers S, Bilgin DD, Radwan O et al (2008) Identification of four soybean reference genes for gene expression normalization. Plant Genome 1(1):44–54. https://doi.org/10.3835/plantgenome2008.02.0091
Liebers M, Chevalier F, Blanvillain R, Pfannschmidt T (2018) PAP genes are tissue-and cell-specific markers of chloroplast development. Planta 248(3):629–646. https://doi.org/10.1007/s00425-018-2924-8
Ma S, Niu H, Liu C, Zhang J et al (2013) Expression stabilities of candidate reference genes for RT-qPCR under different stress conditions in soybean. PLoS ONE 8(10):e75271. https://doi.org/10.1371/journal.pone.0075271
Maheshwari P, Kovalchuk I (2016) Genetic transformation of crops for oil production. Ind Oil Crops. https://doi.org/10.1016/B978-1-893997-98-1.00014-2
Miranda VJ, Coelho RR, Viana AAB, Oliveira Neto OB et al (2013) Validation of reference genes aiming accurate normalization of qPCR data in soybean upon nematode parasitism and insect attack. BMC Res Notes 6(1):196. https://doi.org/10.1186/1756-0500-6-196
Neumaier N, Nepomuceno AL, Farias JRB, Oya T (2000) Estadios de desenvolvimento da cultura de soja. In: Bonato ER (ed) Estresses em soja. Passo Fundo: Embrapa Trigo, 254 p. Available from: https://www.embrapa.br/busca-de-publicacoes/-/publicacao/820371/estresses-em-soja
Patterson E, Wall R, Fitzgerald GF, Ross RP et al (2012) Health implications of high dietary omega-6 polyunsaturated fatty acids. J Nutr Metab. https://doi.org/10.1155/2012/539426
Pham AT, Lee JD, Shannon JG, Bilyeu KD (2010) Mutant alleles of FAD2-1A and FAD2-1B combine to produce soybeans with the high oleic acid seed oil trait. BMC Plant Biol 10:195. https://doi.org/10.1186/1471-2229-10-195
Pham AT, Lee JD, Shannon JG, Bilyeu KD (2011) A novel FAD2-1 A allele in a soybean plant introduction offers an alternate means to produce soybean seed oil with 85% oleic acid content. Theor Appl Genet 123:793–802. https://doi.org/10.1007/s00122-011-1627-3
Pham AT, Shannon JG, Bilyeu KD (2012) Combinations of mutant FAD2 and FAD3 genes to produce high oleic acid and low linolenic acid soybean oil. Theor Appl Genet 125:503–515. https://doi.org/10.1007/s00122-012-1849-z
Pham AT, Bilyeu K, Chen P, Boerma HR et al (2014) Characterization of the fan1 locus in soybean line A5 and development of molecular assays for high-throughput genotyping of FAD3 genes. Mol Breed 33:895–907. https://doi.org/10.1007/s11032-013-0003-1
Pinto MDO, Good-God PIV, Moreira MA, Barros EGD (2013) Associação de marcadores moleculares SNP com o conteúdo de ácido linolênico em sementes de soja. Pesqui Agropecu Bras 48(3):263–269. https://doi.org/10.1590/S0100-204X2013000300004
Reinprecht Y, Luk-Labey SY, Larsen J, Poysa VW et al (2009) Molecular basis of the low linolenic acid trait in soybean EMS mutant line RG10. Plant Breed 128:253–258. https://doi.org/10.1111/j.1439-0523.2008.01571.x
Rodrigues JIDS, Arruda KMA, Cruz CD, Piovesan ND et al (2014) Biometric analysis of protein and oil contents of soybean genotypes in different environments. Pesqui Agropecu Bras 49(6):475–482. https://doi.org/10.1590/S0100-204X2014000600009
Rodrigues JIS, Arruda KMA, Cruz CD, Barros EG, Piovesan ND, Moreira MA (2017) Genetic divergence of soybean genotypes in relation to grain components. Cienc Rural 47(2):e20151258. https://doi.org/10.1590/0103-8478cr20151258
Sandhu D, Alt JL, Scherder CW, Fehr WR, Bhattacharyya MK (2007) Enhanced oleic acid content in the soybean mutant M23 is associated with the deletion in the Fad2-1a gene encoding a fatty acid desaturase. J Am Oil Chem Soc 84(3):229–235. https://doi.org/10.1007/s11746-007-1037-5
Silva LCC, Bueno RD, da Matta LB, Pereira PHS et al (2018) Characterization of a new GmFAD3A allele in Brazilian CS303TNKCA soybean cultivar. Theor Appl Genet 131:1099–1110. https://doi.org/10.1007/s00122-018-3061-2
Silva LCC, Mota LM, Fonseca LABV, Bueno RD et al (2019) Effect of a mutation in Raffinose Synthase 2 (GmRS2) on soybean quality traits. Crop Breed Appl Biotechnol 19(1):62–69. https://doi.org/10.1590/1984-70332019v19n1a09
Swift ML (1997) GraphPad prism, data analysis, and scientific graphing. J Chem Inf Model 37(2):411–412. https://doi.org/10.1021/ci960402j
Tang GQ, Novitzky WP, Griffin HC, Huber SC, Dewey RE (2005) Oleate desaturase enzymes of soybean: evidence of regulation through differential stability and phosphorylation. Plant J 44(3):433–446. https://doi.org/10.1111/j.1365-313X.2005.02535.x
Thapa R, Carrero-Colon M, Crowe M, Gaskin E, Hudson K (2016) Novel FAD2–1A alleles confer an elevated oleic acid phenotype in soybean seeds. Crop Sci 56(1):226–231. https://doi.org/10.2135/cropsci2015.06.0339
Thapa R, Carrero-Colón M, Addo-Quaye C, Held J et al (2018) New alleles of FAD3A lower the linolenic acid content of soybean seeds. Crop Sci 58(2):713–718. https://doi.org/10.2135/cropsci2017.08.0490
Thibivilliers S, Joshi T, Campbell KB, Scheffler B et al (2009) Generation of Phaseolus vulgaris ESTs and investigation of their regulation upon Uromyces appendiculatus infection. BMC Plant Biol 9(1):46. https://doi.org/10.1186/1471-2229-9-46
Trevaskis B, Wandrey M, Colebatch G, Udvardi MK (2002) The soybean GmN6L gene encodes a late nodulin expressed in the infected zone of nitrogen-fixing nodules. Mol Plant Microbe Interact 15(7):630–636. https://doi.org/10.1094/MPMI.2002.15.7.630
Upchurch RG, Ramirez ME (2011) Soybean plastidal omega-3 fatty acid desaturase genes GmFAD7 and GmFAD8: structure and expression. Crop Sci 51(4):1673–1682. https://doi.org/10.2135/cropsci2010.09.0537
USDA (2021) Oilseeds: world markets and trade. September 2021. Available from: https://apps.fas.usda.gov/psdonline/circulars/oilseeds.pdf
Vandesompele J, De Preter K, Pattyn F, Poppe B et al (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. https://doi.org/10.1186/gb-2002-3-7-research0034
Wan Q, Chen S, Shan Z, Yang Z et al (2017) Stability evaluation of reference genes for gene expression analysis by RT-qPCR in soybean under different conditions. PLoS ONE. https://doi.org/10.1371/journal.pone.0189405
Warner K, Fehr W (2008) Mid-oleic/ultra low linolenic acid soybean oil: a healthful new alternative to hydrogenated oil for frying. J Am Oil Chem Soc 85:945–951. https://doi.org/10.1007/s11746-008-1275-1
Wen S, Liu H, Li X, Chen X et al (2018) TALEN-mediated targeted mutagenesis of fatty acid desaturase 2 (FAD2) in peanut (Arachis hypogaea L.) promotes the accumulation of oleic acid. Plant Mol Biol 97(1):177–185. https://doi.org/10.1007/s11103-018-0731-z
Wieczorek P, Wrzesińska B, Obrępalska-Stęplowska A (2013) Assessment of reference gene stability influenced by extremely divergent disease symptoms in Solanum lycopersicum L. J Virol Methods 194(1):161–168. https://doi.org/10.1016/j.jviromet.2013.08.010
Woyann LG, Meira D, Zdziarski AD, Matei G et al (2019) Multiple-trait selection of soybean for biodiesel production in Brazil. Ind Crop Prod 140:111721. https://doi.org/10.1016/j.indcrop.2019.111721
Yan Q, Cui X, Lin S, Gan S et al (2016) GmCYP82A3, a soybean cytochrome P450 family gene involved in the jasmonic acid and ethylene signaling pathway, enhances plant resistance to biotic and abiotic stresses. PLoS ONE 11(9):e0162253. https://doi.org/10.1371/journal.pone.0162253
Yuan FJ, Zhu DH, Tan YY, Dong DK et al (2012) Identification and characterization of the soybean IPK1 ortholog of a low phytic acid mutant reveals an exon-excluding splice-site mutation. Theor Appl Genet 125(7):1413–1423. https://doi.org/10.1007/s00122-012-1922-7
Zhang J, Wang X, Lu Y, Bhusal SJ (2018) Genome-wide scan for seed composition provides insights into soybean quality improvement and the impacts of domestication and breeding. Mol Plant 11:460–472. https://doi.org/10.1016/j.molp.2017.12.016
Funding
Conselho Nacional de Desenvolvimento Científico e Tecnológico (granting doctoral scholarships) and Fundação de Amparo à Pesquisa do Estado de Minas Gerais (Grant APQ-01416-16 and PPM-640-18 to MD).
Author information
Authors and Affiliations
Contributions
LCCS—experiment planning, development of F2 population, marker-assisted selection, interpretation of results, and manuscript writing. DBM—experiment planning, RNA extraction, and reverse transcriptase, RT-qPCR. RDB—searching of candidate genes, primers design, phenotyping. NDP—development of F2 population, advising. CR—interpretation of results and manuscript reviewing. MD-B—experiment planning, interpretation of results, manuscript reviewing, advising.
Corresponding author
Ethics declarations
Conflict of interest
Not applicable.
Ethical Approval
Not applicable.
Informed Consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10528_2021_10135_MOESM1_ESM.xlsx
Supplementary file1 (XLSX 11 kb) Supplementary Table 1 Information about soybean candidates for reference genes in desaturase expression analysis, and desaturase soybean seed genes evaluated for expression gene.
Rights and permissions
About this article
Cite this article
Silva, L.C.C., Mayrink, D.B., Bueno, R.D. et al. Reference Genes and Expression Analysis of Seed Desaturases Genes in Soybean Mutant Accessions. Biochem Genet 60, 937–952 (2022). https://doi.org/10.1007/s10528-021-10135-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-021-10135-x