Developing Actively Targeted Nanoparticles to Fight Cancer: Focus on Italian Research
Abstract
:1. Introduction
2. Ligands for Active Targeting and Receptor-Based Active Targeting Strategies
2.1. Peptide-Protein Ligands
2.2. Non-Peptide Ligands
2.3. Human Epidermal Growth Factor Receptor (HER)
2.4. CD44 Receptor (CD44)
2.5. Folate Receptor (FL)
2.6. Prostate-Specific Membrane Antigen (PSMA)
2.7. Transferrin Receptors (TF)
2.8. Biotin-Specific Receptors
2.9. Interleukin Receptor
2.10. Insulin (INS) and Nnsulin-like Growth Factor (IGF) Receptors
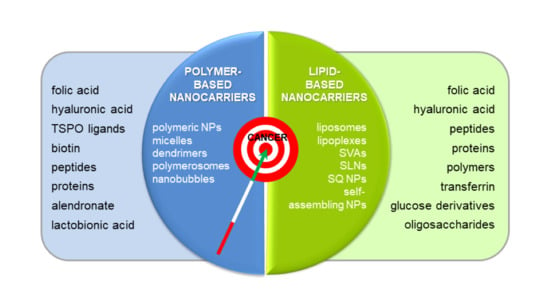
3. Ten Years of Italian Research on Actively Targeted Nanocarriers
3.1. Polymer-Based Nanocarriers
Polymer-Shelled Nanobubbles (NBs)
3.2. Lipid-Based Nanocarriers
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ApoE | apolipoprotein E |
| BBB | blood brain barrier |
| BN | bombesin |
| BTZ | bortezomib |
| CPE | cell-penetration enhancer |
| CUR | curcumin |
| CD | cyclodextrin |
| DC | dendritic cell |
| DDS | drug delivery system |
| DEX | dextran |
| DOPE | 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine |
| DOX | doxorubicin |
| DPPE | 1,2-dipalmitoyl-sn-glycero-3-phosphoethanolamine |
| DSPE | 1,2-distearoyl-sn-glycero-3-phosphoethanolamine |
| DTX | docetaxel |
| EDAC | 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide |
| EGFR | epidermal growth factor receptor |
| EPR | enhanced permeability and retention |
| FA | folic acid |
| FR | folate receptor |
| 5-FU | 5-fluorouracil |
| FZD10 | frizzled 10 protein |
| GEM | gemcitabine |
| HA | hyaluronic acid |
| HEGFR | human epidermal growth factor receptor |
| HPβ-CD | 2-(hydroxypropyl)-β-cyclodextrin |
| INS | insulin |
| Lf | lactoferrin |
| mAb(s) | monoclonal antibody(ies) |
| MDR | multidrug resistance |
| MTX | methotrexate |
| NB(s) | nanobubble(s) |
| NP(s) | nanoparticle(s) |
| PCL | poly(ε-caprolactone) |
| PDAC | pancreatic adenocarcinoma |
| PEG | polyethylene glycol |
| PEI | polyethyleneimine |
| PLGA | poly lactic-co-glycolic acid |
| PLA | poly lactic acid |
| PSMA | prostate-specific membrane antigen |
| PTX | paclitaxel |
| SLN(s) | solid lipid nanoparticle(s) |
| SQ | squalene |
| SVA(s) | supramolecular vesicular aggregate(s) |
| T3 | thyroid hormone triiodothyronine |
| Tf | transferrin |
| TfR | Tf receptor |
| TSH | thyroid-stimulating hormone |
| US | ultrasounds |
| UT | urotensin-II |
| VLDL | very low-density lipoprotein |
References
- Kumari, P.; Ghosh, B.; Biswas, S. Nanocarriers for cancer-targeted drug delivery. J. Drug Target. 2015, 24, 179–191. [Google Scholar] [CrossRef]
- Senapati, S.; Mahanta, A.K.; Kumar, S.; Maiti, P. Controlled drug delivery vehicles for cancer treatment and their performance. Signal. Transduct. Target. Ther. 2018, 3, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Gu, W.; Meng, F.; Haag, R.; Zhong, Z. Actively targeted nanomedicines for precision cancer therapy: Concept, construction, challenges and clinical translation. J. Control. Release 2021, 329, 676–695. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Li, J.; Zhao, Q.; Pan, T.; Zhong, H.; Wang, W. Advanced and Innovative Nano-Systems for Anticancer Targeted Drug Delivery. Pharmaceutics 2021, 13, 1151. [Google Scholar] [CrossRef]
- Dadwal, A.; Baldi, A.; Narang, R.K. Nanoparticles as carriers for drug delivery in cancer. Artif. Cells Nanomed. Biotechnol. 2018, 46, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Pearce, A.K.; O’Reilly, R.K. Insights into active targeting of nanoparticles in drug delivery: Advances in clinical studies and design considerations for cancer nanomedicine. Bioconjug. Chem. 2019, 30, 2300–2311. [Google Scholar] [CrossRef]
- Hejmady, S.; Pradhan, R.; Alexander, A.; Agrawal, M.; Singhvi, G.; Gorain, B.; Tiwari, S.; Kesharwani, P.; Dubey, S.K. Recent advances in targeted nanomedicine as promising antitumor therapeutics. Drug Discov. Today 2020, 25, 2227–2244. [Google Scholar] [CrossRef] [PubMed]
- Zielińska, A.; Szalata, M.; Gorczyński, A.; Karczewski, J.; Eder, P.; Severino, P.; Cabeda, J.; Souto, E.; Słomski, R. Cancer nanopharmaceuticals: Physicochemical characterization and in vitro/in vivo applications. Cancers 2021, 13, 1896. [Google Scholar] [CrossRef]
- Molina-Crespo, A.; Cadete, A.; Sarrio, D.; Gamez-Chiachio, M.; Martinez, L.; Chao, K.; Olivera, A.; Gonella, A.; Diaz, E.; Palacios, J.; et al. Intracellular delivery of an antibody targeting gasdermin-B Reduces HER2 breast cancer aggressiveness. Clin. Cancer Res. 2019, 25, 4846–4858. [Google Scholar] [CrossRef] [Green Version]
- Ragelle, H.; Danhier, F.; Préat, V.; Langer, R.; Anderson, D.G. Nanoparticle-based drug delivery systems: A commercial and regulatory outlook as the field matures. Expert Opin. Drug Deliv. 2016, 14, 851–864. [Google Scholar] [CrossRef]
- Attia, M.F.; Anton, N.; Wallyn, J.; Omran, Z.; Vandamme, T.F. An overview of active and passive targeting strategies to improve the nanocarriers efficiency to tumour sites. J. Pharm. Pharmacol. 2019, 71, 1185–1198. [Google Scholar] [CrossRef] [Green Version]
- Rosenblum, D.; Joshi, N.; Tao, W.; Karp, J.M.; Peer, D. Progress and challenges towards targeted delivery of cancer therapeutics. Nat. Commun. 2018, 9, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Nichols, J.W.; Bae, Y.H. EPR: Evidence and fallacy. J. Control. Release 2014, 190, 451–464. [Google Scholar] [CrossRef] [PubMed]
- Raj, S.; Khurana, S.; Choudhari, R.; Kesari, K.K.; Kamal, M.A.; Garg, N.; Ruokolainen, J.; Das, B.C.; Kumar, D. Specific targeting cancer cells with nanoparticles and drug delivery in cancer therapy. Semin. Cancer Biol. 2019, 69, 166–177. [Google Scholar] [CrossRef] [PubMed]
- Heath, V.L.; Bicknell, R. Anticancer strategies involving the vasculature. Nat. Rev. Clin. Oncol. 2009, 6, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Byrne, J.D.; Betancourt, T.; Brannon-Peppas, L. Active targeting schemes for nanoparticle systems in cancer therapeutics. Adv. Drug Deliv. Rev. 2008, 60, 1615–1626. [Google Scholar] [CrossRef]
- Bazak, R.; Houri, M.; El Achy, S.; Kamel, S.; Refaat, T. Cancer active targeting by nanoparticles: A comprehensive review of literature. J. Cancer Res. Clin. Oncol. 2014, 141, 769–784. [Google Scholar] [CrossRef] [Green Version]
- Muhamad, N.; Plengsuriyakarn, T.; Na-Bangchang, K. Application of active targeting nanoparticle delivery system for chemotherapeutic drugs and traditional/herbal medicines in cancer therapy: A systematic review. Int. J. Nanomed. 2018, 13, 3921–3935. [Google Scholar] [CrossRef] [Green Version]
- Autio, K.A.; Dreicer, R.; Anderson, J.; Garcia, J.A.; Alva, A.; Hart, L.L.; Milowsky, M.I.; Posadas, E.M.; Ryan, C.J.; Graf, R.P.; et al. Safety and efficacy of bind-014, a docetaxel nanoparticle targeting prostate-specific membrane antigen for patients with metastatic castration-resistant prostate cancer. JAMA Oncol. 2018, 4, 1344–1351. [Google Scholar] [CrossRef] [Green Version]
- Bazak, R.; Houri, M.; El Achy, S.; Hussein, W.; Refaat, T. Passive targeting of nanoparticles to cancer: A comprehensive review of the literature. Mol. Clin. Oncol. 2014, 2, 904–908. [Google Scholar] [CrossRef] [Green Version]
- Akhter, H.; Beg, S.; Tarique, M.; Malik, A.; Afaq, S.; Choudhry, H.; Hosawi, S. Receptor-based targeting of engineered nanocarrier against solid tumors: Recent progress and challenges ahead. Biochim. et Biophys. Acta BBA-Gen. Subj. 2020, 1865, 129777. [Google Scholar] [CrossRef]
- Akhtar, M.; Ahamed, M.; Alhadlaq, H.; Alrokayan, S.; Kumar, S. Targeted anticancer therapy: Overexpressed receptors and nanotechnology. Clin. Chim. Acta 2014, 436, 78–92. [Google Scholar] [CrossRef]
- Zhu, Y.; Choi, S.H.; Shah, K. Multifunctional receptor-targeting antibodies for cancer therapy. Lancet Oncol. 2015, 16, 543–554. [Google Scholar] [CrossRef]
- Ahmad, E.; Ali, A.; Fatima, M.T.; Nimisha; Apurva; Kumar, A.; Sumi, M.P.; Sattar, R.S.A.; Mahajan, B.; Saluja, S.S. Ligand decorated biodegradable nanomedicine in the treatment of cancer. Pharmacol. Res. 2021, 167, 105544. [Google Scholar] [CrossRef]
- Sonju, J.J.; Dahal, A.; Singh, S.S.; Jois, S.D. Peptide-functionalized liposomes as therapeutic and diagnostic tools for cancer treatment. J. Control. Release 2020, 329, 624–644. [Google Scholar] [CrossRef]
- Kobayashi, T.; Ishida, T.; Okada, Y.; Ise, S.; Harashima, H.; Kiwada, H. Effect of transferrin receptor-targeted liposomal doxorubicin in P-glycoprotein-mediated drug resistant tumor cells. Int. J. Pharm. 2007, 329, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Lei, Z.; Mengying, Z.; Yifei, G.; Xiangtao, W.; Meihua, H. Alendronate-modified polydopamine-coated paclitaxel nanoparticles for osteosarcoma-targeted therapy. J. Drug Deliv. Sci. Technol. 2019, 53, 101133. [Google Scholar] [CrossRef]
- Gao, X.; Li, L.; Cai, X.; Huang, Q.; Xiao, J.; Cheng, Y. Targeting nanoparticles for diagnosis and therapy of bone tumors: Opportunities and challenges. Biomaterials 2020, 265, 120404. [Google Scholar] [CrossRef]
- Korc, M.; Chandrasekar, B.; Yamanaka, Y.; Friess, H.; Buchier, M.; Beger, H.G. Overexpression of the epidermal growth factor receptor in human pancreatic cancer is associated with concomitant increases in the levels of epidermal growth factor and transforming growth factor alpha. J. Clin. Investig. 1992, 90, 1352–1360. [Google Scholar] [CrossRef] [Green Version]
- Zimmermann, M.; Zouhair, A.; Azria, D.; Ozsahin, M. The epidermal growth factor receptor (EGFR) in head and neck cancer: Its role and treatment implications. Radiat. Oncol. 2006, 1, 11. [Google Scholar] [CrossRef]
- Minner, S.; Rump, D.; Tennstedt, P.; Simon, R.; Burandt, E.; Terracciano, L.; Moch, H.; Wilczak, W.; Bokemeyer, C.; Fisch, M.; et al. Epidermal growth factor receptor protein expression and genomic alterations in renal cell carcinoma. Cancer 2011, 118, 1268–1275. [Google Scholar] [CrossRef] [Green Version]
- Herbst, R.S.; Shin, D.M. Monoclonal antibodies to target epidermal growth factor receptor-positive tumors. Cancer 2002, 94, 1593–1611. [Google Scholar] [CrossRef] [PubMed]
- Senbanjo, L.T.; Chellaiah, M.A. CD44: A Multifunctional Cell Surface Adhesion Receptor Is a Regulator of Progression and Metastasis of Cancer Cells. Front. Cell Dev. Biol. 2017, 5, 18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, C.; Sheng, Y.; Shi, X.; Liu, Y.; He, Y.; Du, Y.; Zhang, G.; Gao, F. CD44/HA signaling mediates acquired resistance to a PI3Kα inhibitor. Cell Death Dis. 2020, 11, 1–11. [Google Scholar] [CrossRef]
- Ledermann, J.A.; Canevari, S.; Thigpen, T. Targeting the folate receptor: Diagnostic and therapeutic approaches to personalize cancer treatments. Ann. Oncol. 2015, 26, 2034–2043. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Diop-Bove, N.; Visentin, M.; Goldman, I.D. Mechanisms of membrane transport of folates into cells and across epithelia. Annu. Rev. Nutr. 2011, 31, 177–201. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, A.; Heston, W.D. Tumor target prostate specific membrane antigen (PSMA) and its regulation in prostate cancer. J. Cell. Biochem. 2004, 91, 528–539. [Google Scholar] [CrossRef] [PubMed]
- Lütje, S.; Slavik, R.; Fendler, W.; Herrmann, K.; Eiber, M. PSMA ligands in prostate cancer—Probe optimization and theranostic applications. Methods 2017, 130, 42–50. [Google Scholar] [CrossRef]
- Saniee, F.; Ravari, N.S.; Goodarzi, N.; Amini, M.; Atyabi, F.; Moghadam, E.S.; Dinarvand, R. Glutamate-urea-based PSMA-targeted PLGA nanoparticles for prostate cancer delivery of docetaxel. Pharm. Dev. Technol. 2021, 26, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Ponka, P.; Lok, C.N. The transferrin receptor: Role in health and disease. Int. J. Biochem. Cell Biol. 1999, 31, 1111–1137. [Google Scholar] [CrossRef]
- Daniels, T.R.; Delgado, T.; Rodriguez, J.A.; Helguera, G.; Penichet, M.L. The transferrin receptor part I: Biology and targeting with cytotoxic antibodies for the treatment of cancer. Clin. Immunol. 2006, 121, 144–158. [Google Scholar] [CrossRef] [PubMed]
- Saxena, M.; Delgado, Y.; Sharma, R.K.; Sharma, S.; Guzmán, S.L.P.D.L.; Tinoco, A.D.; Griebenow, K. Inducing cell death in vitro in cancer cells by targeted delivery of cytochrome c via a transferrin conjugate. PLoS ONE 2018, 13, e0195542. [Google Scholar] [CrossRef]
- Candelaria, P.V.; Leoh, L.S.; Penichet, M.L.; Daniels-Wells, T.R. Antibodies targeting the transferrin receptor 1 (TfR1) as direct anti-cancer agents. Front. Immunol. 2021, 12, 583. [Google Scholar] [CrossRef]
- Deshpande, P.; Jhaveri, A.; Pattni, B.; Biswas, S.; Torchilin, V. Transferrin and octaarginine modified dual-functional liposomes with improved cancer cell targeting and enhanced intracellular delivery for the treatment of ovarian cancer. Drug Deliv. 2018, 25, 517–532. [Google Scholar] [CrossRef]
- Soe, Z.C.; Kwon, J.B.; Thapa, R.K.; Ou, W.; Nguyen, H.T.; Gautam, M.; Oh, K.T.; Choi, H.-G.; Ku, S.K.; Yong, C.S.; et al. Transferrin-conjugated polymeric nanoparticle for receptor-mediated delivery of doxorubicin in doxorubicin-resistant breast cancer cells. Pharmaceutics 2019, 11, 63. [Google Scholar] [CrossRef] [Green Version]
- De Freitas, C.F.; Montanha, M.C.; Pellosi, D.S.; Kimura, E.; Caetano, W.; Hioka, N. Biotin-targeted mixed liposomes: A smart strategy for selective release of a photosensitizer agent in cancer cells. Mater. Sci. Eng. C 2019, 104, 109923. [Google Scholar] [CrossRef]
- Ren, W.X.; Han, J.; Uhm, S.; Jang, Y.J.; Kang, C.; Kim, J.-H. Recent development of biotin conjugation in biological imaging, sensing, and target delivery. Chem. Commun. 2015, 51, 10403–10418. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, Q.; Lv, L.; Fu, J.; Jiang, Y.; Xin, H.; Yao, Q. Glioma and microenvironment dual targeted nanocarrier for improved antiglioblastoma efficacy. Drug Deliv. 2017, 24, 1401–1409. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Reinhard, S.; Li, C.; Qian, M.; Jiang, H.; Du, Y.; Lächelt, U.; Lu, W.; Wagner, E.; Huang, R. Antitumoral cascade-targeting ligand for il-6 receptor-mediated gene delivery to glioma. Mol. Ther. 2017, 25, 1556–1566. [Google Scholar] [CrossRef] [Green Version]
- Malaguarnera, R.; Belfiore, A. The Insulin Receptor: A new target for cancer therapy. Front. Endocrinol. 2011, 2, 93. [Google Scholar] [CrossRef] [Green Version]
- Nicolas, J.; Mura, S.; Brambilla, D.; Mackiewicz, N.; Couvreur, P. Design, functionalization strategies and biomedical applications of targeted biodegradable/biocompatible polymer-based nanocarriers for drug delivery. Chem. Soc. Rev. 2013, 42, 1147–1235. [Google Scholar] [CrossRef] [PubMed]
- Mazzotta, E.; De Benedittis, S.; Qualtieri, A.; Muzzalupo, R. Actively targeted and redox responsive delivery of anticancer drug by chitosan nanoparticles. Pharmaceutics 2019, 12, 26. [Google Scholar] [CrossRef] [Green Version]
- Matini, T.; Francini, N.; Battocchio, A.; Spain, S.; Mantovani, G.; Vicent, M.J.; Sanchis, J.; Gallon, E.; Mastrotto, F.; Salmaso, S.; et al. Synthesis and characterization of variable conformation pH responsive block co-polymers for nucleic acid delivery and targeted cell entry. Polym. Chem. 2014, 5, 1626–1636. [Google Scholar] [CrossRef] [Green Version]
- Gallon, E.; Matini, T.; Sasso, L.; Mantovani, G.; De Benito, A.A.; Sanchis, J.; Caliceti, P.; Alexander, C.; Vicent, M.J.; Salmaso, S. Triblock copolymer nanovesicles for pH-responsive targeted delivery and controlled release of siRNA to cancer cells. Biomacromolecules 2015, 16, 1924–1937. [Google Scholar] [CrossRef] [PubMed]
- Curcio, M.; Paolì, A.; Cirillo, G.; Di Pietro, S.; Forestiero, M.; Giordano, F.; Mauro, L.; Amantea, D.; Di Bussolo, V.; Nicoletta, F.; et al. Combining dextran conjugates with stimuli-responsive and folate-targeting activity: A new class of multifunctional nanoparticles for cancer therapy. Nanomaterials 2021, 11, 1108. [Google Scholar] [CrossRef] [PubMed]
- Venuta, A.; Moret, F.; Poggetto, G.D.; Esposito, D.; Fraix, A.; Avitabile, C.; Ungaro, F.; Malinconico, M.; Sortino, S.; Romanelli, A.; et al. Shedding light on surface exposition of poly(ethylene glycol) and folate targeting units on nanoparticles of poly(ε-caprolactone) diblock copolymers: Beyond a paradigm. Eur. J. Pharm. Sci. 2018, 111, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Conte, C.; Fotticchia, I.; Tirino, P.; Moret, F.; Pagano, B.; Gref, R.; Ungaro, F.; Reddi, E.; Giancola, C.; Quaglia, F. Cyclodextrin-assisted assembly of PEGylated polyester nanoparticles decorated with folate. Colloids Surf. B Biointerfaces 2016, 141, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Poggetto, G.D.; Troise, S.S.; Conte, C.; Marchetti, R.; Moret, F.; Iadonisi, A.; Silipo, A.; Lanzetta, R.; Malinconico, M.; Quaglia, F.; et al. Nanoparticles decorated with folate based on a site-selective αCD-rotaxanated PEG-b-PCL copolymer for targeted cancer therapy. Polym. Chem. 2020, 11, 3892–3903. [Google Scholar] [CrossRef]
- Chiesa, E.; Dorati, R.; Conti, B.; Modena, T.; Cova, E.; Meloni, F.; Genta, I. Hyaluronic acid-decorated chitosan nanoparticles for CD44-targeted delivery of everolimus. Int. J. Mol. Sci. 2018, 19, 2310. [Google Scholar] [CrossRef] [Green Version]
- Chiesa, E.; Riva, F.; Dorati, R.; Greco, A.; Ricci, S.; Pisani, S.; Patrini, M.; Modena, T.; Conti, B.; Genta, I. On-Chip synthesis of hyaluronic acid-based nanoparticles for selective inhibition of CD44+ human mesenchymal stem cell proliferation. Pharmaceutics 2020, 12, 260. [Google Scholar] [CrossRef] [Green Version]
- Curcio, M.; Diaz-Gomez, L.; Cirillo, G.; Nicoletta, F.; Leggio, A.; Iemma, F. Dual-targeted hyaluronic acid/albumin micelle-like nanoparticles for the vectorization of doxorubicin. Pharmaceutics 2021, 13, 304. [Google Scholar] [CrossRef] [PubMed]
- Cosco, D.; Mare, R.; Paolino, D.; Salvatici, M.C.; Cilurzo, F.; Fresta, M. Sclareol-loaded hyaluronan-coated PLGA nanoparticles: Physico-chemical properties and in vitro anticancer features. Int. J. Biol. Macromol. 2019, 132, 550–557. [Google Scholar] [CrossRef] [PubMed]
- Maiolino, S.; Russo, A.; Pagliara, V.; Conte, C.; Ungaro, F.; Russo, G.; Quaglia, F. Biodegradable nanoparticles sequentially decorated with Polyethyleneimine and Hyaluronan for the targeted delivery of docetaxel to airway cancer cells. J. Nanobiotechnol. 2015, 13, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Russo, A.; Maiolino, S.; Pagliara, V.; Ungaro, F.; Tatangelo, F.; Leone, A.; Scalia, G.; Budillon, A.; Quaglia, F.; Russo, G. Enhancement of 5-FU sensitivity by the proapoptotic rpL3 gene in p53 null colon cancer cells through combined polymer nanoparticles. Oncotarget 2016, 7, 79670–79687. [Google Scholar] [CrossRef] [Green Version]
- Gaio, E.; Conte, C.; Esposito, D.; Miotto, G.; Quaglia, F.; Moret, F.; Reddi, E. Co-delivery of docetaxel and disulfonate tetraphenyl chlorin in one nanoparticle produces strong synergism between chemo- and photodynamic therapy in drug-sensitive and -resistant cancer cells. Mol. Pharm. 2018, 15, 4599–4611. [Google Scholar] [CrossRef] [PubMed]
- Gaio, E.; Conte, C.; Esposito, D.; Reddi, E.; Quaglia, F.; Moret, F. CD44 targeting mediated by polymeric nanoparticles and combination of chlorine TPCS2a-PDT and docetaxel-chemotherapy for efficient killing of breast differentiated and stem cancer cells in vitro. Cancers 2020, 12, 278. [Google Scholar] [CrossRef] [Green Version]
- Maiolino, S.; Moret, F.; Conte, C.; Fraix, A.; Tirino, P.; Ungaro, F.; Sortino, S.; Reddi, E.; Quaglia, F. Hyaluronan-decorated polymer nanoparticles targeting the CD44 receptor for the combined photo/chemo-therapy of cancer. Nanoscale 2015, 7, 5643–5653. [Google Scholar] [CrossRef]
- Russo, A.; Pellosi, D.; Pagliara, V.; Milone, M.R.; Pucci, B.; Caetano, W.; Hioka, N.; Budillon, A.; Ungaro, F.; Russo, G.; et al. Biotin-targeted Pluronic ® P123/F127 mixed micelles delivering niclosamide: A repositioning strategy to treat drug-resistant lung cancer cells. Int. J. Pharm. 2016, 511, 127–139. [Google Scholar] [CrossRef] [Green Version]
- Pellosi, D.S.; Calori, I.R.; De Paula, L.B.; Hioka, N.; Quaglia, F.; Tedesco, A.C. Multifunctional theranostic Pluronic mixed micelles improve targeted photoactivity of Verteporfin in cancer cells. Mater. Sci. Eng. C 2017, 71, 1–9. [Google Scholar] [CrossRef]
- Conte, C.; Moret, F.; Esposito, D.; Poggetto, G.D.; Avitabile, C.; Ungaro, F.; Romanelli, A.; Laurienzo, P.; Reddi, E.; Quaglia, F. Biodegradable nanoparticles exposing a short anti-FLT1 peptide as antiangiogenic platform to complement docetaxel anticancer activity. Mater. Sci. Eng. C 2019, 102, 876–886. [Google Scholar] [CrossRef]
- Voltan, R.; Secchiero, P.; Ruozi, B.; Forni, F.; Agostinis, C.; Caruso, L.; Vandelli, M.A.; Zauli, G. nanoparticles engineered with rituximab and loaded with nutlin-3 show promising therapeutic activity in b-leukemic xenografts. Clin. Cancer Res. 2013, 19, 3871–3880. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Belletti, D.; Tosi, G.; Riva, G.; Lagreca, I.; Galliania, M.; Luppi, M.; Vandelli, M.; Forni, F.; Ruozi, B. Nutlin-3 loaded nanocarriers: Preparation, characterization and in vitro antineoplastic effect against primary effusion lymphoma. Int. J. Pharm. 2015, 490, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Akl, M.A.; Kartal-Hodzic, A.; Suutari, T.; Oksanen, T.; Montagner, I.M.; Rosato, A.; Ismael, H.R.; Afouna, M.I.; Caliceti, P.; Yliperttula, M.; et al. Real-time label-free targeting assessment and in vitro characterization of curcumin-loaded poly-lactic-co-glycolic acid nanoparticles for oral colon targeting. ACS Omega 2019, 4, 16878–16890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Colzani, B.; Speranza, G.; Dorati, R.; Conti, B.; Modena, T.; Bruni, G.; Zagato, E.; Vermeulen, L.; Dakwar, G.R.; Braeckmans, K.; et al. Design of smart GE11-PLGA/PEG-PLGA blend nanoparticulate platforms for parenteral administration of hydrophilic macromolecular drugs: Synthesis, preparation and in vitro/ex vivo characterization. Int. J. Pharm. 2016, 511, 1112–1123. [Google Scholar] [CrossRef] [PubMed]
- Laquintana, V.; Denora, N.; Lopalco, A.; Lopedota, A.; Cutrignelli, A.; Lasorsa, F.; Agostino, G.; Franco, M. translocator protein ligand–plga conjugated nanoparticles for 5-fluorouracil delivery to glioma cancer cells. Mol. Pharm. 2014, 11, 859–871. [Google Scholar] [CrossRef] [PubMed]
- Denora, N.; Laquintana, V.; Lopalco, A.; Iacobazzi, R.M.; Lopedota, A.; Cutrignelli, A.; Iacobellis, G.; Annese, C.; Cascione, M.; Leporatti, S.; et al. In vitro targeting and imaging the translocator protein TSPO 18-kDa through G(4)-PAMAM–FITC labeled dendrimer. J. Control. Release 2013, 172, 1111–1125. [Google Scholar] [CrossRef]
- Iacobazzi, R.M.; Porcelli, L.; Lopedota, A.A.; Laquintana, V.; Lopalco, A.; Cutrignelli, A.; Altamura, E.; Di Fonte, R.; Azzariti, A.; Franco, M.; et al. Targeting human liver cancer cells with lactobionic acid-G(4)-PAMAM-FITC sorafenib loaded dendrimers. Int. J. Pharm. 2017, 528, 485–497. [Google Scholar] [CrossRef]
- Salerno, M.; Cenni, E.; Fotia, C.; Avnet, S.; Granchi, D.; Castelli, F.; Micieli, D.; Pignatello, R.; Capulli, M.; Rucci, N.; et al. Bone-targeted doxorubicin-loaded nanoparticles as a tool for the treatment of skeletal metastases. Curr. Cancer Drug Targets 2010, 10, 649–659. [Google Scholar] [CrossRef]
- Cenni, E.; Avnet, S.; Granchi, D.; Fotia, C.; Salerno, M.; Micieli, D.; Sarpietro, M.G.; Pignatello, R.; Castelli, F.; Baldini, N. The effect of poly(d,l-lactide-co-glycolide)-alendronate conjugate nanoparticles on human osteoclast precursors. J. Biomater. Sci. Polym. Ed. 2012, 23, 1285–1300. [Google Scholar] [CrossRef]
- Clementi, C.; Miller, K.; Mero, A.; Satchi-Fainaro, R.; Pasut, G. Dendritic Poly(ethylene glycol) Bearing Paclitaxel and Alendronate for Targeting Bone Neoplasms. Mol. Pharm. 2011, 8, 1063–1072. [Google Scholar] [CrossRef]
- Cavalli, R.; Soster, M.; Argenziano, M. Nanobubbles: A promising efficient tool for therapeutic delivery. Ther. Deliv. 2016, 7, 117–138. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; Chang, J.; Dou, P.; Jin, S.; Jiao, M.; Tang, H.; Jiang, W.; Ren, W.; Zheng, S. Tumor targeted multifunctional magnetic nanobubbles for MR/US dual imaging and focused ultrasound triggered drug delivery. Front. Bioeng. Biotechnol. 2020, 8, 586874. [Google Scholar] [CrossRef] [PubMed]
- Xiao, R.; Zhao, Z.; Chen, J.; He, L.; Wang, H.; Huang, L.; Luo, B. Preparation and ultrasonic imaging investigation of perfluoropentane-filled polylactic acid nanobubbles as a novel targeted ultrasound contrast agent. Front. Mater. 2020, 7, 416. [Google Scholar] [CrossRef]
- Xu, J.S.; Huang, J.; Qin, R.; Hinkle, G.H.; Povoski, S.P.; Martin, E.W.; Xu, R.X. Synthesizing and binding dual-mode poly (lactic-co-glycolic acid) (PLGA) nanobubbles for cancer targeting and imaging. Biomaterials 2010, 31, 1716–1722. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zheng, Y.; Wang, Z.; Huang, S.; Chen, Y.; Jiang, W.; Zhang, H.; Ding, M.; Li, Q.; Xiao, X.; et al. Methotrexate-loaded PLGA nanobubbles for ultrasound imaging and synergistic targeted therapy of residual tumor during HIFU ablation. Biomaterials 2014, 35, 5148–5161. [Google Scholar] [CrossRef] [PubMed]
- Capolla, S.; Argenziano, M.; Bozzer, S.; D’Agaro, T.; Bittolo, T.; De Leo, L.; Not, T.; Busato, D.; Dal Bo, M.; Toffoli, G.; et al. Targeted chitosan nanobubbles as a new delivery approach required for anti-microRNA-17 based systemic therapy in Burkitt lymphoma models. Small 2021. submitted for publication. [Google Scholar]
- Argenziano, M.; Occhipinti, S.; Guiot, C.; Giovarelli, M.; Cavalli, R. Nanobubble-based HER2 immunotherapy through dendritic cells targeting. Presented at the CRS Italy Chapter Workshop 2017, Padova, Italy, 26–28 October 2017. [Google Scholar]
- Gulati, M.; Grover, M.; Singh, S.; Singh, M. Lipophilic drug derivatives in liposomes. Int. J. Pharm. 1998, 165, 129–168. [Google Scholar] [CrossRef]
- Crommelin, D.J.; Van Hoogevest, P.; Storm, G. The role of liposomes in clinical nanomedicine development. What now? Now what? J. Control. Release 2019, 318, 256–263. [Google Scholar] [CrossRef]
- Shah, S.; Dhawan, V.; Holm, R.; Nagarsenker, M.S.; Perrie, Y. Liposomes: Advancements and innovation in the manufacturing process. Adv. Drug Deliv. Rev. 2020, 154–155, 102–122. [Google Scholar] [CrossRef]
- D’Avanzo, N.; Torrieri, G.; Figueiredo, P.; Celia, C.; Paolino, D.; Correia, A.; Moslova, K.; Teesalu, T.; Fresta, M.; Santos, H.A. LinTT1 peptide-functionalized liposomes for targeted breast cancer therapy. Int. J. Pharm. 2021, 597, 120346. [Google Scholar] [CrossRef]
- Corti, A.; Curnis, F.; Rossoni, G.; Marcucci, F.; Gregorc, V. peptide-mediated targeting of cytokines to tumor vasculature: The NGR-hTNF Example. BioDrugs 2013, 27, 591–603. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zuccari, G.; Milelli, A.; Pastorino, F.; Loi, M.; Petretto, A.; Parise, A.; Marchetti, C.; Minarini, A.; Cilli, M.; Emionite, L.; et al. Tumor vascular targeted liposomal-bortezomib minimizes side effects and increases therapeutic activity in human neuroblastoma. J. Control. Release 2015, 211, 44–52. [Google Scholar] [CrossRef]
- Di Paolo, D.; Pastorino, F.; Zuccari, G.; Caffa, I.; Loi, M.; Marimpietri, D.; Brignole, C.; Perri, P.; Cilli, M.; Nico, B.; et al. Enhanced anti-tumor and anti-angiogenic efficacy of a novel liposomal fenretinide on human neuroblastoma. J. Control. Release 2013, 170, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Morelli, G.; Accardo, A.; Tesauro, D.; Cicala, C.; Salzano, G.; De Rosa, G.; Morisco, A.; Aloj, L.; Aurilio, M.; Maione, F.; et al. Peptide-modified liposomes for selective targeting of bombesin receptors overexpressed by cancer cells: A potential theranostic agent. Int. J. Nanomed. 2012, 7, 2007–2017. [Google Scholar] [CrossRef] [Green Version]
- Accardo, A.; Mansi, R.; Salzano, G.; Morisco, A.; Aurilio, M.; Parisi, A.; Maione, F.; Cicala, C.; Ziaco, B.; Tesauro, D.; et al. Bombesin peptide antagonist for target-selective delivery of liposomal doxorubicin on cancer cells. J. Drug Target. 2012, 21, 240–249. [Google Scholar] [CrossRef] [Green Version]
- Burger, J.A.; Kipps, T.J. CXCR4: A key receptor in the crosstalk between tumor cells and their microenvironment. Blood 2006, 107, 1761–1767. [Google Scholar] [CrossRef]
- Ierano’, C.; Portella, L.; Lusa, S.; Salzano, G.; D’Alterio, C.; Napolitano, M.; Buoncervello, M.; Macchia, D.; Spada, M.; Barbieri, A.; et al. CXCR4-antagonist Peptide R-liposomes for combined therapy against lung metastasis. Nanoscale 2016, 8, 7562–7571. [Google Scholar] [CrossRef]
- Ong, K.L.; Lam, K.S.L.; Cheung, B.M.Y. Urotensin II: Its function in health and its role in disease. Cardiovasc. Drugs Ther. 2005, 19, 65–75. [Google Scholar] [CrossRef] [Green Version]
- Zappavigna, S.; Abate, M.; Cossu, A.M.; Lusa, S.; Campani, V.; Scotti, L.; Luce, A.; Yousif, A.M.; Merlino, F.; Grieco, P.; et al. Urotensin-II-targeted liposomes as a new drug delivery system towards prostate and colon cancer cells. J. Oncol. 2019, 2019, 1–14. [Google Scholar] [CrossRef]
- Rinaldi, F.; Hanieh, P.N.; Del Favero, E.; Rondelli, V.; Brocca, P.; Pereira, M.C.; Andreev, O.A.; Reshetnyak, Y.K.; Marianecci, C.; Carafa, M. Decoration of Nanovesicles with pH (Low) Insertion Peptide (pHLIP) for Targeted Delivery. Nanoscale Res. Lett. 2018, 13, 391. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.C.; Pianella, M.; Wei, D.; Moshnikova, A.; Marianecci, C.; Carafa, M.; Andreev, O.A.; Reshetnyak, Y.K. pH-sensitive pHLIP® coated niosomes. Mol. Membr. Biol. 2016, 33, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Uchegbu, I.F.; Vyas, S.P. Non-ionic surfactant based vesicles (niosomes) in drug delivery. Int. J. Pharm. 1998, 172, 33–70. [Google Scholar] [CrossRef]
- Lasic, D. On the thermodynamic stability of liposomes. J. Colloid Interface Sci. 1990, 140, 302–304. [Google Scholar] [CrossRef]
- Handjani-Vila, R.M.; Ribier, A.; Rondot, B.; Vanlerberghie, G. Dispersions of lamellar phases of non-ionic lipids in cosmetic products. Int. J. Cosmet. Sci. 1979, 1, 303–314. [Google Scholar] [CrossRef] [PubMed]
- Florence, A.T. New drug delivery systems. Chem. Ind. 1993, 20, 1000–1004. [Google Scholar]
- Yeo, P.L.; Lim, C.L.; Chye, S.M.; Ling, A.P.K.; Koh, R.Y. Niosomes: A review of their structure, properties, methods of preparation, and medical applications. Asian Biomed. 2017, 11, 301–314. [Google Scholar] [CrossRef] [Green Version]
- Kuotsu, K.; Karim, K.M.; Mandal, A.S.; Biswas, N.; Guha, A.; Chatterjee, S.; Behera, M. Niosome: A future of targeted drug delivery systems. J. Adv. Pharm. Technol. Res. 2010, 1, 374–380. [Google Scholar] [CrossRef]
- Pozza, E.D.; Lerda, C.; Costanzo, C.; Donadelli, M.; Dando, I.; Zoratti, E.; Scupoli, M.; Beghelli, S.; Scarpa, A.; Fattal, E.; et al. Targeting gemcitabine containing liposomes to CD44 expressing pancreatic adenocarcinoma cells causes an increase in the antitumoral activity. Biochim. et Biophys. Acta (BBA) Biomembr. 2013, 1828, 1396–1404. [Google Scholar] [CrossRef] [Green Version]
- Arpicco, S.; Lerda, C.; Pozza, E.D.; Costanzo, C.; Tsapis, N.; Stella, B.; Donadelli, M.; Dando, I.; Fattal, E.; Cattel, L.; et al. Hyaluronic acid-coated liposomes for active targeting of gemcitabine. Eur. J. Pharm. Biopharm. 2013, 85, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Marengo, A.; Forciniti, S.; Dando, I.; Pozza, E.D.; Stella, B.; Tsapis, N.; Yagoubi, N.; Fanelli, G.; Fattal, E.; Heeschen, C.; et al. Pancreatic cancer stem cell proliferation is strongly inhibited by diethyldithiocarbamate-copper complex loaded into hyaluronic acid decorated liposomes. Biochim. et Biophys. Acta (BBA)-Gen. Subj. 2018, 1863, 61–72. [Google Scholar] [CrossRef] [Green Version]
- Gazzano, E.; Buondonno, I.; Marengo, A.; Rolando, B.; Chegaev, K.; Kopecka, J.; Saponara, S.; Sorge, M.; Hattinger, C.M.; Gasco, A.; et al. Hyaluronated liposomes containing H2S-releasing doxorubicin are effective against P-glycoprotein-positive/doxorubicin-resistant osteosarcoma cells and xenografts. Cancer Lett. 2019, 456, 29–39. [Google Scholar] [CrossRef]
- Cano, M.E.; Lesur, D.; Bincoletto, V.; Gazzano, E.; Stella, B.; Riganti, C.; Arpicco, S.; Kovensky, J. Synthesis of defined oligohyaluronates-decorated liposomes and interaction with lung cancer cells. Carbohydr. Polym. 2020, 248, 116798. [Google Scholar] [CrossRef]
- Santana-Armas, M.L.; De Ilarduya, C.T. Strategies for cancer gene-delivery improvement by non-viral vectors. Int. J. Pharm. 2021, 596, 120291. [Google Scholar] [CrossRef] [PubMed]
- Ponti, F.; Campolungo, M.; Melchiori, C.; Bono, N.; Candiani, G. Cationic lipids for gene delivery: Many players, one goal. Chem. Phys. Lipids 2021, 235, 105032. [Google Scholar] [CrossRef] [PubMed]
- Surace, C.; Arpicco, S.; Dufaÿ-Wojcicki, A.; Marsaud, V.; Bouclier, C.; Clay, D.; Cattel, L.; Renoir, J.-M.; Fattal, E. Lipoplexes targeting the CD44 hyaluronic acid receptor for efficient transfection of breast cancer cells. Mol. Pharm. 2009, 4, 1062–1073. [Google Scholar]
- Wojcicki, A.D.; Hillaireau, H.; Nascimento, T.L.; Arpicco, S.; Taverna, M.; Ribes, S.; Bourge, M.; Nicolas, V.; Bochot, A.; Vauthier, C.; et al. Hyaluronic acid-bearing lipoplexes: Physico-chemical characterization and in vitro targeting of the CD44 receptor. J. Control. Release 2012, 162, 545–552. [Google Scholar] [CrossRef]
- Taetz, S.; Bochot, A.; Surace, C.; Arpicco, S.; Renoir, J.-M.; Schaefer, U.F.; Marsaud, V.; Kerdine-Roemer, S.; Lehr, C.-M.; Fattal, E. Hyaluronic acid-modified DOTAP/dope liposomes for the targeted delivery of anti-telomerase siRNA to CD44-expressing lung cancer cells. Oligonucleotides 2009, 19, 103–116. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, T.L.; Hillaireau, H.; Vergnaud, J.; Rivano, M.; Deloménie, C.; Courilleau, D.; Arpicco, S.; Suk, J.S.; Hanes, J.; Fattal, E. Hyaluronic acid-conjugated lipoplexes for targeted delivery of siRNA in a murine metastatic lung cancer model. Int. J. Pharm. 2016, 514, 103–111. [Google Scholar] [CrossRef]
- Nascimento, T.L.; Hillaireau, H.; Noiray, M.; Bourgaux, C.; Arpicco, S.; Pehau-Arnaudet, G.; Taverna, M.; Cosco, D.; Tsapis, N.; Fattal, E. Supramolecular organization and siRNA binding of hyaluronic acid-coated lipoplexes for targeted delivery to the CD44 receptor. Langmuir 2015, 31, 11186–11194. [Google Scholar] [CrossRef]
- Leto, I.; Coronnello, M.; Righeschi, C.; Bergonzi, M.C.; Mini, E.; Bilia, A.R. Enhanced efficacy of artemisinin loaded in transferrin-conjugated liposomes versus stealth liposomes against HCT-8 colon cancer cells. ChemMedChem 2016, 11, 1745–1751. [Google Scholar] [CrossRef]
- Salzano, G.; Marra, M.; Porru, M.; Zappavigna, S.; Abbruzzese, A.; La Rotonda, M.; Leonetti, C.; Caraglia, M.; De Rosa, G. Self-assembly nanoparticles for the delivery of bisphosphonates into tumors. Int. J. Pharm. 2010, 403, 292–297. [Google Scholar] [CrossRef] [PubMed]
- Ristori, S.; Grillo, I.; Lusa, S.; Thamm, J.; Valentino, G.; Campani, V.; Caraglia, M.; Steiniger, F.; Luciani, P.; De Rosa, G. Structural characterization of self-assembling hybrid nanoparticles for bisphosphonate delivery in tumors. Mol. Pharm. 2018, 15, 1258–1265. [Google Scholar] [CrossRef] [PubMed]
- Porru, M.; Zappavigna, S.; Salzano, G.; Luce, A.; Stoppacciaro, A.; Balestrieri Artuso, S.; Lusa, S.; De Rosa, G.; Leonetti, C.; Caraglia, M.; et al. Medical treatment of orthotopic glioblastoma with transferrin-conjugated nanoparticles encapsulating zoledronic acid. Oncotarget 2014, 5, 10446–10459. [Google Scholar] [PubMed] [Green Version]
- Salzano, G.; Zappavigna, S.; Luce, A.; D’Onofrio, N.; Balestrieri, M.L.; Grimaldi, A.; Lusa, S.; Ingrosso, D.; Artuso, S.; Porru, M.; et al. Transferrin-targeted nanoparticles containing zoledronic acid as a potential tool to inhibit glioblastoma growth. J. Biomed. Nanotechnol. 2016, 12, 811–830. [Google Scholar] [CrossRef] [Green Version]
- Scognamiglio, I.; Di Martino, M.T.; Campani, V.; Virgilio, A.; Galeone, A.; Gullà, A.; Cantafio, M.E.G.; Misso, G.; Tagliaferri, P.; Tassone, P.; et al. Transferrin-Conjugated SNALPs Encapsulating 2′-O-Methylated miR-34a for the Treatment of Multiple Myeloma. BioMed Res. Int. 2014, 2014, 1–7. [Google Scholar] [CrossRef]
- Tavano, L.; Muzzalupo, R.; Mauro, L.; Pellegrino, M.; Andò, S.; Picci, N. Transferrin-Conjugated Pluronic Niosomes as a New Drug Delivery System for Anticancer Therapy. Langmuir 2013, 29, 12638–12646. [Google Scholar] [CrossRef]
- Tavano, L.; Mauro, L.; Naimo, G.D.; Bruno, L.; Picci, N.; Andò, S.; Muzzalupo, R. further evolution of multifunctional niosomes based on pluronic surfactant: Dual active targeting and drug combination properties. Langmuir 2016, 32, 8926–8933. [Google Scholar] [CrossRef]
- Paolino, D.; Licciardi, M.; Celia, C.; Giammona, G.; Fresta, M.; Cavallaro, G. Folate-targeted supramolecular vesicular aggregates as a new frontier for effective anticancer treatment in in vivo model. Eur. J. Pharm. Biopharm. 2012, 82, 94–102. [Google Scholar] [CrossRef]
- Scomparin, A.; Salmaso, S.; Eldar-Boock, A.; Ben-Shushan, D.; Ferber, S.; Tiram, G.; Shmeeda, H.; Landa-Rouben, N.; Leor, J.; Caliceti, P.; et al. A comparative study of folate receptor-targeted doxorubicin delivery systems: Dosing regimens and therapeutic index. J. Control. Release 2015, 208, 106–120. [Google Scholar] [CrossRef]
- Gazzano, E.; Rolando, B.; Chegaev, K.; Salaroglio, I.C.; Kopecka, J.; Pedrini, I.; Saponara, S.; Sorge, M.; Buondonno, I.; Stella, B.; et al. Folate-targeted liposomal nitrooxy-doxorubicin: An effective tool against P-glycoprotein-positive and folate receptor-positive tumors. J. Control. Release 2018, 270, 37–52. [Google Scholar] [CrossRef] [Green Version]
- Cheung, N.K.; Saarinen, U.M.; Neely, J.E.; Landmeier, B.; Donovan, D.; Coccia, P.F. Monoclonal antibodies to a glycolipid antigen on human neuroblastoma cells. Cancer Res. 1985, 45, 2642–2649. [Google Scholar] [PubMed]
- Pagnan, G.; Montaldo, P.G.; Pastorino, F.; Raffaghello, L.; Kirchmeier, M.; Allen, T.M.; Ponzoni, M. GD2-mediated melanoma cell targeting and cytotoxicity of liposome-entrapped fenretinide. Int. J. Cancer 1999, 81, 268–274. [Google Scholar] [PubMed]
- Brown, B.S.; Patanam, T.; Mobli, K.; Celia, C.; Zage, P.E.; Bean, A.J.; Tasciotti, E. Etoposide-loaded immunoliposomes as active targeting agents for GD2-positive malignancies. Cancer Biol. Ther. 2014, 15, 851–861. [Google Scholar] [CrossRef] [Green Version]
- Scavo, M.P.; Cutrignelli, A.; DePalo, N.; Fanizza, E.; Laquintana, V.; Gasparini, G.; Giannelli, G.; Denora, N. Effectiveness of a controlled 5-FU delivery based on FZD10 antibody-conjugated liposomes in colorectal cancer in vitro models. Pharmaceutics 2020, 12, 650. [Google Scholar] [CrossRef]
- Ruozi, B.; Riva, G.; Belletti, D.; Tosi, G.; Barozzi, P.; Luppi, M.; Forni, F.; Vandelli, M.A. Immunoliposomal systems targeting primary effusion lymphoma: In vitro study. Nanomedicine 2010, 5, 1051–1064. [Google Scholar] [CrossRef] [PubMed]
- Belletti, D.; Vandelli, M.A.; Tonelli, M.; Zapparoli, M.; Forni, F.; Tosi, G.; Ruozi, B. Functionalization of liposomes: Microscopical methods for preformulative screening. J. Liposome Res. 2014, 25, 1–7. [Google Scholar] [CrossRef]
- Bragagni, M.; Mennini, N.; Ghelardini, C.; Mura, P. Development and characterization of niosomal formulations of doxorubicin aimed at brain targeting. J. Pharm. Pharm. Sci. 2012, 15, 184–196. [Google Scholar] [CrossRef] [Green Version]
- Barattin, M.; Mattarei, A.; Balasso, A.; Paradisi, C.; Cantù, L.; Del Favero, E.; Viitala, T.; Mastrotto, F.; Caliceti, P.; Salmaso, S. pH-controlled liposomes for enhanced cell penetration in tumor environment. ACS Appl. Mater. Interfaces 2018, 10, 17646–17661. [Google Scholar] [CrossRef]
- Popilski, H.; Feinshtein, V.; Kleiman, S.; Mattarei, A.; Garofalo, M.; Salmaso, S.; Stepensky, D. Doxorubicin liposomes cell penetration enhancement and its potential drawbacks for the tumor targeting efficiency. Int. J. Pharm. 2020, 592, 120012. [Google Scholar] [CrossRef]
- Iwamaru, Y.; Shimizu, Y.; Imamura, M.; Murayama, Y.; Endo, R.; Tagawa, Y.; Ushiki-Kaku, Y.; Takenouchi, T.; Kitani, H.; Mohri, S.; et al. Lactoferrin induces cell surface retention of prion protein and inhibits prion accumulation. J. Neurochem. 2008, 107, 636–646. [Google Scholar] [CrossRef]
- Pireddu, R.; Pibiri, M.; Valenti, D.; Sinico, C.; Fadda, A.M.; Simbula, G.; Lai, F. A novel lactoferrin-modified stealth liposome for hepatoma-delivery of triiodothyronine. Int. J. Pharm. 2018, 537, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Gérard, A.-C.; Daumerie, C.; Mestdagh, C.; Gohy, S.; De Burbure, C.; Costagliola, S.; Miot, F.; Nollevaux, M.-C.; Denef, J.-F.; Rahier, J.; et al. correlation between the loss of thyroglobulin iodination and the expression of thyroid-specific proteins involved in iodine metabolism in thyroid carcinomas. J. Clin. Endocrinol. Metab. 2003, 88, 4977–4983. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paolino, D.; Cosco, D.; Gaspari, M.; Celano, M.; Wolfram, J.; Voce, P.; Puxeddu, E.; Filetti, S.; Celia, C.; Ferrari, M.; et al. Targeting the thyroid gland with thyroid-stimulating hormone (TSH)-nanoliposomes. Biomaterials 2014, 35, 7101–7109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duong, V.-A.; Nguyen, T.-T.; Maeng, H.-J. Preparation of Solid lipid nanoparticles and nanostructured lipid carriers for drug delivery and the effects of preparation parameters of solvent injection method. Molecules 2020, 25, 4781. [Google Scholar] [CrossRef]
- Magro, R.D.; Ornaghi, F.; Cambianica, I.; Beretta, S.; Re, F.; Musicanti, C.; Rigolio, R.; Donzelli, E.; Canta, A.R.; Ballarini, E.; et al. ApoE-modified solid lipid nanoparticles: A feasible strategy to cross the blood-brain barrier. J. Control. Release 2017, 249, 103–110. [Google Scholar] [CrossRef]
- Béduneau, A.; Saulnier, P.; Benoit, J.-P. Active targeting of brain tumors using nanocarriers. Biomaterials 2007, 28, 4947–4967. [Google Scholar] [CrossRef]
- Cortesi, R.; Menegatti, E.; Esposito, E.; Ravani, L.; Drechsler, M. Colloidal dispersions for the delivery of acyclovir: A comparative study. Indian J. Pharm. Sci. 2011, 73, 687–693. [Google Scholar] [CrossRef] [Green Version]
- Mohammadi-Samani, S.; Ghasemiyeh, P. Solid lipid nanoparticles and nanostructured lipid carriers as novel drug delivery systems: Applications, advantages and disadvantages. Res. Pharm. Sci. 2018, 13, 288–303. [Google Scholar] [CrossRef]
- Battaglia, L.; Muntoni, E.; Chirio, D.; Peira, E.; Annovazzi, L.; Schiffer, D.; Mellai, M.; Riganti, C.; Salaroglio, I.C.; Lanotte, M.; et al. Solid lipid nanoparticles by coacervation loaded with a methotrexate prodrug: Preliminary study for glioma treatment. Nanomedicine 2017, 12, 639–656. [Google Scholar] [CrossRef]
- Muntoni, E.; Martina, K.; Marini, E.; Giorgis, M.; Lazzarato, L.; Salaroglio, I.C.; Riganti, C.; Lanotte, M.; Battaglia, L. Methotrexate-loaded solid lipid nanoparticles: Protein functionalization to improve brain biodistribution. Pharmaceutics 2019, 11, 65. [Google Scholar] [CrossRef] [Green Version]
- Desmaële, D.; Gref, R.; Couvreur, P. Squalenoylation: A generic platform for nanoparticular drug delivery. J. Control. Release 2012, 161, 609–618. [Google Scholar] [CrossRef] [PubMed]
- Valetti, S.; Maione, F.; Mura, S.; Stella, B.; Desmaële, D.; Noiray, M.; Vergnaud, J.; Vauthier, C.; Cattel, L.; Giraudo, E.; et al. Peptide-functionalized nanoparticles for selective targeting of pancreatic tumor. J. Control. Release 2014, 192, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Valetti, S.; Mura, S.; Noiray, M.; Arpicco, S.; Dosio, F.; Vergnaud, J.; Desmaële, D.; Stella, B.; Couvreur, P. Peptide Conjugation: Before or After Nanoparticle Formation? Bioconjug. Chem. 2014, 25, 1971–1983. [Google Scholar] [CrossRef]
- Chirio, D.; Peira, E.; Sapino, S.; Chindamo, G.; Oliaro-Bosso, S.; Adinolfi, S.; Dianzani, C.; Baratta, F.; Gallarate, M. A new bevacizumab carrier for intravitreal administration: Focus on stability. Pharmaceutics 2021, 13, 560. [Google Scholar] [CrossRef] [PubMed]



| Ligand | Nanosystem | Conjugation Strategy | Drug/Payload | Receptor | Cancer Type | Ref. |
|---|---|---|---|---|---|---|
| Small molecules | ||||||
| FA | Polymeric NPs (CHIT) | amide bond | MTX | FR | cervical adenocarcinoma | [52] |
| FA | Polymerosomes | amide bond | ds-DNA and ds-siRNA | FR | cervical carcinoma, breast adenocarcinoma | [53,54] |
| FA | Polymeric NPs (DEX) | ester bond | DOX | FR | breast adenocarcinoma breast | [55] |
| FA | Polymeric NPs (PCL) | amide bond | fluorescent probe (Nile red) | FR | cervical carcinoma, lung carcinoma | [56,57,58] |
| biotin | Micelles | ester bond | niclosamide | biotin receptor | lung carcinoma | [68] |
| biotin | Micelles | ester bond | verteporfin | biotin receptor | prostate cancer, breast adenocarcinoma | [69] |
| TSPO ligands | Polymeric NPs (PLGA) | ester bond | 5-FU | TSPO | glioma | [75] |
| TSPO ligands | G(4)-PAMAM dendrimers | amide bond | fluorescent probe (fluorescein) | TSPO | glioma | [76] |
| lactobionic acid | G(4)-PAMAM dendrimers | amide bond | sorafenib | asialoglycoprotein receptor | liver cancer | [77] |
| alendronate | Polymeric NPs (PLGA) | amide bond | DOX | bone hydroxyapatite | bone cancer | [78] |
| alendronate | PEG dendrimers | amide bond | PTX | bone hydroxyapatite | prostate cancer | [80] |
| FA | SVAs | covalent bond (FA conjugated polyasparthydrazide) | GEM | FR | breast cancer | [129] |
| FA | Liposomes | covalent bond (FA-DSPE conjugate post insertion) | DOX | FR | cervical carcinoma | [130] |
| FA | Liposomes | covalent bond (FA-DSPE conjugate in mixture and post insertion) | a synthetic DOX conjugated with nitric oxide (NO)-releasing group | FR | breast cancer | [131] |
| Peptides and proteins | ||||||
| GNQWFI peptide | Polymeric NPs (PCL) | amide bond | DTX | VEGFR1 | breast adenocarcinoma | [70] |
| GE11 peptide | Polymeric NPs (PLGA) | amide bond | CUR | EGFR | colon adenocarcinoma | [73] |
| GE11 peptide | Polymeric NPs (PLGA) | amide bond | model drug (myoglobin) | EGFR | lung carcinoma | [74] |
| LinTT1 peptide | Liposomes | covalent bond on the surface of preformed liposomes | DOX and sorafenib | p32 protein | breast cancer | [91] |
| NGR motif-containing peptide | Liposomes | covalent bond on the surface of preformed liposomes | BTZ | aminopeptidase N-positive tumor vessels | neuroblastoma | [93] |
| NGR motif-containing peptide | Liposomes | covalent bond on the surface of preformed liposomes | Fenretinide | aminopeptidase N-positive tumor vessels | neuroblastoma | [94] |
| BN(7–14) peptide | Liposomes | amphiphilic peptide derivative mixed with the other lipids | DOX and 111InCl3 | BN receptors | prostate adenocarcinoma | [95] |
| BN-AA1 | Liposomes | amphiphilic peptide derivative mixed with the other lipids | DOX | BN antagonist | prostate cancer | [96] |
| Peptide R | Liposomes | covalent bond on the surface of preformed liposomes | DOX | CXCR4 receptor-antagonist | melanoma lung metastasis | [98] |
| UT-II | Liposomes | covalent bond on the surface of preformed liposomes | DOX | UT-receptor | colon, bladder and prostate cancer | [100] |
| Tf | Liposomes | inserted on the surface of preformed liposome | artemisinin | TfR | colon cancer | [121] |
| Tf | hybrid self-assembling NPs | pre-formed cationic liposomes mixed with Tf | ZOL | TfR | glioblastoma | [124,125] |
| Tf | SNALPs | covalent bond on the preformed NC | miR-34a | TfR | mieloma | [126] |
| Tf | Niosomes | covalent bond on the external surface of the niosomes | DOX | TfR | breast cancer | [127] |
| Tf and FA | Niosomes | covalent bond on the external surface of the niosomes | DOX and CURC or DOX and quercetin | TfR | breast cancer | [128] |
| CPE (Arg4-DAG) | Liposomes | association to the liposome bilayer | model drugs (calcein or BSA) | cervical cancer | [139] | |
| CPE (Arg4-DAG) | Liposomes | association to the liposome bilayer | DOX | breast cancer | [140] | |
| Lf | Liposomes | covalent bond on the surface of preformed liposomes | T3 | multiple receptors | hepatocellular carcinoma | [142] |
| TSH | Nanoliposomes | covalent bond | GEM | TSHR | thyroid carcinoma | [144] |
| ApoE | SLNs | electrostatic interaction | MTX derivative | VLDL | glioblastoma multiforme | [150] |
| Tf/INS | SLNs | covalent bond | MTX derivative | TfR/HIR | glioblastoma multiforme | [151] |
| CKAAKN peptide | SQ NPs | thioether bond | GEM | frizzled receptors | pancreatic cancer | [153] |
| CKAAKN peptide | SQ NPs | thioether bond | GEM | frizzled receptors | pancreatic cancer | [154] |
| Antibodies | ||||||
| rituximab | Polymeric NPs (PLGA) | amide bond | Nutlin-3 | CD20 | chronic lymphocytic leukemia | [71] |
| rituximab | Polymeric NBs (CHIT) | reductive amination | antagomir17 | CD20 | Burkitt limphoma | [86] |
| Anti-CD11c or anti-CD1a monoclonal antibody | Polymeric NBs (CHIT) | reductive amination | plasmid coding for HER2 | murine DC marker CD11c or human DC marker CD1a | breast cancer | [87] |
| anti-Syndecan-1 antibody | Polymeric NPs (PLGA) | amide bond | Nutlin-3 | CD-138/Syndecan-1 | lymphoma | [72] |
| mAb 3F8 | Liposomes | covalent bond on the surface of preformed liposomes | etoposide | anti-GD2 | neuroblastoma, cervical carcinoma, breast carcinoma, melanoma, osteosarcoma | [134] |
| Ab against Frizzled 10 (FZD10) protein | Liposomes | covalent bond on the surface of preformed liposomes | 5-FU | anti-Frizzled 10 protein (FZD10) | colorectal cancer | [135] |
| Hyaluronic acid | ||||||
| HA (HMW)+50:56 | Polymeric NPs (CHIT) | ionotropic gelation | everolimus | CD44 | human mesenchymal stem cells (hMSCs) | [59,60] |
| HA (LMW) | Micelle-like albumin NPs | amide bond | DOX | CD44 | breast adenocarcinoma | [61] |
| HA (HMW) | Polymeric NPs (PLGA) | physical adsorption | sclareol | CD44 | breast cancer | [62] |
| HA (LMW) | Polymeric NPs (PLGA) | layer-by-layer deposition | DTX | CD44 | lung cancer | [63] |
| HA (LMW) | Polymeric NPs (PLGA) | layer-by-layer deposition | 5-FU/plasmid pL3 | CD44 | colon cancer | [64] |
| HA (LMW) | Polymeric NPs (PLGA) | layer-by-layer deposition | DTX/photosensitizer (disulfonate tetraphenyl chlorin) | CD44 | breast cancer and cervix cancer | [65,66] |
| HA (LMW) | Polymeric NPs (PLGA) | layer-by-layer deposition | DTX/photosensitizer (tetrasodium-meso-tetra- (4-sulfonatophenyl)porphyrin) | CD44 | breast cancer | [67] |
| HA (LMW) | Liposomes | HA-phospholpid conjugate inserted in liposome | GEM lipophilic prodrug | CD44 | pancreatic cancer | [109,110] |
| HA (LMW) | Liposomes | HA-phospholpid conjugate inserted in liposome | diethyldithiocarbamate-copper | CD44 | pancreatic cancer | [111] |
| HA (LMW) | Liposomes | HA-phospholpid conjugate inserted in liposome | synthetic DOX conjugated with a H2S-releasing moiety | CD44 | osteosarcoma | [112] |
| HA oligosaccharides | Liposomes | conjugate inserted in liposome | fluorescently labeled | CD44 | lung cancer | [113] |
| HA (HMW) | Lipoplexes | HA-phospholpid conjugate inserted during lipoplexes formation | plasmid DNA | CD44 | breast cancer | [116] |
| HA (HMW) | Lipoplexes | HA-phospholipid conjugate inserted during lipoplexes formation | plasmid DNA | CD44 | lung cancer | [117] |
| HA (HMW) | Lipoplexes | HA-phospholipid conjugate inserted during lipoplexes formation | anti-telomerase siRNA | CD44 | lung cancer | [118] |
| HA (HMW) | Lipoplexes | HA-phospholipid conjugate inserted during lipoplexes formation | anti-Luc siRNA | CD44 | metastatic lung cancer | [119] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Argenziano, M.; Arpicco, S.; Brusa, P.; Cavalli, R.; Chirio, D.; Dosio, F.; Gallarate, M.; Peira, E.; Stella, B.; Ugazio, E. Developing Actively Targeted Nanoparticles to Fight Cancer: Focus on Italian Research. Pharmaceutics 2021, 13, 1538. https://doi.org/10.3390/pharmaceutics13101538
Argenziano M, Arpicco S, Brusa P, Cavalli R, Chirio D, Dosio F, Gallarate M, Peira E, Stella B, Ugazio E. Developing Actively Targeted Nanoparticles to Fight Cancer: Focus on Italian Research. Pharmaceutics. 2021; 13(10):1538. https://doi.org/10.3390/pharmaceutics13101538
Chicago/Turabian StyleArgenziano, Monica, Silvia Arpicco, Paola Brusa, Roberta Cavalli, Daniela Chirio, Franco Dosio, Marina Gallarate, Elena Peira, Barbara Stella, and Elena Ugazio. 2021. "Developing Actively Targeted Nanoparticles to Fight Cancer: Focus on Italian Research" Pharmaceutics 13, no. 10: 1538. https://doi.org/10.3390/pharmaceutics13101538