Abstract
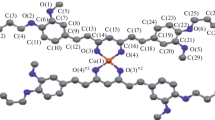
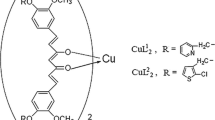
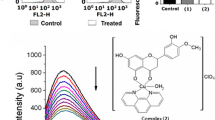
Co(II) complexes having a ferrocene-based curcuminoid (Fc-curH) ligand viz. [Co(L)2(Fc-cur)]ClO4 (1, 2), where L is phenanthroline base, namely, 1,10-phenanthroline (phen in 1) and dipyrido[3,2-a:2’,3’-c]phenazine (dppz in 2) have been synthesized, characterized and evaluated as photochemotherapeutic agents in vitro. The corresponding Co(II) complexes of the naturally occurring polyphenol curcumin (curH), namely, [Co(L)2(cur)]ClO4 (3, 4), where L is phen (in 3) and dppz (in 4) were synthesized and their photo-induced anticancer activities compared with their ferrocene containing counterparts 1 and 2. The Co(II) acetylacetonato complex viz. [Co(phen)2(acac)]ClO4 (5) was structurally characterized through X-ray crystallography and used as control for cellular experiments. The Co(II) complexes having ferrocene-based curcuminoid are remarkably stable at physiological condition with higher lipophilicity compared to their curcumin analogues. The complexes display significant binding propensity to calf thymus (ct) DNA and human serum albumin (HSA). The complexes 1–4 display remarkable visible light induced cytotoxicity with the ferrocenyl analogues showing more phototoxic index (PI). The Co(II) curcumin complexes localize in the nucleus and mitochondria of A549 cells. The primary cell death mechanism is believed to be apoptotic in nature induced by light assisted generation of reactive oxygen species (ROS).
Graphic abstract








Similar content being viewed by others
Change history
25 October 2021
A Correction to this paper has been published: https://doi.org/10.1007/s00775-021-01907-2
References
Johnstone TC, Suntharalingam K, Lippard SJ (2016) Chem Rev 116:3436–3486
Kelland L (2007) Nat Rev Cancer 7:573–584
Gibson D (2009) Dalton Trans 10681–10689
Oun R, Moussa YE, Wheate NJ (2018) Dalton Trans 47:6645–6653
Galluzzi L, Senovilla L, Vitale I, Michels J, Martins I, Kepp O, Castedo M, Kroemer G (2012) Oncogene 31:1869–1883
Kwiatkowski S, Knap B, Przystupski D, Saczko J, Kędzierska E, Knap-Czop K, Kotlińska J, Michel O, Kotowski K, Kulbacka J (2018) Biomed Pharmacother 106:1098–1107
Allison RR, Moghissi K (2013) Clin Endosc 46:24–29
Zhang J, Jiang C, Longo JPF, Azevedo RB, Zhang H, Muehlmann LA (2018) Acta Pharm Sin B 8:137–146
Kou J, Dou D, Yang L (2017) Oncotarget 8:81591–81603
O’Connor AE, Gallagher WM, Byrne AT (2009) Photochem Photobiol 85:1053–1074
Moriwaki SI, Misawa J, Yoshinari Y, Yamada I, Takigawa M, Tokura Y (2001) Photodermatol Photoimmunol Photomed 17:241–243
Wang X, Wang X, Jin S, Muhammad N, Guo Z (2019) Chem Rev 119:1138–1192
Monro S, Colón KL, Yin H, Roque J, Konda P, Gujar S, Thummel RP, Lilge L, Cameron CG, McFarland SA (2019) Chem Rev 119:797–828
Reeßing F, Szymanski W (2017) Curr Med Chem 24:4905–4950
Liu J, Zhang C, Rees TW, Ke L, Ji L, Chao H (2018) Coord Chem Rev 363:17–28
Heinemann F, Karges J, Gasser G (2017) Acc Chem Res 50:2727–2736
Pandey V, Raza MK Joshi P, Gupta I (2020) J Org Chem 85:6309-6322
Imberti C, Zhang P, Huang H, Sadler PJ (2020) Angew Chem Int Ed 59:61–73
Fernandez-Moreira V, Gimeno MC (2018) Chem Eur J 24:3345–3353
Ramu V, Gautam S, Kondaiah P, Chakravarty AR (2019) Inorg Chem 58:9067–9075
Zhang P, Sadler PJ (2017) J Organomet Chem 839:5–14
Gasser G, Ott I, Metzler-Nolte N (2011) J Med Chem 54:3–25
Jaouen G, Vessièresab A, Top S (2015) Chem Soc Rev 44:8802–8817
Bruyère C, Mathieu V, Vessières A, Pigeon P, Top S, Jaouen G, Kiss R (2014) J Inorg Biochem 141:144–151
Patra M, Gasser G (2017) Nat Rev Chem 1:0066
Meier-Menches SM, Gerner C, Berger W, Hartinger CG, Keppler BK (2018) Chem Soc Rev 47:909–928
Lazic S, Kaspler P, Shi G, Monro S, Sainuddin T, Forward S, Kasimova K, Hennigar R, Mandel A, McFarland S, Lilge L (2017) Photochem Photobiol 93:1248–1258
Zamora A, Vigueras G, Rodríguez V, Santana MD, Ruiz J (2018) Coord Chem Rev 360:34–76
Lopresti AL (2018) Adv Nutr 9:41–50
Nelson KM, Dahlin JL, Bisson J, Graham J, Pauli GF, Walters MA (2017) J Med Chem 60:1620–1637
Musib D, Pal M, Raza MK, Roy M (2020) Dalton Trans 49:10786–10798
Ferrari E, Pignedoli F, Imbriano C, Marverti G, Basile V, Venturi E, Saladini M (2011) J Med Chem 54:8066–8077
Khatun B, Baishya P, Ramteke A, Maji TK (2020) New J Chem 44:4887–4897
Arezki A, Chabot GG, Quentin L, Scherman D, Jaouen G, Brulé E (2011) Med Chem Commun 2:190–195
Banerjee S, Chakravarty AR (2015) Acc Chem Res 48:2075–2083
Pröhl M, Schubert US, Weigand W, Gottschaldt M (2016) Coord Chem Rev 307:32–41
Wanninger S, Lorenz V, Subhan A, Edelmann FT (2015) Chem Soc Rev 44:4986–5002
Das D, Banaspati A, Das N, Bora B, Raza MK, Goswami TK (2019) Dalton Trans 48:12933–12942
Handler N, Jaeger W, Puschacher H, Leisser K, Erker T (2007) Chem Pharm Bull 55:64–71
Amouyal E, Homsi A, Chambron J-C, Sauvage J-P (1990) J Chem Soc Dalton Trans 1841–1845
SADABS, Bruker AXS Inc., Madison, Wisconsin, USA (2012).
Sheldrick GM (2015) Acta Cryst C 71:3–8
Mcghee JD, Hippel PHV (1974) J Mol Biol 86:469–489
Carter MT, Rodriguez M, Bard AJ (1989) J Am Chem Soc 111:8901–8911
Banaspati A, Das D, Choudhury CJ, Bhattacharyya A, Goswami TK (2019) J Inorg Biochem 191:60–68
Banaspati A, Raza MK, Goswami TK (2020) Eur J Med Chem 204:112632
Das D, Raza MK, Goswami TK (2020) Polyhedron 186:114614
Raza MK, Gautam S, Howlader P, Bhattacharyya A, Kondaiah P, Chakravarty AR (2018) Inorg Chem 57:14374–14385
Raza MK, Mitra K, Shettar A, Basu U, Kondaiah P, Chakravarty AR (2016) Dalton Trans 45:13234–13243
Upadhyay A, Gautam S, Ramu V, Kondaiah P, Chakravarty AR (2019) Dalton Trans 48:17556–17565
Mitra K, Gautam S, Kondaiah P, Chakravarty AR (2015) Angew Chemie-Int Ed 54:13989–13993
Dougan SJ, Habtemariam A, McHale SE, Parsons S, Sadler PJ (2008) PNAS 105:11628–11633
Romero-Canelón I, Mos M, Sadler PJ (2015) J Med Chem 58:7874–7880
Goswami TK, Gadadhar S, Roy M, Nethaji M, Karande AA, Chakravarty AR (2012) Organometallics 31:3010–3021
Goswami TK, Gadadhar S, Gole B, Karande AA, Chakravarty AR (2012) Eur J Med Chem 63:800–810
Maity B, Roy M, Banik B, Majumdar R, Dighe RR, Chakravarty AR (2010) Organometallics 29:3632–3641
Kellett A, Molphy Z, Slator C, McKee V, Farrell NP (2019) Chem Soc Rev 48:971–988
Zhang Y, Sun T, Jiang C (2018) Acta Pharm Sin B 8:34–50
İnci D, Aydın R, Vatan Ö, Sevgi T, Yılmaz D, Zorlu Y, Yerli Y, Çoşut B, Demirkan E, Çinkılıç N (2017) J Biol Inorg Chem 22:61–85
Ammar AA, Raveendran R, Gibson D, Nassar T, Benita S (2016) J Med Chem 59:9035–9046
Banerjee S, Prasad P, Hussain A, Khan I, Kondaiah P, Chakravarty AR (2012) Chem Commun 48:7702–7704
Delaey E, Van Larr F, De Vos D, Kamuhabwa A, Jacobs P, De Witte P (2000) J Photochem Photobiol B 55:27–36
Wu L, Yang L, Huang J, Zhang L, Weng X, Zhang X, Shen C, Zhou X, Zheng C (2009) Chem Biodivers 6:1066–1076
Sarkar T, Banerjee S, Hussain A (2015) RSC Adv 5:16641–16653
Mjos KD, Orvig C (2014) Chem Rev 114:4540–4563
Qin J-L, Shen W-Y, Chen Z-F, Zhao L-F, Qin Q-P, Yu Y-C, Liang H (2017) Sci Rep 7(46056):1–18
Abyar S, Khandar AA, Salehi R, Hosseini-Yazdi SA, Alizadeh E, Mahkam M, Jamalpoor A, White JM, Shojaei M, Aizpurua-olaizola O, Masereeuw R, Janssen MJ (2019) Sci Rep 9(14686):1–11
Acknowledgements
We are greatful to Department of Science and Technology (DST) INSPIRE Program, Government of India for financial support (IFA14-CH-164). The authors thank DST and Department of SAIF, Gauhati University for a CCD difractometer facility. We are thankful to Dr. Ranjit Thakuria for his kind help in X-ray crystallographic procedures. The authors sincerely thank Prof. A. R. Chakravarty, Indian Institute of Science, Bangalore for allowing us to use his cell culture facility.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised due to correction in figure 6
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Das, D., Noor, A., Raza, M.K. et al. Co(II) complexes of curcumin and a ferrocene-based curcuminoid: a study on photo-induced antitumor activity. J Biol Inorg Chem 26, 881–893 (2021). https://doi.org/10.1007/s00775-021-01899-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-021-01899-z