Abstract
Six artesunate (ART) conjugated ruthenium(II) complexes (Ru(II)-ART conjugates) with the formula [Ru(N^N)2bpy(4-CH3-4′-CH2OART)](PF6)2 (Ru-ART-1-3) and [Ru(N^N)2bpy(4-CH2OART-4′-CH2OART)](PF6)2 (Ru-ART-4–6) (N^N = 2,2′-bipyridine (bpy, in Ru-ART-1 and Ru-ART-4), 1,10-phenanthroline (phen, in Ru-ART-2 and Ru-ART-5) and 4,7-diphenyl-1,10-phenanthroline (DIP, in Ru-ART-3 and Ru-ART-6)), were synthesized and characterized. Among them, Ru-ART-1-3 and Ru-ART-4-6 carry one and two ART moieties, respectively. Ru-ART-3 and Ru-ART-6 exhibit better cytotoxicity among six Ru(II)-ART conjugates. These two complexes can be effectively taken up by human cervical carcinoma (HeLa) cells. In addition, they selectively kill cancer cell lines while mildly affect normal cells. Mechanism studies have shown that HeLa cells treated with Ru-ART-3 and Ru-ART-6 show typical apoptotic characteristics (morphology changes, mitochondrial dysfunction, caspase cascade, etc.). On the other hand, the up regulation of Beclin-1 and conversion of LC3-I to LC3-II note the appearance of autophagy. As a result, Ru-ART-3 and Ru-ART-6 induce autophagy-dependent cell apoptosis via mitochondrial dysfunction and reactive oxygen species (ROS) accumulation.
Graphic abstract

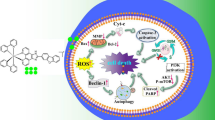
In this work, six artesunate (ART) conjugated ruthenium(II) complexes (Ru(II)-ART conjugates) have been synthesized and characterized. Among them, Ru-ART-3 and Ru-ART-6 exhibit better cytotoxicity. Mechanism studies have shown that HeLa cells treated with Ru-ART-3 and Ru-ART-6 show typical apoptotic characteristics (morphology changes, mitochondrial dysfunction, caspase cascade, etc.). On the other hand, the up regulation of Beclin-1 and conversion of LC3-I to LC3-II note the appearance of autophagy










Similar content being viewed by others
References
Velozo-Sá VS, Pereira LR, Lima AP, Mello-Andrade F, Rezende MRM, Goveia RM, Pires WC, Silva MM, Oliveira KM, Ferreira AG, Ellena J, Deflon VM, Grisolia CK, Batista AA, Silveira-Lacerda EP (2019) Dalton Trans 48:6026–6039
Adeyemo AA, Shettar A, Bhat IA, Kondaiah P, Mukherjee PS (2018) Dalton Trans 47:8466–8475
Zhao J, Zhang D, Hua W, Li W, Xu G, Gou S (2018) Organometallics 37:441–447
Thota S, Rodrigues DA, Crans DC, Barreiro EJ (2018) J Med Chem 61:5805–5821
Flocke LS, Trondl R, Jakupec MA, Keppler BK (2016) Invest New Drugs 34:261–268
Alessio E, Messori L (2019) Molecules 24:10
Trondl R, Heffeter P, Kowol CR, Jakupec MA, Bergerbd W, Keppler BK (2014) Chem Sci 5:2925–2932
Alessio E, Mestroni G, Bergamo A, Sava G (2004) Curr Top Med Chem 4:1525–1535
Alessio E, Mestroni G, Bergamo A, Sava G (2004) Met Ions Biol Syst 42:323–351
Wernitznig D, Kiakos K, Del Favero G, Harrer N, Machat H, Osswald A, Jakupec MA, Wernitznig A, Sommegruber W, Keppler BK (2019) Metallomics 11:1044–1048
Wernitznig D, Meier-Menches SM, Cseh K, Theiner S, Wenisch D, Schweikert A, Jakupec MA, Koellensperger G, Wernitznig A, Sommergruber W, Keppler BK (2020) Metallomics 12:2121–2133
Xie L, Wang L, Guan R, Ji L, Chao H (2021) J Inorg Biochem 217:111380
Luo Z, Yu L, Yang F, Zhao Z, Yu B, Lai H, Wong KH, Ngai SM, Zheng W, Chen T (2014) Metallomics 6:1480–1490
Ye RR, Ke ZF, Tan CP, He L, Ji LN, Mao ZW (2013) Chem Eur J 19:10160–10169
Chou TC (2010) Cancer Res 70:440–446
Cheng Q, Shi H, Wang H, Min Y, Wang J, Liu Y (2014) Chem Commun 50:7427–7430
Lin K, Rong Y, Chen D, Zhao Z, Bo H, Qiao A, Hao X, Wang J (2020) Front Oncol 10:141
Efferth T (2017) Semin Cancer Biol 46:65–83
Feng FB, Qiu HY (2018) Biomed Pharmacother 102:1209–1220
Zhou X, Chen Y, Wang F, Wu H, Zhang Y, Liu J, Cai Y, Huang S, He N, Hu Z, Jin X (2020) Chem Biol Interact 331:109273
Yang ND, Tan SH, Ng S, Shi Y, Zhou J, Tan KS, Wong WS, Shen HM (2014) J Biol Chem 289:33425–33441
Zhang S, Yuan H, Guo Y, Wang K, Wang X, Guo Z (2018) Chem Commun 54:11717–11720
Ye RR, Peng W, Chen BC, Jiang N, Chen XQ, Mao ZW, Li RT (2020) Metallomics 12:1131–1141
Elmore S (2007) Toxicol Pathol 35:495–516
Pfeffer CM, Singh ATK (2018) Int J Mol Sci 19:448
Johnstone RW, Ruefli AA, Lowe SW (2002) Cell 108:153–164
Dower CM, Wills CA, Frisch SM, Wang HG (2018) Autophagy 14:1110–1128
Yang ZJ, Chee CE, Huang S, Sinicrope FA (2011) Mol Cancer Ther 10:1533–1541
Singh N, Jang S, Jo JH, Kim DH, Park DW, Kim I, Kim H, Kang SC, Chi KW (2016) Chem Eur J 22:16157–16164
Chen S, Zhou L, Zhang Y, Leng Y, Pei XY, Lin H, Jones R, Orlowski RZ, Dai Y, Grant S (2014) Mol Cell Biol 34:3435–3449
Zhang P, Zheng Z, Ling L, Yang X, Zhang N, Wang X, Hu M, Xia Y, Ma Y, Yang H, Wang Y, Liu H (2017) Autophagy 13:1093–1112
Koceva-Chyla A, Matczak K, Hikisz MP, Durka MK, Kochel MK, Suss-Fink G, Furrer J, Kowalski K (2016) ChemMedChem 11:2171–2187
Lenis-Rojas OA, Fernandes AR, Roma-Rodrigues C, Baptista PV, Marques F, Perez-Fernandez D, Guerra-Varela J, Sanchez L, Vazquez-Garcia D, Torres ML, Fernandez A, Fernandez JJ (2016) Dalton Trans 45:19127–19140
Wan D, Lai SH, Zeng CC, Zhang C, Tang B, Liu YJ (2017) J Inorg Biochem 173:1–11
Sullivan BP, Salmon DJ, Meyer TJ (2002) Inorg Chem 17:3334–3341
Gopinath J, Park KH, Kim S-J, Santosh V, Sainath AVS, Dhayal M (2017) J Mater Sci 52:10545–10556
Clede S, Lambert F, Saint-Fort R, Plamont MA, Bertrand H, Vessieres A, Policar C (2014) Chem Eur J 20:8714–8722
Lameijer LN, Breve TG, van Rixel VHS, Askes SHC, Siegler MA, Bonnet S (2018) Chem Eur J 24:2709–2717
McKeage MJ, Berners-Price SJ, Galettis P, Bowen RJ, Brouwer W, Ding L, Zhuang L, Baguley BC (2000) Cancer Chemother Pharmacol 46:343–350
Lv G, Guo L, Qiu L, Yang H, Wang T, Liu H, Lin J (2015) Dalton Trans 44:7324–7331
Delasoie J, Pavic A, Voutier N, Vojnovic S, Crochet A, Nikodinovic-Runic J, Zobi F (2020) Eur J Med Chem 204:112583
Yang YL, Guo LH, Tian ZZ, Gong YT, Liu Z (2018) Inorg Chem 17:11087–11098
Sathyadevi P, Krishnamoorthy P, Bhuvanesh NSP, Kalaiselvi P, Dharmaraj N (2021) Chem Eur J 55:420–431
Maniti O, Piao HR, Ayala-Sanmartin J (2014) Int J Biochem Cell Biol 50:73–81
Wallace DC (2012) Nat Rev Cancer 12:685–698
DeHart DN, Fang D, Heslop K, Li L, Lemasters JJ, Maldonado EN (2018) Biochem Pharmacol 148:155–162
Martins WK, Santos NF, Rocha CS, Bacellar IOL, Tsubone TM, Viotto AC, Matsukuma AY, Abrantes ABP, Siani P, Dias LG, Baptista MS (2019) Autophagy 15:259–279
Venkatesan R, Subedi L, Yeo EJ, Kim SY (2016) Neurochem Int 99:133–146
Garedew A, Henderson SO, Moncada S (2010) Cell Death Differ 17:1540–1550
Zorov DB, Juhaszova M, Sollott SJ (2014) Physiol Rev 94:909–950
Luptak I, Qin F, Sverdlov AL, Pimentel DR, Panagia M, Croteau D, Siwik DA, Bachschmid MM, He H, Balschi JA, Colucci WS (2019) Antioxid Redox Signal 31:539–549
Dong K, Lei Q, Guo R, Wu X, Zhang Y, Cui N, Shi JY, Lu T (2019) Int J Nanomed 14:5713–5728
Thornberry NA, Lazebnik Y (1998) Science 281:1312–1316
Hanahan D, Weinberg RA (2011) Cell 144:646–674
Kaur S, Changotra H (2020) Biochimie 175:34–49
Wang AL, Boulton ME, Dunn WA Jr, Rao HV, Cai J, Lukas TJ, Neufeld AH (2009) Autophagy 5:1190–1193
Lamark T, Svenning S, Johansen T (2017) Essays Biochem 61:609–624
Acknowledgements
This work was supported by National Natural Science Foundation of China (21967014, 22007042), Applied Basic Research Projects of Yunnan Province (202001AT070036), the Innovative Team of Yunnan Province (No. 2019HC018), High-level Scientific Research Foundation for Talent Introduction of Kunming University of Science and Technology (KKKP201826008).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, BC., Lu, JJ., Jiang, N. et al. Synthesis, characterization and antitumor mechanism investigation of ruthenium(II) polypyridyl complexes with artesunate moiety. J Biol Inorg Chem 26, 909–918 (2021). https://doi.org/10.1007/s00775-021-01901-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-021-01901-8