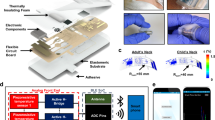
Abstract
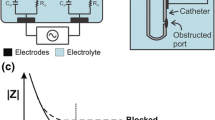
External ventricular drains (EVDs) are used clinically to relieve excess fluid pressure in the brain. However, EVD outflow rate is highly variable and typical clinical flow tracking methods are manual and low resolution. To address this problem, we present an integrated multi-sensor module (IMSM) containing flow, temperature, and electrode/substrate integrity sensors to monitor the flow dynamics of cerebrospinal fluid (CSF) drainage through an EVD. The impedimetric sensors were microfabricated out of biocompatible polymer thin films, enabling seamless integration with the fluid drainage path due to their low profile. A custom measurement circuit enabled automated and portable sensor operation and data collection in the clinic. System performance was verified using real human CSF in a benchtop EVD model. Impedimetric flow sensors tracked flow rate through ambient temperature variation and biomimetic pulsatile flow, reducing error compared with previous work by a factor of 6.6. Detection of sensor breakdown using novel substrate and electrode integrity sensors was verified through soak testing and immersion in bovine serum albumin (BSA). Finally, the IMSM and measurement circuit were tested for 53 days with an RMS error of 61.4 μL/min.






source beaker to enable automatic testing. For calibration, the IMSM was placed inside the oven







Similar content being viewed by others
References
A.A. Al-Ali, B.J. Maundy, A.S. Elwakil, Design and Implementation of Portable Impedance Analyzers (Springer International Publishing, Cham, Switzerland, 2019)
Al-Ali, Abdulwadood, Ahmed Elwakil, Abdelaziz Ahmad, and Brent Maundy. 2017. “Design of a Portable Low-Cost Impedance Analyzer:” In Proceedings of the 10th International Joint Conference on Biomedical Engineering Systems and Technologies, 104–9. Porto, Portugal: SCITEPRESS. https://doi.org/10.5220/0006121901040109.
D.J. Apigo, P.L. Bartholomew, T. Russell, A. Kanwal, R.C. Farrow, G.A. Thomas, An angstrom-sensitive, differential mems capacitor for monitoring the milliliter dynamics of fluids. Sens. Actuators, A 251(November), 234–240 (2016). https://doi.org/10.1016/j.sna.2016.10.030
D.J. Apigo, P.L. Bartholomew, T. Russell, A. Kanwal, R.C. Farrow, G. Thomas, Evidence of an application of a variable MEMS capacitive sensor for detecting shunt occlusions. Sci. Rep. (2017). https://doi.org/10.1038/srep46039
A. Baldwin, Yu. Lawrence, E. Meng, An electrochemical impedance-based thermal flow sensor for physiological fluids. J. Microelectromech. Syst. 25(6), 1015–1024 (2016). https://doi.org/10.1109/JMEMS.2016.2614664
A. Baldwin, E. Yoon, T. Hudson, E. Meng, Fluid temperature measurement in aqueous solution via electrochemical impedance. J. Microelectromech. Syst. 28(6), 1060–1067 (2019). https://doi.org/10.1109/JMEMS.2019.2939811
D. Bigio, Biological reactions to cerebrospinal fluid shunt devices: a review of the cellular pathology. Neurosurgery 42(2), 319–326 (1998). https://doi.org/10.1097/00006123-199802000-00064
T.L. Bonfield, E. Colton, J.M. Anderson, Plasma protein adsorbed biomedical polymers: activation of human monocytes and induction of interleukin 1. J. Biomed. Mater. Res. 23(6), 535–548 (1989). https://doi.org/10.1002/jbm.820230602
T.L. Bonfield, E. Colton, J.M. Anderson, protein adsorption of biomedical polymers influences activated monocytes to produce fibroblast stimulating factors. J. Biomed. Mater. Res. 26(4), 457–465 (1992). https://doi.org/10.1002/jbm.820260404
T. Bork, A. Hogg, M. Lempen, D. Müller, D. Joss, T. Bardyn, P. Büchler et al., Development and in-vitro characterization of an implantable flow sensing transducer for hydrocephalus. Biomed. Microdevice 12(4), 607–618 (2010). https://doi.org/10.1007/s10544-010-9413-6
T.M. Clark, S.C. Malpas, D. McCormick, P. Heppner, D.M. Budgett, Implantable multi-modal sensor to improve outcomes in hydrocephalus management. IEEE Sens. J. 15(10), 6027–6035 (2015). https://doi.org/10.1109/JSEN.2015.2451655
N.P. Cosman, S.G. Roscoe, Electrochemical quartz crystal nanobalance (EQCN) studies of protein interfacial behavior at Pt. Langmuir 20(5), 1711–1720 (2004). https://doi.org/10.1021/la035154h
C.N. Cottonaro, H.V. Roohk, G. Shimizu, D.R. Sperling, Quantitation and characterization of competitive protein binding to polymers. ASAIO J. 27(1), 391 (1981)
Cvancara, P., S. Lauser, L. Rudmann, and T. Stieglitz. 2016. “Investigations on Different Epoxies for Electrical Insulation of Microflex Structures.” In 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), 1963–66. Orlando, FL, USA: IEEE. https://doi.org/10.1109/EMBC.2016.7591108.
M. Czosnyka, J.D. Pickard, Monitoring and interpretation of intracranial pressure. J. Neurol. Neurosurg. Psychiatry 75(6), 813–821 (2004). https://doi.org/10.1136/jnnp.2003.033126
Analog Devices. 2005. “AD5933 Data Sheet.” https://www.analog.com/media/en/technical-documentation/data-sheets/AD5933.pdf.
J.M. Drake, C. Sainte-Rose, M. DaSilva, H. Jean-François, Cerebrospinal fluid flow dynamics in children with external ventricular drains. Neurosurgery 28(2), 242–250 (1991). https://doi.org/10.1227/00006123-199102000-00011
E.T. Enikov, G. Édes, J. Skoch, R. Anton, Application of GMR sensors to liquid flow sensing. J. Microelectromech. Syst. 24(4), 914–921 (2015). https://doi.org/10.1109/JMEMS.2014.2359174
P.A. Gardner, J. Engh, D. Atteberry, J.J. Moossy, Hemorrhage rates after external ventricular drain placement: clinical article. J. Neurosurg. 110(5), 1021–1025 (2009). https://doi.org/10.3171/2008.9.JNS17661
Á. Giménez, M. Galarza, U. Thomale, M.U. Schuhmann, J. Valero, J.M. Amigó, Pulsatile flow in ventricular catheters for hydrocephalus. Philosophical Transact. Royal Soc. 375(2096), 20160294 (2017). https://doi.org/10.1098/rsta.2016.0294
R. Gomez-Sjoberg, D.T. Morisette, R. Bashir, Impedance Microbiology-on-a-Chip: Microfluidic Bioprocessor for Rapid Detection of Bacterial Metabolism. J. Microelectromech. Syst. 14(4), 829–838 (2005). https://doi.org/10.1109/JMEMS.2005.845444
W.F. Gorham, A new, general synthetic method for the preparation of linear poly-p-Xylylenes. J. Poly. Sci. 4(12), 3027–3039 (1966). https://doi.org/10.1002/pol.1966.150041209
C.A. Gutierrez, C. Lee, B. Kim, E. Meng, 16th International Solid-State Sensors. Actuat. Microsys. Conf (2011). https://doi.org/10.1109/TRANSDUCERS.2011.5969538
H. Hämmerle, K. Kobuch, K. Kohler, W. Nisch, H. Sachs, M. Stelzle, Biostability of micro-photodiode arrays for subretinal implantation. Biomaterials 23(3), 797–804 (2002). https://doi.org/10.1016/S0142-9612(01)00185-5
B.W. Hanak, R.H. Bonow, C.A. Harris, S.R. Browd, Cerebrospinal fluid shunting complications in children. Pediatr. Neurosurg. 52(6), 381–400 (2017). https://doi.org/10.1159/000452840
C.A. Harris, J.H. Resau, E.A. Hudson, R.A. West, C. Moon, J.P. McAllister, Mechanical contributions to astrocyte adhesion using a novel in vitro model of catheter obstruction. Exp. Neurol. 222(2), 204–210 (2010). https://doi.org/10.1016/j.expneurol.2009.12.027
Hudson, Trevor, Alex Baldwin, and Ellis Meng. (2019) “A Continuous, Drift-Compensated Impedimetric Thermal Flow Sensor for in Vivo Applications. In 2019 IEEE 32nd International Conference on Micro Electro Mechanical Systems (MEMS), 731–34. https://doi.org/10.1109/MEMSYS.2019.8870746.
T. Hudson, E. Meng, A continuous, impedimetric parylene flow sensor. J. Microelectromechan. Syst. 30, 456–471 (2021)
C. Kadowaki, M. Hara, M. Numoto, K. Takeuchi, I. Saito, CSF shunt physics: factors influencing inshunt CSF flow. Child’s Nervous System 11(4), 203–206 (1995). https://doi.org/10.1007/BF00277654
A.A. Khan, A. Jabbar, A. Banerjee, G. Hinchley, Cerebrospinal shunt malfunction recognition and emergency management. British J. Hospital Med. 68(12), 651–655 (2007). https://doi.org/10.12968/hmed.2007.68.12.651
B.J. Kim, E. Meng, Micromachining of Parylene C for BioMEMS. Polym. Adv. Technol. 27(5), 564–576 (2016). https://doi.org/10.1002/pat.3729
S. Kim, Yu. Guiduk, T. Kim, K. Shin, J. Yoon, Rapid bacterial detection with an interdigitated array electrode by electrochemical impedance spectroscopy. Electrochim. Acta 82, 126–131 (2012). https://doi.org/10.1016/j.electacta.2012.05.131
B.J. Kim, W. Jin, A. Baldwin, Yu. Lawrence, E. Christian, M.D. Krieger, J. Gordon McComb, E. Meng, Parylene MEMS patency sensor for assessment of hydrocephalus shunt obstruction. Biomed. Microdevice 18(5), 87 (2016). https://doi.org/10.1007/s10544-016-0112-9
A.R. Kirmani, A.H. Sarmast, A.R. Bhat, “Role of External Ventricular Drainage in the Management of Intraventricular Hemorrhage (surgical neurology international, Its Complications and Management, 2015). https://doi.org/10.4103/2152-7806.172533
J. Klener, K. Hofbauerová, A. Bartoš, J. Říčný, D. Řípová, V. Kopecký, Instability of cerebrospinal fluid after delayed storage and repeated freezing: a holistic study by drop coating deposition raman spectroscopy. Clinic. Chem. Lab. Med. (CCLM) 52(5), 657–664 (2014). https://doi.org/10.1515/cclm-2013-0800
S. Krause, M. Hahne, M. Walter, S. Kiefer, K.R. Linke, S. Leonhardt, Brain Pressure Dynamics and Control with an External Ventricular Drainage In 5th European Conference of the International Federation for Medical and Biological Engineering, edited by Ákos Jobbágy. Berlin Heidelberg Berlin, Heidelberg (2011). https://doi.org/10.1007/978-3-642-23508-5_82
S.R. Krishnan, T.R. Ray, A.B. Ayer, Y. Ma, P. Gutruf, KunHyuck Lee, J.Y. Lee et al., Epidermal electronics for noninvasive, wireless, quantitative assessment of ventricular shunt function in patients with hydrocephalus. Sci. Translat. Med. 10(465), 8437 (2018). https://doi.org/10.1126/scitranslmed.aat8437
P. Lackner, R. Beer, G. Broessner, R. Helbok, K. Galiano, C. Pleifer, B. Pfausler et al., Efficacy of silver nanoparticles-impregnated external ventricular drain catheters in patients with acute occlusive hydrocephalus. Neurocrit. Care 8(3), 360–365 (2008). https://doi.org/10.1007/s12028-008-9071-1
D.D. Limbrick, S. Lake, M. Talcott, B. Alexander, S. Wight, J.T. Willie, W.D. Richard, G.M. Genin, E.C. Leuthardt, The baric probe: a novel long-term implantable intracranial pressure monitor with ultrasound-based interrogation: technical note. J. Neurosurg. Pediatr. 10(6), 518–524 (2012). https://doi.org/10.3171/2012.8.PEDS11565
A.A. Linninger, M. Xenos, D.C. Zhu, M.R. Somayaji, S. Kondapalli, R.D. Penn, Cerebrospinal fluid flow in the normal and hydrocephalic human brain. IEEE Trans. Biomed. Eng. 54(2), 291–302 (2007). https://doi.org/10.1109/TBME.2006.886853
B.R. Lutz, P. Venkataraman, S.R. Browd, New and improved ways to treat hydrocephalus: pursuit of a smart shunt. Surg. Neurol. Int. 4(1), S38-50 (2013). https://doi.org/10.4103/2152-7806.109197
J.R. Madsen, T.P. Boyle, M.I. Neuman, E.-H. Park, M.S. Tamber, R.W. Hickey, G.G. Heuer et al., Diagnostic accuracy of non-invasive thermal evaluation of ventriculoperitoneal shunt flow in shunt malfunction: a prospective, multi-site, operator-blinded study. Neurosurgery 87(5), 939–948 (2020). https://doi.org/10.1093/neuros/nyaa128
E. Meng, P.-Y. Li, Y.-C. Tai, Plasma removal of parylene C. J. Micromech. Microeng. 18(4), 045004 (2008). https://doi.org/10.1088/0960-1317/18/4/045004
R.P. Metzen, von, and Thomas Stieglitz. , The effects of annealing on mechanical, chemical, and physical properties and structural stability of parylene C. Biomed. Microdevice 15(5), 727–735 (2013). https://doi.org/10.1007/s10544-013-9758-8
B.J.E. Misgeld, I.M. Elixmann, L. Fahnster, M. Walter, M. Weinzierl, W. Steudel, S. Leonhardt, Design and evaluation of an automatic extraventricular drainage control system. IEEE Trans. Control Syst. Technol. 23(6), 2283–2292 (2015). https://doi.org/10.1109/TCST.2015.2413377
P. Moshayedi, G. Ng, J.C.F. Kwok, G.S.H. Yeo, C.E. Bryant, J.W. Fawcett, K. Franze, J. Guck, The relationship between glial cell mechanosensitivity and foreign body reactions in the central nervous system. Biomaterials 35(13), 3919–3925 (2014). https://doi.org/10.1016/j.biomaterials.2014.01.038
R. Muralidharan, External ventricular drains: management and complications. Surg. Neurol. Int. 6(6), S271–S274 (2015). https://doi.org/10.4103/2152-7806.157620
Nordbotten, Bernt Jørgen. 2008. “Bioimpedance Measurements Using the Integrated Circuit AD5933.” M.S. Thesis, University of Oslo.
S. Omanovic, S.G. Roscoe, Electrochemical studies of the adsorption behavior of bovine serum albumin on stainless steel. Langmuir 15(23), 8315–8321 (1999). https://doi.org/10.1021/la990474f
J. Ortigoza-Diaz, K. Scholten, E. Meng, Characterization and modification of adhesion in dry and wet environments in thin-film parylene systems. J. Microelectromech. Syst. 27(5), 874–885 (2018). https://doi.org/10.1109/JMEMS.2018.2854636
T. Pennell, J.L. Yi, B.A. Kaufman, S. Krishnamurthy, Noninvasive measurement of cerebrospinal fluid flow using an ultrasonic transit time flow sensor: a preliminary study. J. Neurosurg. Pediatr. 17(3), 270–277 (2016). https://doi.org/10.3171/2015.7.PEDS1577
C.P. Pennisi, C. Sevcencu, A. Dolatshahi-Pirouz, M. Foss, J. Lundsgaard Hansen, A. Nylandsted Larsen, V. Zachar, F. Besenbacher, K. Yoshida, Responses of fibroblasts and glial cells to nanostructured platinum surfaces. Nanotechnology 20(38), 385103 (2009). https://doi.org/10.1088/0957-4484/20/38/385103
R.K.R. Phillips, S. Omanovic, S.G. Roscoe, Electrochemical studies of the effect of temperature on the adsorption of yeast alcohol dehydrogenase at Pt. Langmuir 17(8), 2471–2477 (2001). https://doi.org/10.1021/la0007729
J. Prusseit, M. Simon, C. von der Brelie, A. Heep, E. Molitor, S. Völz, A. Simon, Epidemiology, prevention and management of ventriculoperitoneal shunt infections in children. Pediatr. Neurosurg. 45(5), 325–336 (2009). https://doi.org/10.1159/000257520
E. Pyshnov, Electrochemical Studies of the Adsorption of Bovine Serum Albumin on a Platinum Surface (Thesis, McGill University, M.E, 2004)
C. Qin, A.H. Olivencia-Yurvati, A.G. Williams, D. Eskildsen, R.T. Mallet, P.K. Dasgupta, Inline flow sensor for ventriculoperitoneal shunts: experimental evaluation in swine. Med. Eng. Phys. 67(May), 66–72 (2019). https://doi.org/10.1016/j.medengphy.2019.03.010
R. Raj, S. Lakshmanan, D. Apigo, A. Kanwal, S. Liu, T. Russell, J.R. Madsen, G.A. Thomas, R.C. Farrow, “Demonstration that a new flow sensor can operate in the clinical range for cerebrospinal fluid flow.” Sens. Actuators, A 234, 223–231 (2015). https://doi.org/10.1016/j.sna.2015.08.023
S. Rammos, J. Klopfenstein, L. Augsburger, H. Wang, A. Wagenbach, J. Poston, G. Lanzino, Conversion of external ventricular drains to ventriculoperitoneal shunts after aneurysmal subarachnoid hemorrhage: effects of site and protein/red blood cell counts on shunt infection and malfunction: clinical article. J. Neurosurg. 109(6), 1001–1004 (2008). https://doi.org/10.3171/JNS.2008.109.12.1001
Rasalingam, Vinnothini. 2018. “A System for Electrochemical Impedance Spectroscopy of Electorchemical Sensors.” M.S. Thesis, Imperial College London.
D. Rodger, A. Fong, W. Li, H. Ameri, A. Ahuja, C. Gutierrez, I. Lavrov, H. Zhong, P. Menon, E. Meng, Flexible parylene-based multielectrode array technology for high-density neural stimulation and recording. Sens. Actuators, B Chem. 132(2), 449–460 (2008). https://doi.org/10.1016/j.snb.2007.10.069
B.Z. Roitberg, N. Khan, M.S. Alp, T. Hersonskey, F.T. Charbel, J.I. Ausman, Bedside external ventricular drain placement for the treatment of acute hydrocephalus. British J Neurosur 15, 324–327 (2001)
R. Rouhana, S.M. Budge, S.M. MacDonald, S.G. Roscoe, Electrochemical studies of the interfacial behaviour of a-lactalbumin and bovine serum albumin. Food Res. Int. 30(1), 13–20 (1997)
K. Scholten, E. Meng, Materials for microfabricated implantable devices: a review. Lab Chip 15(22), 4256–4272 (2015). https://doi.org/10.1039/C5LC00809C
J.P. Seymour, Y.M. Elkasabi, H.-Y. Chen, J. Lahann, D.R. Kipke, The insulation performance of reactive parylene films in implantable electronic devices. Biomaterials 30(31), 6158–6167 (2009). https://doi.org/10.1016/j.biomaterials.2009.07.061
P. Takmakov, K. Kiersten Ruda, S. Phillips, I.S. Isayeva, V. Krauthamer, C.G. Welle, Rapid evaluation of the durability of cortical neural implants using accelerated aging with reactive oxygen species. J. Neural Eng. 12(2), 026003 (2015). https://doi.org/10.1088/1741-2560/12/2/026003
Di. Terlizzi, Roberta, and Simon Platt. , The function, composition and analysis of cerebrospinal fluid in companion animals: part i - function and composition. Vet. J. 172(3), 422–431 (2006). https://doi.org/10.1016/j.tvjl.2005.07.021
M. Turgut, D. Alabaz, F. Erbey, E. Kocabas, T. Erman, E. Alhan, N. Aksaray, Cerebrospinal fluid shunt infections in children. Pediatr. Neurosurg. 41(3), 131–136 (2005). https://doi.org/10.1159/000085869
N. Turner, M. Armitage, R. Butler, G. Ireland, An in vitro model to evaluate cell adhesion to metals used in implantation shows significant differences between palladium and gold or platinum. Cell Biol. Int. 28(7), 541–547 (2004). https://doi.org/10.1016/j.cellbi.2004.04.009
E.A. Vogler, Protein adsorption in three dimensions. Biomaterials 33(5), 1201–1237 (2012). https://doi.org/10.1016/j.biomaterials.2011.10.059
A. Wang, X. Liang, J.P. McAllister II, J. Li, K. Brabant, C. Black, P. Finlayson et al., Stability of and inflammatory response to silicon coated with a fluoroalkyl self-assembled monolayer in the central nervous system. J. Biomed. Mater. Res., Part A 81A(2), 363–372 (2007). https://doi.org/10.1002/jbm.a.31034
P. Webb, Temperatures of skin, subcutaneous tissue, muscle and core in resting men in cold, comfortable and hot conditions. Eur. J. Appl. Physiol. 64(5), 471–476 (1992). https://doi.org/10.1007/BF00625070
J.E.I. Wright, K. Fatih, C.L. Brosseau, S. Omanovic, S.G. Roscoe, L-Phenylalanine adsorption on pt: electrochemical impedance spectroscopy and quartz crystal nanobalance studies. J. Electroanal. Chem. 550–551, 41–51 (2003). https://doi.org/10.1016/S0022-0728(03)00026-3
L. Yang, Y. Li, Detection of viable salmonella using microelectrode-based capacitance measurement coupled with immunomagnetic separation. J. Microbiol. Methods 64(1), 9–16 (2006). https://doi.org/10.1016/j.mimet.2005.04.022
Acknowledgements
The authors would like to thank Dr. Marek Czosnyka of the University of Cambridge for providing access to his clinical ICP dataset, Dr. Donghai Zhu of the USC Core Nanofabrication Facilities for his fabrication assistance and the members of the Biomedical Microsystems Lab for their helpful discussion. This work was funded in part by the National Science Foundation (NSF) under EFRI-1332394 and PFI-TT-1827773, the Rudi Schulte Research Institute, and the Coulter Translational Research Partnership Program.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hudson, T.Q., Baldwin, A., Samiei, A. et al. A portable multi-sensor module for monitoring external ventricular drains. Biomed Microdevices 23, 45 (2021). https://doi.org/10.1007/s10544-021-00579-8
Accepted:
Published:
DOI: https://doi.org/10.1007/s10544-021-00579-8