Abstract
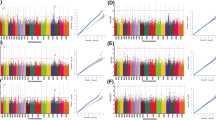
Genetic improvement of seed yield per plant (SY) is one of the major objectives in Brassica napus breeding programme. SY, being a complex quantitative trait is directly and indirectly influenced by yield-component traits such as siliqua length (SL), number of seeds per siliqua (NSS), and thousand seed weight (TSW). Therefore, concurrent improvement in SL, NSS and TSW can lead to higher SY in B. napus. This study was conducted to identify significant SNPs and putative candidate genes governing SY and its component traits (SL, NSS, TSW). All these traits were evaluated in a diverse set of 200 genotypes representing diversity from wide geographical locations. Of these, a set of 125 genotypes were chosen based on pedigree diversity and multi-location trait variation for genotyping by sequencing (GBS). Best linear unbiased predictors (BLUPs) of all the traits were used for genome-wide association study (GWAS) with 85,126 SNPs obtained from GBS. A total of 16, 18, 27 and 18 SNPs were found to be significantly associated for SL, NSS, TSW and SY respectively. Based on linkage disequilibrium decay analysis, 150 kb genomic region flanking the SNP was used for the identification of underlying candidate genes for each test trait. Important candidate genes involved in phytohormone signaling (WAT1, OSR1, ARR8, CKX1, REM7, REM9, BG1) and seed storage proteins (Cruciferin) were found to have significant influence on seed weight and yield. Genes involved in sexual reproduction and fertilization (PERK7, PERK13, PRK3, GATA15, NFD6) were found to determine the number of seeds per siliqua. Several genes found in this study namely ATS3A, CKX1, SPL2, SPL6, SPL9, WAT1 showed pleiotropic effect with yield component traits. Significant SNPs and putative candidate genes identified for SL, NSS, TSW and SY could be used in marker-assisted breeding for improvement of crop yield in B. napus. Genotypes identified with high SL, NSS, TSW and SY could serve as donors in crop improvement programs in B. napus.





Similar content being viewed by others
Abbreviations
- SL:
-
Siliqua length
- NSS:
-
Number of seeds per siliqua
- TSW:
-
Thousand seed weight
- SY:
-
Seed yield per plant
- BLUPs:
-
Best linear unbiased predictors
- GWAS:
-
Genome wide association study
- SNPs:
-
Single nucleotide polymorphisms
- PAU:
-
Punjab Agricultural University
- BTI:
-
Bathinda
- LDH:
-
Ludhiana
- ANOVA:
-
Analysis of variance
- CV:
-
Coefficient of variance
- CTAB:
-
Cetyl trimethyl ammonium bromide
- GBS:
-
Genotyping by sequencing
- FarmCPU:
-
Fixed and Random Model Circulating Unification
- GAPIT:
-
Genomic Association and Prediction Integrated Tool
- PCs:
-
Principal Components
- LD:
-
Linkage disequilibrium
- ARF:
-
Auxin response factor
- AuxREs:
-
Auxin response elements
- OSR:
-
ORGAN SIZE RELATED
- BR:
-
Brassinosteroid
- SSP:
-
Seed storage proteins
- ER:
-
Endoplasmic reticulum
- PSV:
-
Protein storage vacuoles
- SERK:
-
Somatic embryogenesis receptor kinase
- LRR-VII-2 RKs:
-
Leucine rich repeat receptor kinases
- SPL:
-
Squamosa promoter binding like protein
References
Alvarado G, Rodríguez FM, Pacheco A, Burgueño J, Crossa J, Vargas M, Pérez-Rodríguez P, Lopez-Cruz MA (2020) META-R: A software to analyze data from multi-environment plant breeding trials. Crop J. https://doi.org/10.1016/j.cj.2020.03.010
Anonymous (2020) Package of Practices for Rabi Crops. Pp 20–31. Punjab Agricultural University, Ludhiana
Bassham DC, Raikhel NV (1998) An Arabidopsis VPS45p homolog implicated in protein transport to the vacuole. Plant Physiol 117(2):407–415
Bennett EJ, Roberts JA, Wagstaff C (2011) The role of the pod in seed development: strategies for manipulating yield. New Phytol 190:838–853
Cao J, Li G, Qu D, Li X, Wang Y (2020) Into the seed: Auxin controls seed development and grain yield. Int J Mol Sci 21(1662):1–17
Cao R, Guo L, Ma M, Zhang W, Liu X, Zhao H (2019) Identification and functional characterization of Squamosa promoter binding protein-like gene TaSPL16 in wheat (Triticum aestivum L.). Front Plant Sci 10:212
Cardon G, Höhmann S, Klein J, Nettesheim K, Saedler H, Huijser P (1999) Molecular characterisation of the Arabidopsis SBP-box genes. Gene 237(1):91–104
Carruthers JM, Cook SM, Wright GA, Osborne JL, Clark SJ, Swain JL, Haughton AJ (2017) Oilseed rape (Brassica napus) as a resource for farmland insect pollinators: quantifying floral traits in conventional varieties and breeding systems. GCB Bioenergy 9(8):1370–1379
Chakraborty S, Pan H, Tang Q, Woolard C, Xu G (2018) The Extracellular Domain of Pollen Receptor Kinase 3 is structurally similar to the SERK family of co-receptors. Sci Rep 8(1):1–2
Chay P, Thurling N (1989) Identification of genes controlling pod length in spring rapeseed, Brassica napus L., and their utilization for yield improvement. Plant Breed 103:54–62
Danecek P, Auton A, Abecasis G, Albers CA, Banks E, DePristo MA, Handsaker RE, Lunter G, Marth GT, Sherry ST, McVean G (2011) The variant call format and VCFtools. J Bioinform 27(15):2156–2158
Day RC, Herridge RP, Ambrose BA, Macknight RC (2008) Transcriptome analysis of proliferating Arabidopsis endosperm reveals biological implications for the control of syncytial division, cytokinin signaling, and gene expression regulation. Plant Physiol 148(4):1964–1984
Diepenbrock W (2000) Yield analysis of winter oilseed rape (Brassica napus L.): a review. Field Crops Res 67:35–49
Doyle JJ, Doyle JL, Hortoriun LB (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
Drews GN, Yadegari R (2002) Development and function of the angiosperm female gametophyte. Annu Rev Genet 36:99–124
Duggal P, Gillanders EM, Holmes TN, Bailey-Wilson JE (2008) Establishing an adjusted p-value threshold to control the family-wide type 1 error in genome wide association studies. BMC Genom 9:1–8
Feng G, Qin Z, Yan J, Zhang X, Hu Y (2011) Arabidopsis ORGAN SIZE RELATED1 regulates organ growth and final organ size in orchestration with ARGOS and ARL. New Phytol 191(3):635–646
Fujiki Y, Teshima H, Kashiwao S, Kawano-Kawada M, Ohsumi Y, Kakinuma Y, Sekito T (2017) Functional identification of AtAVT3, a family of vacuolar amino acid transporters. Arabidopsis FEBS Lett 591(1):5–15
Gehrig PM, Krzyzaniak A, Barciszewski J, Biemann K (1996) Mass spectrometric amino acid sequencing of a mixture of seed storage proteins (napin) from Brassica napus, products of a multigene family. Proc Natl Acad Sci USA 93:3647–3652
Haffani Y, Silva-Gagliardi N, Sewter S, Aldea MG, Zhao Z, Nakhamchik A, Cameron R, Goring D (2006) Altered expression of PERK receptor kinases in Arabidopsis leads to changes in growth and floral organ formation. Plant Signal Behav 1(5):251–260
Hara-Nishimura I, Shimada T, Hatano K, Takeuchi Y, Nishimura M (1998) Transport of storage proteins to protein storage vacuoles is mediated by large precursor-accumulating vesicles. Plant Cell 10:825–836
Hasan M, Seyis F, Badani AG, Pons-Kuhnemann J, Friedt W, Luhs W, Snowdon RJ (2006) Analysis of genetic diversity in the Brassica napus L. gene pool using SSR markers. Genet Resour Crop Evol 53:793–802
Hirner A, Ladwig F, Stransky H, Okumoto S, Keinath M, Harms A, Frommer WB, Koch W (2006) Arabidopsis LHT1 is a high-affinity transporter for cellular amino acid uptake in both root epidermis and leaf mesophyll. Plant Cell 18(8):1931–1946
Hohl I, Robinson DG, Chrispeels MJ, Hinz G (1996) Transport of storage proteins to the vacuole is mediated by vesicles without a clathrin coat. J Cell Sci 109:2539–2550
Kaler AS, Gillman JD, Beissinger T, Purcell LC (2020) Comparing different statistical models and multiple testing corrections for association mapping in soybean and maize. Front Plant Sci 10:1794. https://doi.org/10.3389/fpls.2019.01794
Kaur H, Kaur P (2015) Temperature features in different agroclimatic zones of Punjab. Agricultural Res J 52(4):32–35
Kaur H, Wang L, Stawniak N, Sloan R, Van Erp H, Eastmond P, Bancroft I (2020) The impact of reducing fatty acid desaturation on the composition and thermal stability of rapeseed oil. Plant Biotechnol J 18(4):983–991
Kaur P, Hundal SS (2008) Climatic trends in different agroclimatic zones of Punjab: II Rainfall features and its variability. J Res 45(1–2):41–50
Lebowitz R (1989) Image analysis measurements and repeatability estimates of siliqua morphological traits in Brassica campestris L. Euphytica 43:113–116
Lee HK, Goring D (2020) Two groups of Arabidopsis receptor kinases preferentially regulate the growth of intra-species pollen tubes in the female reproductive tract. Curr Bio. https://doi.org/10.2139/ssrn.3564989
Letham DS (1973) Cytokinins from Zea mays. Phytochem 12(10):2445–2455
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinform 25(14):1754–1760. https://doi.org/10.1093/bioinformatics/btp324
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Marth G, Abecasis G, Durbin R (2009) The sequence alignment/map format and SAMtools. Bioinform 25(16):2078–2079
Li HJ, Zhu SS, Zhang MX, Wang T, Liang L, Xue Y, Shi DQ, Liu J, Yang WC (2015) Arabidopsis CBP1 is a novel regulator of transcription initiation in central cell-mediated pollen tube guidance. Plant Cell 27(10):2880–28893
Li J, Nie X, Tan JL, Berger F (2013) Integration of epigenetic and genetic controls of seed size by cytokinin in Arabidopsis. Proc Natl Acad Sci USA 110(38):15479–15484
Li N, Song D, Peng W, Zhan J, Shi J, Wang X, Liu G, Wang H (2019) Maternal control of seed weight in rapeseed (Brassica napus L.): the causal link between the size of pod (mother, source) and seed (offspring, sink). Plant Biotechnol J 17(4):736–749
Lipka AE, Tian F, Wang Q, Peiffer J, Li M, Bradbury PJ, Gore MA, Buckler ES, Zhang Z (2012) GAPIT: genome association and prediction integrated tool. Bioinform 28:2397–2399
Liu J, Hua W, Hu Z, Yang H, Zhang L, Li R, Deng L, Sun X, Wang X, Wang H (2015a) Natural variation in ARF18 gene simultaneously affects seed weight and silique length in polyploid rapeseed. Proc Natl Acad Sci 112(37):E5123-5132
Liu L, Tong H, Xiao Y, Che R, Xu F, Hu B, Liang C, Chu J, Li J, Chu C (2015b) Activation of Big Grain1 significantly improves grain size by regulating auxin transport in rice. Proc Natl Acad Sci 112(35):11102–11107
Liu P, Zhang C, Ma JQ, Zhang LY, Yang B, Tang XY, Huang L, Zhou XT, Lu K, Li JN (2018) Genome-wide identification and expression profiling of cytokinin oxidase/dehydrogenase (CKX) genes reveal likely roles in pod development and stress responses in oilseed rape (Brassica napus L.). Genes 9(3):168
Liu X, Huang M, Fan B, Buckler ES, Zhang Z (2016) Iterative usage of fixed and random effect models for powerful and efficient genome-wide association studies. PLoS Genet 12(2):1005767
Mizzotti C, Rotasperti L, Moretto M, Tadini L, Resentini F, Galliani BM, Galbiati M, Engelen K, Pesaresi P, Masiero S (2018) Time-course transcriptome analysis of Arabidopsis siliques discloses genes essential for fruit development and maturation. Plant Physiol 178(3):1249–1268
Mok DW, Mok MC (2001) Cytokinin metabolism and action. Annu Rev Plant Biol 52:89–118
Na G, Mu X, Grabowski P, Schmutz J, Lu C (2019) Enhancing micro-RNA 167A expression in seed decreases the α-linolenic acid content and increases seed size in Camelina sativa. Plant J 98(2):346–358
Nagaharu U (1935) Genome analysis in Brassica with special reference to the experimental formation of B. napus and peculiar mode of fertilization. J Jap Bot 7:389–452
Nuccio ML, Thomas TL (1999) ATS1 and ATS3: Two novel embryo-specific genes in Arabidopsis thaliana. Plant Mol Biol 391(1):1153–1163
Oufattole M, Park JH, Poxleitner M, Jiang L, Rogers JC (2005) Selective membrane protein internalization accompanies movement from the endoplasmic reticulum to the protein storage vacuole pathway in Arabidopsis. Plant Cell 17(11):3066–3080
Pechan PA, Morgan DG (1985) Defoliation and its effects on pod and seed development in oil seed rape (Brassica napus L.). J Exp Bot 36(3):458–468
Portereiko MF, Sandaklie-Nikolova L, Lloyd A, Dever CA, Otsuga D, Drews GN (2006) NUCLEAR FUSION DEFECTIVE1 encodes the Arabidopsis RPL21M protein and is required for karyogamy during female gametophyte development and fertilization. Plant Physiol 141(3):957–965
Prakash S, Wu X, Bhat SR (2012) History, evolution and domestication of Brassica crops. Plant Breed Rev 35:19–84
Priyamedha BK, Thomas L, Bala M, Singh VV, Singh D (2015) Status and perspective of canola quality rapeseed-mustard cultivation in India: a review. J Oilseed Brassica 1(1):142–151
R Core Team (2018) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Ranocha P, Dima O, Nagy R, Felten J, Corratgé-Faillie C, Novák O, Morreel K, Lacombe B, Martinez Y, Pfrunder S, Jin X (2013) Arabidopsis WAT1 is a vacuolar auxin transport facilitator required for auxin homoeostasis. Nat Commun 4(1):1–9
RStudio Team (2020) RStudio: Integrated Development for R. RStudio, Inc., Boston, MA URL http://www.rstudio.com/
Sandhu SK, Pal L, Kaur J, Bhatia D (2019) Genome wide association studies for yield and its component traits under terminal heat stress in Indian mustard (Brassica juncea L.). Euphytica 215(11):1–5
Shi J, Zhan J, Yang Y, Ye J, Huang S, Li R, Wang X, Liu G, Wang H (2015) Linkage and regional association analysis reveal two new tightly-linked major-QTLs for pod number and seed number per pod in rapeseed (Brassica napus L.). Sci Rep 5(1):1–8
Steketee CJ, Sinclair TR, Riar MK, Schapaugh WT, Li Z (2019) Unraveling the genetic architecture for carbon and nitrogen related traits and leaf hydraulic conductance in soybean using genome-wide association analyses. BMC Genom 20(1):1–8
Takeuchi H, Higashiyama T (2016) Tip-localized receptors control pollen tube growth and LURE sensing in Arabidopsis. Nature 531:245–248
Ulmasov T, Hagen G, Guilfoyle TJ (1997) ARF1, a transcription factor that binds to auxin response elements. Science 276(5320):1865–1868
Ulmasov T, Hagen G, Guilfoyle TJ (1999) Dimerization and DNA binding of auxin response factors. Plant J 19(3):309–319
Wang H, Yan M, Xiong M, Wang P, Liu Y, Xin Q, Wan L, Yang G, Hong D (2020) Genetic dissection of thousand-seed weight and fine mapping of cqSW. A03–2 via linkage and association analysis in rapeseed (Brassica napus L.). Theor Appl Genet 133(4):1321–1335
Wang L, Yin H, Qian Q, Yang J, Huang C, Hu X, Luo D (2009) NECK LEAF 1, a GATA type transcription factor, modulates organogenesis by regulating the expression of multiple regulatory genes during reproductive development in rice. Cell Res 19(5):598–611
Wang S, Wu K, Yuan Q, Liu X, Liu Z, Lin X et al (2012) Control of grain size, shape and quality by OsSPL16 in rice. Nat Genet 44:950–954
Xie K, Wu C, Xiong L (2006) Genomic organization, differential expression, and interaction of SQUAMOSA promoter-binding-like transcription factors and microRNA156 in rice. Plant Physiol 142:280–293. https://doi.org/10.1104/pp.106.084475
Yang P, Shu C, Chen L, Xu J, Wu J, Liu K (2012) Identification of a major QTL for silique length and seed weight in oilseed rape (Brassica napus L.). Theor Appl Genet 125(2):285–296
Yang W, Guo Z, Huang C, Duan L, Chen G, Jiang N, Fang W, Feng H, Xie W, Lian X, Wang G (2014) Combining high-throughput phenotyping and genome-wide association studies to reveal natural genetic variation in rice. Nat Commun 5:5087
Yang Y, Shi J, Wang X, Liu G, Wang H (2016) Genetic architecture and mechanism of seed number per pod in rapeseed: elucidated through linkage and near-isogenic line analysis. Sci Rep 6(1):1–10
Zhang B, Liu X, Zhao G, Mao X, Li A, Jing R (2014) Molecular characterization and expression analysis of Triticum aestivum squamosa promoter binding protein-box genes involved in ear development. J Integr Plant Biol 56:571–581
Zhang C, Dong SS, Xu JY, He WM, Yang TL (2019) PopLDdecay: a fast and effective tool for linkage disequilibrium decay analysis based on variant call format files. Bioinform 35(10):1786–1788
Zhang L, Yang G, Liu P, Hong D, Li S, He Q (2011) Genetic and correlation analysis of silique-traits in Brassica napus L. by quantitative trait locus mapping. Theor Appl Genet 122(1):21–31
Zhu Y, Ye J, Zhan J, Zheng X, Zhang J, Shi J, Wang X, Liu G, Wang H (2020) Validation and characterization of a seed number per silique quantitative trait locus qSN A7 in Rapeseed (Brassica napus L.). Front Plant Sci 11:68
Zouhar J, Rojo E, Bassham DC (2009) AtVPS45 is a positive regulator of the SYP41/SYP61/VTI12 SNARE complex involved in trafficking of vacuolar cargo. Plant Physiol 149(4):1668–1678
Acknowledgements
The B. napus germplasm and advance breeding lines, used in this study, were collected/developed and maintained by ICAR National Professor Dr. S. S. Banga. The authors duly acknowledge Dr. S. S. Banga for providing germplasm and valuable guidance. Authors also acknowledge Dr. Paramjit Singh (Director, Regional Research Station, Bathinda) for providing necessary facilities required for conducting field experiments at Bathinda. The fellowship from University Grant Commission (UGC) under CSIR-UGC JRF for the Ph.D. programme of Lalit Pal is duly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pal, L., Sandhu, S.K., Bhatia, D. et al. Genome-wide association study for candidate genes controlling seed yield and its components in rapeseed (Brassica napus subsp. napus). Physiol Mol Biol Plants 27, 1933–1951 (2021). https://doi.org/10.1007/s12298-021-01060-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-021-01060-9