Abstract
Background and objective
Pathogenic variants in KCNT1 have been associated with severe forms of epilepsy, typically sleep-related hypermotor epilepsy or epilepsy of infancy with migrating focal seizures. To show that pathogenic variants in KCNT1 can be associated with mild extra-frontal epilepsy, we report a KCNT1 family with a wide spectrum of phenotypes ranging from developmental and epileptic encephalopathy to mild focal epilepsy without cognitive regression and not consistent with sleep-related hypermotor epilepsy.
Methods
A large Canadian family of Caucasian descent including 9 affected family members was recruited. Family members were phenotyped by direct interview and review of existing medical records. Clinical epilepsy gene panel analysis and exome sequencing were performed.
Results
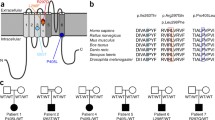
Phenotypic information was available for five family members of which two had developmental and epileptic encephalopathy and three had normal development and focal epilepsy with presumed extra-frontal onset. All three had predominantly nocturnal seizures that did not show hyperkinetic features. All three reported clusters of seizures at night with a feeling of being unable to breathe associated with gasping for air, choking and/or repetitive swallowing possibly suggesting insular or opercular involvement. Genetic analysis identified a heterozygous KCNT1 c.2882G > A, p.Arg961His variant that was predicted to be deleterious.
Discussion
This family demonstrates that the phenotypic spectrum associated with KCNT1 pathogenic variants is broader than previously assumed. Our findings indicate that variants in KCNT1 can be associated with mild focal epilepsy and should not be excluded during variant interpretation in such patients based solely on gene–disease validity.

Similar content being viewed by others
Availability of data and material
All de-identified data is included in the article.
Code availability
Not applicable.
References
Heron SE, Smith KR, Bahlo M et al (2012) Missense mutations in the sodium-gated potassium channel gene KCNT1 cause severe autosomal dominant nocturnal frontal lobe epilepsy. Nat Genet 44:1188–1190. https://doi.org/10.1038/ng.2440
Barcia G, Fleming MR, Deligniere A et al (2012) De novo gain-of-function KCNT1 channel mutations cause malignant migrating partial seizures of infancy. Nat Genet 44:1255–1259. https://doi.org/10.1038/ng.2441
Borlot F, Abushama A, Morrison-Levy N et al (2020) KCNT1-related epilepsy: an international multicenter cohort of 27 pediatric cases. Epilepsia 61:679–692. https://doi.org/10.1111/epi.16480
Ohba C, Kato M, Takahashi N et al (2015) De novo KCNT1 mutations in early-onset epileptic encephalopathy. Epilepsia 56:e121–e128. https://doi.org/10.1111/epi.13072
McTague A, Appleton R, Avula S et al (2013) Migrating partial seizures of infancy: expansion of the electroclinical, radiological and pathological disease spectrum. Brain : a journal of neurology 136:1578–1591. https://doi.org/10.1093/brain/awt073
Routier L, Verny F, Barcia G et al (2019) Exome sequencing findings in 27 patients with myoclonic-atonic epilepsy: is there a major genetic factor? Clin Genet 96:254–260. https://doi.org/10.1111/cge.13581
Bonardi CM, Heyne HO, Fiannacca M et al (2021) KCNT1-related epilepsies and epileptic encephalopathies: phenotypic and mutational spectrum. Brain. https://doi.org/10.1093/brain/awab219
Møller RS, Heron SE, Larsen LHG et al (2015) Mutations in KCNT1 cause a spectrum of focal epilepsies. Epilepsia 56:e114–e120. https://doi.org/10.1111/epi.13071
Rubboli G, Plazzi G, Picard F et al (2019) Mild malformations of cortical development in sleep-related hypermotor epilepsy due to KCNT1 mutations. Ann Clin Transl Neurol 6:386–391. https://doi.org/10.1002/acn3.708
Barcia G, Chemaly N, Kuchenbuch M et al (2019) Epilepsy with migrating focal seizures: KCNT1 mutation hotspots and phenotype variability. Neurol Genet 5:e363. https://doi.org/10.1212/NXG.0000000000000363
Cataldi M, Nobili L, Zara F et al (2019) Migrating focal seizures in autosomal dominant sleep-related hypermotor epilepsy with KCNT1 mutation. Seizure 67:57–60. https://doi.org/10.1016/j.seizure.2019.02.019
Licchetta L, Pippucci T, Baldassari S et al (2020) Sleep-related hypermotor epilepsy (SHE): contribution of known genes in 103 patients. Seizure 74:60–64. https://doi.org/10.1016/j.seizure.2019.11.009
Kerr M, Hume S, Omar F et al (2020) MITO-FIND: a study in 390 patients to determine a diagnostic strategy for mitochondrial disease. Mol Genet Metab 131:66–82. https://doi.org/10.1016/j.ymgme.2020.08.009
Richards S, Aziz N, Bale S et al (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17:405–424. https://doi.org/10.1038/gim.2015.30
Köhler S, Carmody L, Vasilevsky N et al (2019) Expansion of the Human Phenotype Ontology (HPO) knowledge base and resources. Nucleic Acids Res 47:D1018–D1027. https://doi.org/10.1093/nar/gky1105
Singleton MV, Guthery SL, Voelkerding KV et al (2014) Phevor combines multiple biomedical ontologies for accurate identification of disease-causing alleles in single individuals and small nuclear families. Am J Human Genet 94:599–610. https://doi.org/10.1016/j.ajhg.2014.03.010
Genome Aggregation Database Consortium, Karczewski KJ, Francioli LC et al (2020) The mutational constraint spectrum quantified from variation in 141,456 humans. Nature 581:434–443. https://doi.org/10.1038/s41586-020-2308-7
Zhu X, Padmanabhan R, Copeland B et al (2017) A case-control collapsing analysis identifies epilepsy genes implicated in trio sequencing studies focused on de novo mutations. PLoS Genet 13:e1007104. https://doi.org/10.1371/journal.pgen.1007104
Parrini E, Marini C, Mei D et al (2017) Diagnostic targeted resequencing in 349 patients with drug-resistant pediatric epilepsies identifies causative mutations in 30 different genes. Hum Mutat 38:216–225. https://doi.org/10.1002/humu.23149
Scheffer IE, Zhang Y-H, Jansen FE, Dibbens L (2009) Dravet syndrome or genetic (generalized) epilepsy with febrile seizures plus? Brain Develop 31:394–400. https://doi.org/10.1016/j.braindev.2009.01.001
Hansen N, Widman G, Hattingen E et al (2017) Mesial temporal lobe epilepsy associated with KCNT1 mutation. Seizure 45:181–183. https://doi.org/10.1016/j.seizure.2016.12.018
Rehm HL, Berg JS, Brooks LD et al (2015) ClinGen–the clinical genome resource. N Engl J Med 372:2235–2242. https://doi.org/10.1056/NEJMsr1406261
Proserpio P, Cossu M, Francione S et al (2011) Insular-opercular seizures manifesting with sleep-related paroxysmal motor behaviors: a stereo-EEG study. Epilepsia 52:1781–1791. https://doi.org/10.1111/j.1528-1167.2011.03254.x
Milligan CJ, Li M, Gazina EV et al (2014) KCNT1 gain of function in 2 epilepsy phenotypes is reversed by quinidine. Ann Neurol 75:581–590. https://doi.org/10.1002/ana.24128
Mullen SA, Carney PW, Roten A et al (2018) Precision therapy for epilepsy due to KCNT1 mutations: a randomized trial of oral quinidine. Neurology 90:e67–e72. https://doi.org/10.1212/WNL.0000000000004769
Mikati MA, Jiang YH, Carboni M et al (2015) Quinidine in the treatment of KCNT1-positive epilepsies. Ann Neurol 78:995–999. https://doi.org/10.1002/ana.24520
Acknowledgements
We thank the family for participation in our study. The study was supported by the Department of Clinical Neurosciences, Hotchkiss Brain Institute and Alberta Children’s Hospital Research Institute, University of Calgary, to KMK.
Funding
The study was supported by the Department of Clinical Neurosciences, Hotchkiss Brain Institute and Alberta Children’s Hospital Research Institute, University of Calgary, to KMK.
Author information
Authors and Affiliations
Contributions
CC: analyzed the data; drafted the manuscript for intellectual content. JPA: Designed and conceptualized study; analyzed the data; revised the manuscript for intellectual content. Setareh Ashtiani: Major role in the acquisition of data; revised the manuscript for intellectual content. PF: major role in the acquisition of data; analyzed the data; revised the manuscript for intellectual content. CPM: major role in the acquisition of data; analyzed the data; revised the manuscript for intellectual content. MK: major role in the acquisition of data; analyzed the data; revised the manuscript for intellectual content. AK: major role in the acquisition of data; analyzed the data; revised the manuscript for intellectual content. P-YBA: designed and conceptualized study; analyzed the data; revised the manuscript for intellectual content. KMK: designed and conceptualized study; analyzed the data; drafted the manuscript for intellectual content.
Corresponding author
Ethics declarations
Conflicts of interest
C. Cherian reports no disclosures relevant to the manuscript. J.P. Appendino reports personal and educational fees from UCB Pharma, PendoPharm Inc., and Sunovion Pharmaceuticals Inc. S. Ashtiani reports no disclosures relevant to the manuscript. P. Federico reports no disclosures relevant to the manuscript. C.P. Molnar reports no disclosures relevant to the manuscript. A. Khan and M. Kerr received part of the funding for the MITO-FIND project from MITOCANADA through the Alberta Children’s Hospital Research Foundation. P. Y. B. Au reports no disclosures relevant to the manuscript. K.M. Klein reports personal fees from UCB Pharma, Novartis Pharma AG, Eisai, and GW Pharmaceuticals, grants from the federal state Hessen, Germany, through the LOEWE program and from the Canadian Institutes of Health Research.
Ethical approval
The study was approved by the Conjoint Health Research Ethics Board at University of Calgary and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Consent to participate
All participants or their legal guardians provided written informed consent prior to inclusion in the study.
Consent for publication
The consent includes publication of de-identified health information.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cherian, C., Appendino, J.P., Ashtiani, S. et al. The phenotypic spectrum of KCNT1: a new family with variable epilepsy syndromes including mild focal epilepsy. J Neurol 269, 2162–2171 (2022). https://doi.org/10.1007/s00415-021-10808-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-021-10808-y