Abstract
Background
Thyroid hormones are critical regulators of metabolism, development and growth in mammals. However, the genetic association of thyroid-related hormones in the Chinese Han population is not fully understood.
Objective
We aimed to identify the genetic loci associated with circulating thyroid-related hormones concentrations in the healthy Chinese Han population.
Methods
Genotyping was performed in 124 individuals using Applied Biosystems™ Axiom™ PMDA, and 796,288 single nucleotide polymorphisms (SNPs) were available for the GWAS analysis. For replication, eleven SNPs were selected as candidate loci for genotyping by Agena MassARRAY platform in additional samples (313 subjects). The values of p < 5 × 10− 6 suggest a suggestively significant genome-wide association with circulating thyroid-related hormones concentrations.
Results
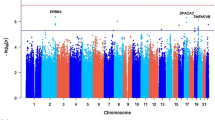
We identified that rs11178277 (PTPRB, p = 4.88 × 10− 07) and rs7320337 (LMO7DN-KCTD12, p = 1.22 × 10− 06) were associated with serum FT3 level. Three SNPs (rs4850041 in LOC105373394-LINC01249: p = 3.55 × 10− 06, rs6867291 in LINC02208: p = 2.40 × 10− 06 and rs79508321 in WWOX: p = 3.35 × 10− 06) were related to circulating T3 level. Rs12474167 (LOC105373394-LINC01249, p = 1.65 × 10− 06) and rs1864553 (IWS1, p = 2.00 × 10− 06) were associated with circulating T4 concentration. The association with TGA concentration was for rs17163542 in DISP1 (p = 3.46 × 10− 06) and rs12601151 in NOG-C17orf67 (p = 2.72 × 10− 07). Two genome-level significant SNPs (rs2114707 in LINC01314, p = 1.69 × 10− 06 and rs12601151, p = 1.41 × 10− 07) associated with serum TMA concentration were identified. Moreover, rs6083269 (CST1-CST2, p = 3.36 × 10− 06) was a significant locus for circulating TSH level. In replication, rs12601151 in NOG-C17orf67 was still associated with serum TGA level (p = 0.012).
Conclusions
The GWAS reported 11 new suggestively significant loci associated with circulating thyroid-related hormones levels among the Chinese Han population. These findings represented suggestively biological candidates for circulating thyroid-related hormones levels and provided new insights into the mechanisms of regulating serum TGA concentration.



Similar content being viewed by others
References
Bavarsad K, Hosseini M, Hadjzadeh MA, Sahebkar A (2019) The effects of thyroid hormones on memory impairment and Alzheimer’s disease. J Cell Physiol. January 24 (Online ahead of print)
Bednarek AK, Laflin KJ, Daniel RL, Liao Q, Hawkins KA, Aldaz CM (2000) WWOX, a novel WW domain-containing protein mapping to human chromosome 16q23.3-24.1, a region frequently affected in breast cancer. Cancer Res 60(8):2140–2145
Cancemi L, Romei C, Bertocchi S, Tarrini G, Spitaleri I, Cipollini M, Landi D, Garritano S, Pellegrini G, Cristaudo A et al (2011) Evidences that the polymorphism Pro-282-Ala within the tumor suppressor gene WWOX is a new risk factor for differentiated thyroid carcinoma. Int J Cancer 129(12):2816–2824
Carvalho DP, Dupuy C (2017) Thyroid hormone biosynthesis and release. Mol Cell Endocrinol 458:6–15
de Vries PS, Brown MR, Bentley AR, Sung YJ, Winkler TW, Ntalla I, Schwander K, Kraja AT, Guo X, Franceschini N et al (2019) Multiancestry genome-wide association study of lipid levels incorporating gene–alcohol interactions. Am J Epidemiol 188(6):1033–1054
Dias EP, Pimenta FJ, Sarquis MS, Dias Filho MA, Aldaz CM, Fujii JB, Gomez RS, De Marco L (2007) Association between decreased WWOX protein expression and thyroid cancer development. Thyroid 17(11):1055–1059
Duntas LH (2002) Thyroid disease and lipids. Thyroid 12(4):287–293
Fang Y, Ma X, Zeng J, Jin Y, Hu Y, Wang J, Liu R, Cao C (2018) The profile of genetic mutations in papillary thyroid cancer detected by whole exome sequencing. Cell Physiol Biochem 50(1):169–178
Feitosa MF, Lunetta KL, Wang L, Wojczynski MK, Kammerer CM, Perls T, Schupf N, Christensen K, Murabito JM, Province MA (2020) Gene discovery for high-density lipoprotein cholesterol level change over time in prospective family studies. Atherosclerosis 297:102–110
Gallois A, Mefford J, Ko A (2019) A comprehensive study of metabolite genetics reveals strong pleiotropy and heterogeneity across time and context. Nat Commun 10(1):4788
Hadlow NC, Rothacker KM, Wardrop R, Brown SJ, Lim EM, Walsh JP (2013) The relationship between TSH and free T4, in a large population is complex and nonlinear and differs by age and sex. J Clin Endocrinol Metab 98(7):2936–2943
Haines JL, Hauser MA, Schmidt S, Scott WK, Olson LM, Gallins P, Spencer KL, Kwan SY, Noureddine M, Gilbert JR et al (2005) Complement factor H variant increases the risk of age-related macular degeneration. Science 308(5720):419–421
Jabbar A, Pingitore A, Pearce SH, Zaman A, Iervasi G, Razvi S (2017) Thyroid hormones and cardiovascular disease. Nat Rev Cardiol 14(1):39–55
Koblinski JE, Ahram M, Sloane BF (2000) Unraveling the role of proteases in cancer. Clin Chim Acta 291(2):113–135
Krause C, Guzman A, Knaus P (2011) Noggin. Int J Biochem Cell Biol 43(4):478–481
Larsen PR (1982) Thyroid-pituitary interaction: feedback regulation of thyrotropin secretion by thyroid hormones. N Engl J Med 306(1):23–32
Lassová L, Niu Z, Golden EB, Cohen AJ, Adams SL (2009) Thyroid hormone treatment of cultured chondrocytes mimics in vivo stimulation of collagen X mRNA by increasing BMP 4 expression. J Cell Physiol 219(3):595–605
Matheis N, Okrojek R, Grus FH, Kahaly GJ (2012) Proteomics of tear fluid in thyroid-associated orbitopathy. Thyroid 22(10):1039–1045
Medici M, Porcu E, Pistis G, Teumer A, Brown SJ, Jensen RA, Rawal R, Roef GL, Plantinga TS, Vermeulen SH et al (2014) Identification of novel genetic Loci associated with thyroid peroxidase antibodies and clinical thyroid disease. PLoS Genet 10(2):e1004123
Nielsen TR, Appel EV, Svendstrup M, Ohrt JD, Dahl M, Fonvig CE, Hollensted M, Have CT, Kadarmideen HN, Pedersen O et al (2017) A genome-wide association study of thyroid stimulating hormone and free thyroxine in Danish children and adolescents. PLoS One 12(3):e0174204
Nunez MI, Ludes-Meyers J, Aldaz CM (2006) WWOX protein expression in normal human tissues. J Mol Histol 37(3–4):115–125
Pujari V, Radebaugh CA, Chodaparambil JV, Muthurajan UM, Almeida AR, Fischbeck JA, Luger K, Stargell LA (2010) The transcription factor Spn1 regulates gene expression via a highly conserved novel structural motif. J Mol Biol 404(1):1–15
Randolph JF, Lamb SV, Cheraskin JL, Schanbacher BJ, Salerno VJ, Mack KM, Scarlett JM, Place NJ (2015) Free thyroxine concentrations by equilibrium dialysis and chemiluminescent immunoassays in 13 hypothyroid dogs positive for thyroglobulin antibody. J Vet Intern Med 29(3):877–881
Rankin SA, Kormish J, Kofron M, Jegga A, Zorn AM (2011) A gene regulatory network controlling hhex transcription in the anterior endoderm of the organizer. Dev Biol 351(2):297–310
Ruf J, Feldt-Rasmussen U, Hegedüs L, Ferrand M, Carayon P (1994) Bispecific thyroglobulin and thyroperoxidase autoantibodies in patients with various thyroid and autoimmune diseases. J Clin Endocrinol Metab 79(5):1404–1409
Shuwen H, Xi Y, Miao D (2020) Nine genes mediate the therapeutic effects of iodine-131 radiotherapy in thyroid carcinoma patients. Dis Mark 2020:9369341
Sim X, Ong RT, Suo C, Tay WT, Liu J, Ng DP, Boehnke M, Chia KS, Wong TY, Seielstad M et al (2011) Transferability of type 2 diabetes implicated loci in multi-ethnic cohorts from Southeast Asia. PLoS Genet 7(4):e1001363
Sinha RA, Singh BK, Yen PM (2018) Direct effects of thyroid hormones on hepatic lipid metabolism. Nat Rev Endocrinol 14(5):259–269
Sinha RA, Bruinstroop E, Singh BK, Yen PM (2019) Nonalcoholic fatty liver disease and hypercholesterolemia: roles of thyroid hormones, metabolites, and agonists. Thyroid 29(9):1173–1191
Tang L, Wen JB, Wen P, Li X, Gong M, Li Q (2019) Long non-coding RNA LINC01314 represses cell migration, invasion, and angiogenesis in gastric cancer via the Wnt/β-catenin signaling pathway by down-regulating KLK4. Cancer Cell Int 19:94
Taylor PN, Porcu E, Chew S, Campbell PJ, Traglia M, Brown SJ, Mullin BH, Shihab HA, Min J, Walter K et al (2015) Whole-genome sequence-based analysis of thyroid function. Nat Commun 6:5681
Teumer A, Chaker L, Groeneweg S, Li Y, Di Munno C, Barbieri C, Schultheiss UT, Traglia M, Ahluwalia TS (2018) Genome-wide analyses identify a role for SLC17A4 and AADAT in thyroid hormone regulation. Nat Commun 9(1):4455
Tsiairis CD, McMahon AP (2008) Disp1 regulates growth of mammalian long bones through the control of Ihh distribution. Dev Biol 317(2):480–485
Walsh JP (2016) Managing thyroid disease in general practice. Med J Aust 205(4):179–184
Funding
This study was funded by the Hainan Provincial Natural Science Foundation of China (819QN354), National Natural Science Foundation of China (No. 81660013 and No.81860015) and Key Research and Development Plan of Hainan Province (No. ZDYF2018116).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Liang Huang, Fenghua Bai, Yutian Zhang, Shanshan Zhang, Tianbo Jin, Xingwei Wei, Xiaoli Zhou, Mei Lin, Yufei Xie, Chanyi He, Qi Lin, Tian Xie and Yipeng Ding declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
13258_2021_1165_MOESM1_ESM.tif
Supplementary material 1 Regional plots of significant Loci associated with serum FT3 level. Chromosomes are shown on the x-axis, and the − log10 p value on the y-axis. The color of each SNP spot, ranging from red to blue, reflects the degree of linkage disequilibrium (r2) with between other SNPs and top SNP (purple) (TIF 4657.8 kb)
13258_2021_1165_MOESM2_ESM.tif
Supplementary material 2 Regional plots of significant Loci associated with serum T3 level. Chromosomes are shown on the x-axis, and the − log10 p value on the y-axis. The color of each SNP spot, ranging from red to blue, reflects the degree of linkage disequilibrium (r2) with between other SNPs and top SNP (purple) (TIF 6976.1 kb)
13258_2021_1165_MOESM3_ESM.tif
Supplementary material 3 Regional plots of significant Loci associated with serum T4 level. Chromosomes are shown on the x-axis, and the − log10 p value on the y-axis. The color of each SNP spot, ranging from red to blue, reflects the degree of linkage disequilibrium (r2) with between other SNPs and top SNP (purple) (TIF 4876.9 kb)
13258_2021_1165_MOESM4_ESM.tif
Supplementary material 4 Regional plots of significant Loci associated with serum TGA level. Chromosomes are shown on the x-axis, and the − log10 p value on the y-axis. The color of each SNP spot, ranging from red to blue, reflects the degree of linkage disequilibrium (r2) with between other SNPs and top SNP (purple) (TIF 4749.0 kb)
13258_2021_1165_MOESM5_ESM.tif
Supplementary material 5 Regional plots of significant Loci associated with serum TMA level. Chromosomes are shown on the x-axis, and the − log10 p value on the y-axis. The color of each SNP spot, ranging from red to blue, reflects the degree of linkage disequilibrium (r2) with between other SNPs and top SNP (purple) (TIF 4628.3 kb)
13258_2021_1165_MOESM6_ESM.tif
Supplementary material 6 Regional plots of significant Loci associated with serum TSH level. Chromosomes are shown on the x-axis, and the − log10 p value on the y-axis. The color of each SNP spot, ranging from red to blue, reflects the degree of linkage disequilibrium (r2) with between other SNPs and top SNP (purple) (TIF 2230.7 kb)
Rights and permissions
About this article
Cite this article
Huang, L., Bai, F., Zhang, Y. et al. Preliminary study of genome-wide association identified novel susceptibility genes for thyroid-related hormones in Chinese population. Genes Genom 44, 1031–1038 (2022). https://doi.org/10.1007/s13258-021-01165-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-021-01165-1