Abstract
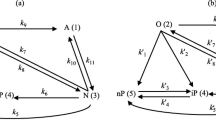
In the current research, a mathematical model is developed to simulate a commercial ethylene dichloride thermal cracker assuming pseudo-steady state condition. The considered reaction network includes 19 elementary and reversible reactions, 14 molecules, and 6 radical species. To prove the validity of the proposed model, the simulation results are compared with the available plant data. The results show that the coke build-up and deposition on the inner surface of the coil tube increases the heat transfer resistance during the process run time and has a negative effect on the production rate and pressure drop. Afterward, a sensitivity analysis is performed to investigate the effects of the feed and firebox temperatures on the ethylene dichloride cracking severity and selectivity. The results show that increasing the firebox and feed temperatures enhance the vinyl chloride monomer production rate. In the next step, a dynamic optimization problem is formulated and considered to calculate the optimal dynamic trajectory of the feed and furnace temperatures to achieve the maximum vinyl chloride monomer production rate. The programmed optimization problem is solved by the genetic algorithm method. The results show that applying the optimal trajectory of the decision variables can enhance the VCM production rate by 2.5%.









Similar content being viewed by others
Abbreviations
- Ac :
-
Surface area (m2)
- CP :
-
Specific heat capacity (J mol−1 K−1)
- Dt :
-
Coil diameter (m)
- Ft :
-
Total flow rate (mol s−1)
- Fr:
-
Friction factor
- ff,w :
-
Shape coefficient between tube and radiation chamber
- G:
-
Mass flux (kg m−2 s−1)
- g :
-
Acceleration of gravity (m s−2)
- h:
-
Heat transfer coefficient (W m−2 K−1)
- J:
-
Objective function
- ∆Hj :
-
Heat of reaction j (J mol−1)
- k:
-
Thermal conductivity coefficient (W m−1 K−1)
- Mc :
-
Coke molecular weight (g mol−1)
- Mwi :
-
Molecular weight of component i (g mol−1)
- P:
-
Total pressure (bar)
- Q(z):
-
Heat transfer rate (J)
- ri :
-
Rate of reaction (mol m−3 h−1)
- \({\mathrm{r}}_{c}\) :
-
Rate of coke formation reaction (mol m−3 s−1)
- R:
-
Universal gas constant (m3 Pa K−1 mol−1)
- Rb :
-
Tube curvature radius
- Ri :
-
Heat transfer resistance
- Re:
-
Reynolds number
- T:
-
Temperature (K)
- U:
-
Overall heat transfer coefficient (W m−2 K−1)
- ug :
-
Gas velocity (m s−1)
- yi :
-
Mole fraction of component i in gas phase
- z:
-
Axial reactor coordinate (m)
- Z:
-
Compressibility factor, dimensionless
- ρg :
-
Density of gas phase (kg m−3)
- \({\uprho }_{c}\) :
-
Coke density (kg m−3)
- \(\upxi\) :
-
Tube curvature parameter
- \(\varepsilon\) :
-
Emissivity factor
- \(\omega\) :
-
Weight of objectives
- \(\Lambda\) :
-
Tube curvature angle
- \(\upsilon _{{\text{i}}}\) :
-
Stoichiometric coefficient of component i
References
Nyeng I (2008) Modeling of a 1, 2-dichloroethane cracker. Master Tgesis in NTNU, Trondheim
Zhang Y, Hu G, Du W, Qian F (2016) Steam cracking and EDC furnace simulation. Adv Chem Eng 49:199–272
Mark HF, Kroschwitz JI (1985) Encyclopedia of polymer science and engineering. Pergamon Press, New York
Schirmeister R, Kahsnitz J, Träger M (2009) Influence of EDC cracking severity on the marginal costs of vinyl chloride production. Ind Eng Chem Res 48(6):2801–2809
Moradi Z, Farsi M (2020) Simulation of direct chlorination of ethylene in a two-phase reactor by coupling equilibrium, kinetic and population balance models. Chem Prod Process Model. https://doi.org/10.1515/cppm-2020-0061
Borsa AG, Herring AM, McKinnon JT, McCormick RL, Ko GH (2001) Coke and byproduct formation during 1,2-dichloroethane pyrolysis in a laboratory tubular reactor. Ind Eng Chem Res 40(11):2428–2436
Flid M, Trushechkina M, Treger Y (2017) Theoretical and applied aspects of 1,2-dichloroethane pyrolysis. J Thermodyn Catal 8(189):2–18
Borsa AG (1999) Industrial plant/laboratory investigation and computer modeling of 1,2-dichloroethane pyrolysis. Doctoral dissertations, Colorado
Mochida I, Tsunawaki T, Sotowa C, Korai Y, Higuchi K (1996) Coke produced in the commercial pyrolysis of ethylene dichloride into vinyl chloride. Ind Eng Chem Res 35(10):3803–3807
Choi B-S, Oh JS, Lee S-W, Kim H, Yi J (2001) Simulation of the effects of CCl4 on the ethylene dichloride pyrolysis process. Ind Eng Chem Res 40(19):4040–4049
Oliveira TCLd, Pereira Neto AT, Alves JJN, Morais Júnior AA (2017) CFD simulation of an industrial reactor for thermal cracking of 1, 2-dichloroethane. Braz J Chem Eng 34(2):541–555
Lee KY (2002) Numerical simulations of the pyrolysis of 1, 2 dichloroethane. KSME Int J 16(1):102–108
Fahiminezhad A, Peyghambarzadeh SM, Rezaeimanesh M (2020) Numerical modelling and industrial verification of ethylene dichloride cracking furnace. J Chem Pet Eng 54(2):165–185
Li C, Hu G, Zhong W, He W, Du W, Qian F (2013) Coke deposition influence based on a run length simulation of a 1, 2-dichloroethane cracker. Ind Eng Chem Res 52(49):17501–17516
Czechorski TJ (2019) A kinetic and thermodynamic model of ethylene dichloride pyrolysis. Master's Thesis in University of Louisville, Kentucky
Mousavipour S, Saheb V, Pirhadi F, Dehbozorgi M (2007) Experimental and theoretical study on the kinetics and mechanism of thermal decomposition of 1, 2-dichloroethane. J Iran Chem Soc 4(3):279–298
Lakshmanan A, Rooney WC, Biegler LT (1999) A case study for reactor network synthesis: the vinyl chloride process. Comput Chem Eng 23:479–495
Alam I, West DH, Balakotaiah V (2016) Transport effects on pattern formation and maximum temperature in homogeneous–heterogeneous combustion. Chem Eng J 288:99–115
Abbasali SZ, Farsi M, Rahimpour M (2017) Simulation and dynamic optimization of an industrial naphtha thermal cracking furnace based on time variant feeding policy. Chem Prod Process Model. https://doi.org/10.1515/cppm-2017-0032
Rice RG, Do DD (2012) Applied mathematics and modeling for chemical engineers. Wiley, New York
Rao SS (2019) Engineering optimization: theory and practice. Wiley3, New York
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Raoof, M., Farsi, M. & Setoodeh, P. Kinetic modeling and dynamic optimization of a commercial dichloroethane thermal cracker. Reac Kinet Mech Cat 134, 331–346 (2021). https://doi.org/10.1007/s11144-021-02069-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-021-02069-7