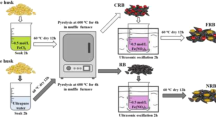
Abstract
The presence of arsenic (As) and lead (Pb) in water above tolerable levels is a global concern, as both elements are extremely toxic and can compromise the health of millions of people. In this study, we evaluated biochars pyrolyzed at 350 (BC350) and 750 °C (BC750) and then modified with ferric chloride (FeCl3) to remove As and Pb from water samples contaminated by mining activity. The use of FeCl3-modified biochar was an efficient strategy to remove As and Pb from water. Sorption on biochars followed the pseudo-second-order kinetics, represented by two sorption speeds, being higher at the beginning of the reaction and falling over time. The modification with FeCl3 increased the specific surface area, removed the dissolved organic carbon, and increased the capacity of BC350 to sorb Pb. These physical and chemical alterations of the biochar played a significant role in the sorption capacity and speed of As and Pb.






Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Adegbeye, M. J., Salem, A. Z. M., Reddy, P. R. K., Elghandour, M. M. M., & Oyebamiji, K. J. (2020). Waste recycling for the eco-friendly input use efficiency in agriculture and livestock feeding, in: Resources use efficiency in agriculture. Springer Singapore, pp. 1–45. https://doi.org/10.1007/978-981-15-6953-1_1
Ahmad, M., Rajapaksha, A. U., Lim, J. E., Zhang, M., Bolan, N., Mohan, D., Vithanage, M., Lee, S. S., & Ok, Y. S. (2014). Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere. https://doi.org/10.1016/j.chemosphere.2013.10.071
Ahmad, M., Usman, A. R. A., Hussain, Q., Al-Farraj, A. S. F., Tsang, Y. F., Bundschuh, J., & Al-Wabel, M. I. (2020). Fabrication and evaluation of silica embedded and zerovalent iron composited biochars for arsenate removal from water. Environmental Pollution, 266, 115256. https://doi.org/10.1016/j.envpol.2020.115256
Ahmed, M. B., Zhou, J. L., Ngo, H. H., Guo, W., & Chen, M. (2016). Progress in the preparation and application of modified biochar for improved contaminant removal from water and wastewater. Bioresource Technology, 214, 836–851. https://doi.org/10.1016/j.biortech.2016.05.057
ASTM (2007). ASTM D1762–84 - Standard Test Method for Chemical Analysis of Wood. https://doi.org/10.1520/D1762-84R13
Bakshi, S., Banik, C., Rathke, S. J., & Laird, D. A. (2018). Arsenic sorption on zero-valent iron-biochar complexes. Water Research, 137, 153–163. https://doi.org/10.1016/j.watres.2018.03.021
Barreto, M. S. C., Elzinga, E. J., & Alleoni, L. R. F. (2020). Hausmannite as potential As(V) filter. Macroscopic and spectroscopic study of As(V) adsorption and desorption by citric acid. Environmental Pollution, 262, 114196. https://doi.org/10.1016/j.envpol.2020.114196
Beibei, Z., Jinbang, L., & Quanjiu, W. (2015). Chloride transport in an aggregated clay soil column. Journal of Hydrologic Engineering, 20, 04015032. https://doi.org/10.1061/(asce)he.1943-5584.0001227
Boim, A. G. F., Patinha, C., Wragg, J., Cave, M., & Alleoni, L. R. F. (2020). Respiratory bioaccessibility and solid phase partitioning of potentially harmful elements in urban environmental matrices. Science of the Total Environment, 765, 142791. https://doi.org/10.1016/j.scitotenv.2020.142791
CETESB. (2016). Valores orientadores para solo e água subterrânea de São Paulo [WWW Document]. URL https://cetesb.sp.gov.br/aguas-subterraneas/valores-orientadores-para-solo-e-agua-subterranea/. Accessed 19–21 Jan 2021.
Chakraborty, S., Alam, M. O., Bhattacharya, T., & Singh, Y. N. (2014). Arsenic accumulation in food crops: A potential threat in Bengal Delta Plain. Water Quality Exposure and Health, 6, 233–246. https://doi.org/10.1007/s12403-014-0122-x
Chisolm, J. J., & Harisson, H. E. (1998). Home management of acute respiratory infections: A challenge to the family and the community. International Journal of Sociology & Social Policy, 18, 102–127. https://doi.org/10.1108/01443339810788489
Christie, D. (2004). Resampling with Excel. Teaching Statistics, 26, 9–14. https://doi.org/10.1111/j.1467-9639.2004.00136.x
CONAMA (2009) Resolução No 420 sobre critérios e valores orientadores da qualidade do solo e água subterrâneas [WWW Document]
Dougherty, D. A. (2013). The cation-π interaction. Accounts of Chemical Research, 46, 885–893. https://doi.org/10.1021/ar300265y
Essington, M. E. (2003). Soil and water chemistry: An integrative approach (1st ed.). CRC Press.
Hale, S. E., Nurida, N. L., Jubaedah, Mulder, J., Sørmo, E., Silvani, L., Abiven, S., Joseph, S., Taherymoosavi, S., & Cornelissen, G. (2020). The effect of biochar, lime and ash on maize yield in a long-term field trial in a Ultisol in the humid tropics. Science of the Total Environment, 719, 137455. https://doi.org/10.1016/j.scitotenv.2020.137455
He, X.-S., Xi, B.-D., Wei, Z.-M., Jiang, Y.-H., Geng, C.-M., Yang, Y., Yuan, Y., & Liu, H.-L. (2011). Physicochemical and spectroscopic characteristics of dissolved organic matter extracted from municipal solid waste (MSW) and their influence on the landfill biological stability. Bioresource Technology, 102, 2322–2327. https://doi.org/10.1016/J.BIORTECH.2010.10.085
He, J., Strezov, V., Zhou, X., Kumar, R., & Kan, T. (2020). Pyrolysis of heavy metal contaminated biomass pre-treated with ferric salts: Product characterisation and heavy metal deportment. Bioresource Technology, 313, 123641. https://doi.org/10.1016/j.biortech.2020.123641
Hu, X., & Gholizadeh, M. (2019). Biomass pyrolysis: A review of the process development and challenges from initial researches up to the commercialisation stage. Journal of Energy Chemistry. https://doi.org/10.1016/j.jechem.2019.01.024
Hu, X., Ding, Z., Zimmerman, A. R., Wang, S., & Gao, B. (2015). Batch and column sorption of arsenic onto iron-impregnated biochar synthesized through hydrolysis. Water Research, 68, 206–216. https://doi.org/10.1016/j.watres.2014.10.009
Huang, M., Li, Z., Chen, M., Wen, J., Luo, N., Xu, W., Ding, X., & Xing, W. (2020). Dissolved organic matter released from rice straw and straw biochar: Contrasting molecular composition and lead binding behaviors. Science of the Total Environment, 739, 140378. https://doi.org/10.1016/j.scitotenv.2020.140378
Inyang, M. I., Gao, B., Yao, Y., Xue, Y., Zimmerman, A., Mosa, A., Pullammanappallil, P., Ok, Y. S., & Cao, X. (2016). A review of biochar as a low-cost adsorbent for aqueous heavy metal removal. Critical Reviews in Environment Science and Technology, 46, 406–433. https://doi.org/10.1080/10643389.2015.1096880
Johs, A., Eller, V. A., Mehlhorn, T. L., Brooks, S. C., Harper, D. P., Mayes, M. A., Pierce, E. M., & Peterson, M. J. (2019). Dissolved organic matter reduces the effectiveness of sorbents for mercury removal. Science of the Total Environment, 690, 410–416. https://doi.org/10.1016/j.scitotenv.2019.07.001
Karim, M. M. (2000). Arsenic in groundwater and health problems in Bangladesh. Water Research, 34, 304–310. https://doi.org/10.1016/S0043-1354(99)00128-1
Kottek, M., Grieser, J., Beck, C., Rudolf, B., & Rubel, F. (2006). World Map of the Köppen-Geiger Climate Classification Updated, 15, 259–263. https://doi.org/10.1127/0941-2948/2006/0130
Lado, L. R., Hengl, T., & Reuter, H. I. (2008). Heavy metals in European soils: A geostatistical analysis of the FOREGS Geochemical database. Geoderma, 148, 189–199. https://doi.org/10.1016/j.geoderma.2008.09.020
Langmuir, D. (1979). Techniques of estimating thermodynamic properties for some aqueous complexes of geochemical interest, in: Chemical modeling in aqueous systems. Am Chem Soc, pp. 353–387. https://doi.org/10.1021/bk-1979-0093.ch018
Li, H., Dong, X., da Silva, E. B., de Oliveira, L. M., Chen, Y., & Ma, L. Q. (2017). Mechanisms of metal sorption by biochars: Biochar characteristics and modifications. Chemosphere, 178, 466–478. https://doi.org/10.1016/J.CHEMOSPHERE.2017.03.072
Li, G., Khan, S., Ibrahim, M., Sun, T. R., Tang, J. F., Cotner, J. B., & Xu, Y. Y. (2018). Biochars induced modification of dissolved organic matter (DOM) in soil and its impact on mobility and bioaccumulation of arsenic and cadmium. Journal of Hazardous Materials, 348, 100–108. https://doi.org/10.1016/j.jhazmat.2018.01.031
Li, S., Zeng, W., Jia, Z., Wu, G., Xu, H., & Peng, Y. (2020). Phosphorus species transformation and recovery without apatite in FeCl3-assisted sewage sludge hydrothermal treatment. Chemical Engineering Journal, 399, 125735. https://doi.org/10.1016/J.CEJ.2020.125735
Maneechakr, P., & Mongkollertlop, S. (2020). Investigation on adsorption behaviors of heavy metal ions (Cd2+, Cr3+, Hg2+and Pb2+) through low-cost/active manganese dioxide-modified magnetic biochar derived from palm kernel cake residue. Journal of Environmental Chemical Engineering, 8, 104467. https://doi.org/10.1016/j.jece.2020.104467
Martinsen, V., Alling, V., Nurida, N. L., Mulder, J., Hale, S. E., Ritz, C., Rutherford, D. W., Heikens, A., Breedveld, G. D., & Cornelissen, G. (2015). pH effects of the addition of three biochars to acidic Indonesian mineral soils. Soil Science and Plant Nutrition, 61, 821–834. https://doi.org/10.1080/00380768.2015.1052985
Mólgora, C. C., Domínguez, A. M., Avila, E. M., Drogui, P., & Buelna, G. (2013). Removal of arsenic from drinking water: A comparative study between electrocoagulation-microfiltration and chemical coagulation-microfiltration processes. Separation and Purification Technology, 118, 645–651. https://doi.org/10.1016/j.seppur.2013.08.011
Obasi, P. N., & Akudinobi, B. B. (2020). Potential health risk and levels of heavy metals in water resources of lead–zinc mining communities of Abakaliki, southeast Nigeria. Applied Water Science, 10, 184. https://doi.org/10.1007/s13201-020-01233-z
Pan, J., Jiang, J., & Xu, R. (2013). Adsorption of Cr(III) from acidic solutions by crop straw derived biochars. Journal of Environmental Sciences (china), 25, 1957–1965. https://doi.org/10.1016/S1001-0742(12)60305-2
Panagos, P., Van Liedekerke, M., Jones, A., & Montanarella, L. (2012). European Soil Data Centre: Response to European policy support and public data requirements. Land Use Policy, 29, 329–338. https://doi.org/10.1016/j.landusepol.2011.07.003
Papanikolaou, N. C., Hatzidaki, E. G., Belivanis, S., Tzanakakis, G. N., & Tsatsakis, A. M. (2005). Lead toxicity update. A brief review. Medical Science Monitor, 11, RA329–RA336.
Park, J. H., Ok, Y. S., Kim, S. H., Cho, J. S., Heo, J. S., Delaune, R. D., & Seo, D. C. (2016). Competitive adsorption of heavy metals onto sesame straw biochar in aqueous solutions. Chemosphere, 142, 77–83. https://doi.org/10.1016/j.chemosphere.2015.05.093
Ren, J., Li, N., Wei, H., Li, A., & Yang, H. (2020). Efficient removal of phosphorus from turbid water using chemical sedimentation by FeCl3 in conjunction with a starch-based flocculant. Water Research, 170, 115361. https://doi.org/10.1016/J.WATRES.2019.115361
Saharan, P., Chaudhary, G. R., Mehta, S. K., & Umar, A. (2014). Removal of water contaminants by iron oxide nanomaterials. Journal of Nanoscience and Nanotechnology, 14, 627–643. https://doi.org/10.1166/jnn.2014.9053
Shaji, E., Santosh, M., Sarath, K. V., Prakash, P., Deepchand, V., & Divya, B. V. (2020). Arsenic contamination of groundwater: A global synopsis with focus on the Indian Peninsula. Geoscience Frontiers. https://doi.org/10.1016/j.gsf.2020.08.015
Singh, S. K., & Das, A. (2015). The n → π∗ interaction: A rapidly emerging non-covalent interaction. Physical Chemistry Chemical Physics: PCCP, 17, 9596–9612. https://doi.org/10.1039/c4cp05536e
Singh, B., Camps-Arbestain, M., & Lehmann, J. (2017). Biochar: A guide to analytical methods. Taylor and .ed. CSIRO.
Smebye, A., Alling, V., Vogt, R. D., Gadmar, T. C., Mulder, J., Cornelissen, G., & Hale, S. E. (2016). Biochar amendment to soil changes dissolved organic matter content and composition. Chemosphere, 142, 100–105. https://doi.org/10.1016/j.chemosphere.2015.04.087
Song, W., & Guo, M. (2012). Quality variations of poultry litter biochar generated at different pyrolysis temperatures. Journal of Analytical and Applied Pyrolysis, 94, 138–145. https://doi.org/10.1016/J.JAAP.2011.11.018
Soukup, D. A., Drees, L. R., & Lynn, W. C. (2008). Sampling soils for mineralogical analyses. In A. L. Ulery & L. R. Drees (Eds.), Methods of soil analysis part 5—Mineralogical methods (pp. 1–11). John Wiley & Sons Ltd. https://doi.org/10.2136/sssabookser5.5.c1
Sun, Y., Yu, I. K. M., Tsang, D. C. W., Cao, X., Lin, D., Wang, L., Graham, N. J. D., Alessi, D. S., Komárek, M., Ok, Y. S., Feng, Y., & Li, X. D. (2019). Multifunctional iron-biochar composites for the removal of potentially toxic elements, inherent cations, and hetero-chloride from hydraulic fracturing wastewater. Environment International, 124, 521–532. https://doi.org/10.1016/j.envint.2019.01.047
Tan, Z., Yuan, S., Hong, M., Zhang, L., & Huang, Q. (2020). Mechanism of negative surface charge formation on biochar and its effect on the fixation of soil Cd. Journal of Hazardous Materials, 384, 121370. https://doi.org/10.1016/j.jhazmat.2019.121370
Tomczyk, A., Sokołowska, Z., & Boguta, P. (2020). Biochar physicochemical properties: Pyrolysis temperature and feedstock kind effects. Reviews in Environmental Science & Biotechnology, 19, 191–215. https://doi.org/10.1007/s11157-020-09523-3
Tuna, A. Ö. A., Özdemir, E., Şimşek, E. B., & Beker, U. (2013). Removal of As(V) from aqueous solution by activated carbon-based hybrid adsorbents: Impact of experimental conditions. Chemical Engineering Journal, 223, 116–128. https://doi.org/10.1016/j.cej.2013.02.096
USEPA (2007). Method 3051A: Microwave assisted acid digestion of sediments, sludges, soils and oils, EPA. ed. Washington
USEPA. (2020). Regional Screening Levels (RSLs) - Generic tables [WWW Document]. URL https://www.epa.gov/risk/regional-screening-levels-rsls-generic-tables. Accessed 05–10 Feb 2021.
Wang, S., Gao, B., Zimmerman, A. R., Li, Y., Ma, L., Harris, W. G., & Migliaccio, K. W. (2015). Removal of arsenic by magnetic biochar prepared from pinewood and natural hematite. Bioresource Technology, 175, 391–395. https://doi.org/10.1016/j.biortech.2014.10.104
Wang, D., Jiang, P., Zhang, H., & Yuan, W. (2020). Biochar production and applications in agro and forestry systems: A review. Science of the Total Environment, 723, 137775. https://doi.org/10.1016/j.scitotenv.2020.137775
Wu, C., Cui, M. Q., Xue, S. G., Li, W. C., Huang, L., Jiang, X. X., & Qian, Z. Y. (2018). Remediation of arsenic-contaminated paddy soil by iron-modified biochar. Environmental Science and Pollution Research, 25, 20792–20801. https://doi.org/10.1007/s11356-018-2268-8
Wu, L., Zhang, S., Wang, J., & Ding, X. (2020). Phosphorus retention using iron (II/III) modified biochar in saline-alkaline soils: Adsorption, column and field tests. Environmental Pollution, 261, 114223. https://doi.org/10.1016/j.envpol.2020.114223
Xing, J., Li, L., Li, G., & Xu, G. (2019). Feasibility of sludge-based biochar for soil remediation: Characteristics and safety performance of heavy metals influenced by pyrolysis temperatures. Ecotoxicology and Environmental Safety, 180, 457–465. https://doi.org/10.1016/J.ECOENV.2019.05.034
Xu, L., Fu, B., Sun, Y., Jin, P., Bai, X., Jin, X., Shi, X., Wang, Y., & Nie, S. (2020). Degradation of organic pollutants by Fe/N co-doped biochar via peroxymonosulfate activation: Synthesis, performance, mechanism and its potential for practical application. Chemical Engineering Journal, 400, 125870. https://doi.org/10.1016/j.cej.2020.125870
Yang, F., Zhao, L., Gao, B., Xu, X., & Cao, X. (2016). The interfacial behavior between biochar and soil minerals and its effect on biochar stability. Environmental Science and Technology, 50, 2264–2271. https://doi.org/10.1021/acs.est.5b03656
Yoon, G., Park, S.-M., Yang, H., Tsang, D. C. W., Alessi, D. S., & Baek, K. (2018). Selection criteria for oxidation method in total organic carbon measurement. Chemosphere, 199, 453–458. https://doi.org/10.1016/J.CHEMOSPHERE.2018.02.074
Zhang, Q., & Wang, C. (2020). Natural and human factors affect the distribution of soil heavy metal pollution: A review. Water, Air, and Soil Pollution, 231, 1–13. https://doi.org/10.1007/s11270-020-04728-2
Zhang, M., Gao, B., Varnoosfaderani, S., Hebard, A., Yao, Y., & Inyang, M. (2013). Preparation and characterization of a novel magnetic biochar for arsenic removal. Bioresource Technology, 130, 457–462. https://doi.org/10.1016/j.biortech.2012.11.132
Zhang, M. M., Liu, Y. G., Li, T. T., Xu, W. H., Zheng, B. H., Tan, X. F., Wang, H., Guo, Y. M., Guo, F. Y., & Wang, S. F. (2015). Chitosan modification of magnetic biochar produced from Eichhornia crassipes for enhanced sorption of Cr(VI) from aqueous solution. RSC Advances, 5, 46955–46964. https://doi.org/10.1039/c5ra02388b
Zhao, S. X., Ta, N., & Wang, X. D. (2017). Effect of temperature on the structural and physicochemical properties of biochar with apple tree branches as feedstock material. Energies, 10, 1293. https://doi.org/10.3390/en10091293
Zhu, J., Cozzolino, V., Pigna, M., Huang, Q., Caporale, A. G., & Violante, A. (2011). Sorption of Cu, Pb and Cr on Na-montmorillonite: Competition and effect of major elements. Chemosphere, 84, 484–489. https://doi.org/10.1016/j.chemosphere.2011.03.020
Acknowledgements
The 1st author gratefully thanks the São Paulo Research Foundation (FAPESP) (grant #2019/06897-9) and the Brazilian Council for Scientific and Technological Development—Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (grant #140830/2018-9) for the scholarships granted for this research. The 2nd author thanks CNPq (grant#132590/2019-0) for the scholarship granted for this research. The 3rd author thanks CNPq (grant #306429/2018-7) for the scholarship granted for this research. This study was partially funded by CNPq and the Coordination for the Improvement of Higher Education Personnel—Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) – Finance Code 001.
Author information
Authors and Affiliations
Contributions
MBS: conceived and designed the analyses, performed the formal analysis, wrote the manuscript. FHS: contributed analysis tools, contributed to sample preparation, revised the manuscript. LRFA: acquisition of financing, conceived and designed the analyses, encouraged MBS and FHS to investigate biochars from sugarcane straw to remove As and Pb from water samples contaminated by mining activity, supervised the findings of this research, revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Soares, M.B., dos Santos, F.H. & Alleoni, L.R.F. Iron-Modified Biochar from Sugarcane Straw to Remove Arsenic and Lead from Contaminated Water. Water Air Soil Pollut 232, 391 (2021). https://doi.org/10.1007/s11270-021-05343-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-021-05343-5