Abstract
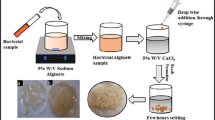
In the present work, the biodegradation of phenol by alginate immobilized Chlamydomonas reinhardtii cells was investigated. Immobilized Chlamydomonas reinhardtii could remove up to 1300 μmol/L of phenol within 10 days of cultivation. Metabolic activity was demonstrated by the extracellular release of catechol. Beads prepared at high concentrations of alginate (5–6% w/v) were found to protect microalgae against the strong inhibitory effects of phenol on the photosynthetic apparatus. Cells immobilized in beads of higher concentrations of alginate exhibited higher metabolic efficiencies compared to those prepared by lower alginate concentrations. Lower alginate concentrations (3–4% w/v) led to increased cell leakage, while the presence of phenol in the medium had the opposite effect in all alginate concentrations. Resuspension of immobilized microalgae in a medium containing a growth-promoting substrate, led to colony formation only on the external surface of alginate beads, indicating that acetic acid and consequently phenol, could not penetrate the internal of alginate beads. The significance of the work is that alginate immobilized Chlamydomonas substantially minimize the required volume of the aqueous medium and improve the economics and commercial application prospects of the process.









Similar content being viewed by others
References
Bailliez C, Largeau C, Casadevall E (1985) Growth and hydrocarbon production of Botryococcus braunii immobilized in calcium alginate gel. Appl Microb Biot 23(2):99–105. https://doi.org/10.1007/bf01982724
Bailliez C, Largeau C, Berkaloff C, Casadevall E (1986) Immobilization of Botryococcus braunii in alginate: influence on chlorophyll content, photosynthetic activity and degeneration during batch cultures. Appl Microb Biot 23(5):361–366. https://doi.org/10.1007/bf00257033
Canbay E, Kose A, Oncel SS (2018) Photobiological hydrogen production via immobilization: understanding the nature of the immobilization and investigation on various conventional photobioreactors. 3 Biotech 8(5):244. https://doi.org/10.1007/s13205-018-1266-3
Castro-Ceseña AB, del Pilar S-S (2015) Evaluation of sodium tripolyphosphate-alginate coating and re-calcifying on the entrapment of microalgae in alginate beads. J Appl Phycol 27(3):1205–1212. https://doi.org/10.1007/s10811-014-0411-6
Chu WL, See YC, Phang SM (2008) Use of immobilised Chlorella vulgaris for the removal of colour from textile dyes. J Appl Phycol 21(6):641–648. https://doi.org/10.1007/s10811-008-9396-3
De Vos P, Lazarjani HA, Poncelet D, Faas MM (2014) Polymers in cell encapsulation from an enveloped cell perspective. Adv Drug Deliver Rev 67–68:15–34. https://doi.org/10.1016/j.addr.2013.11.005
De-Bashan LE, Bashan Y (2010) Immobilized microalgae for removing pollutants: review of practical aspects. Bioresource Technol 101(6):1611–1627. https://doi.org/10.1016/j.biortech.2009.09.043
Donot F, Fontana A, Baccou JC, Schorr-Galindo S (2012) Microbial exopolysaccharides: Main examples of synthesis, excretion, genetics and extraction. Carbohyd Polym 87(2):951–962. https://doi.org/10.1016/j.carbpol.2011.08.083
Duarte JC, Rodrigues JA, Moran PJ, Valenca GP, Nunhez JR (2013) Effect of immobilized cells in calcium alginate beads in alcoholic fermentation. AMB Express 3(1):31. https://doi.org/10.1186/2191-0855-3-31
Eroglu E, Smith SM, Raston CL (2015) Application of various immobilization techniques for algal bioprocesses. In: Moheimani NR, McHenry MP, de Boer K, Bahri PA (eds) Biomass and Biofuels from Microalgae: Advances in Engineering and Biology. Springer International Publishing, Cham, pp 19–44. https://doi.org/10.1007/978-3-319-16640-7_2
Forootanfar H, Shakibaie M, Bagherzadeh Z, Aghaie-Khozani M, Nafissi-Varcheh N, Monsef-Esfahani HR, Faramarzi MA (2013) The removal of ρ-chlorophenol in aqueous cultures with free and alginate-immobilized cells of the microalga Tetraselmis suecica. J Appl Phycol 25(1):51–57. https://doi.org/10.1007/s10811-012-9837-x
Harris E (2009) The Chlamydomonas sourcebook: Introduction to Chlamydomonas and its laboratory use. In: The Chlamydomonas sourcebook, vol I. 2nd edn. Academic Press, Canada
Jaromir M, Ozadowicz R, Duda W (2005) Analysis of chlorophenols, chlorocatechols, chlorinated methoxyphenols and monoterpenes in communal sewage of Lodz and in the Ner river n 1999–2000. Water Air Soil Pollut 164(1–4):205–222
Johnson X, Alric J (2013) Central carbon metabolism and electron transport in Chlamydomonas reinhardtii: metabolic constraints for carbon partitioning between oil and starch. Eukaryot Cell 12(6):776–793. https://doi.org/10.1128/EC.00318-12
Keweloh H, Heipieper H-J, Rehm H-J (1989) Protection of bacteria against toxicity of phenol by immobilization in calcium alginate. Appl Microb Biot 31(4):383–389. https://doi.org/10.1007/bf00257609
León R, Pizzano JA, Galván F (1996) Studies of cell viability of immobilized Chlamydomonas reinhardtii and glycerol photoproduction. In: Wijffels RH, Buitelaar RM, Bucke C, Tramper J (eds) Progress in Biotechnology, vol 11. Elsevier, pp 221–228. https://doi.org/10.1016/S0921-0423(96)80031-1
Matsubara Y, Kawada R, Iwasaki K-I, Kimura Y, Oda T, Muramatsu T (2000) Cloning and sequence analysis of a gene (aly PG) encoding poly(α-l-guluronate)lyase from Corynebacterium sp. strain ALY-1. J Biosci Bioeng 89(2):199–202. https://doi.org/10.1016/S1389-1723(00)88738-1
Meera G, Abraham TE (2006) Polyionic hydrocolloids for the intestinal delivery of protein drugs: Alginate and chitosan - a review. J Control Release 114(1):1–14. https://doi.org/10.1016/j.jconrel.2006.04.017
Michalowicz J, Duda W (2007) Phenols – Sources and Toxicity. Pol J Environ Stud 16(3):347–362
Moreno-Garrido I (2008) Microalgae immobilization: current techniques and uses. Biores Technol 99(10):3949–3964. https://doi.org/10.1016/j.biortech.2007.05.040
Morita H (1975) Calcium binding to catechols related to organic soil compounds. Soil Sci 120(4):261–263
Nazos TT, Kokarakis EJ, Ghanotakis DF (2017) Metabolism of xenobiotics by Chlamydomonas reinhardtii: Phenol degradation under conditions affecting photosynthesis. Photosynth Res 131(1):31–40. https://doi.org/10.1007/s11120-016-0294-2
Nazos TT, Mavroudakis L, Pergantis SA, Ghanotakis DF (2020a) Biodegradation of phenol by Chlamydomonas reinhardtii. Photosynth Res 144:383–395. https://doi.org/10.1007/s11120-020-00756-5
Nazos TT, Kokarakis EJ, Valsami EA, Stratigakis NC, Poloniataki EG, Sfendourakis GP, Ghanotakis DF (2020) Characterization of a novel herbicide and antibiotic-resistant Chlorella sp. with an extensive extracellular matrix. Photosynth Res 143(3):315–334. https://doi.org/10.1007/s11120-020-00710-5
Østgaard K, Knutsen SH, Dyrset N, Aasen IM (1993) Production and characterization of guluronate lyase from Klebsiella pneumoniae for applications in seaweed biotechnology. Enzyme Microb Tech 15(9):756–763. https://doi.org/10.1016/0141-0229(93)90006-N
Paredes JGA, Spasojevic M, Faas MM, De Vos P (2014) Immunological and technical considerations in application of alginate-based microencapsulation systems. Front Bioeng Biotechnol 2(26):1–15. https://doi.org/10.3389/fbioe.2014.00026
Ruiz-Marin A, Mendoza-Espinosa LG, Stephenson T (2010) Growth and nutrient removal in free and immobilized green algae in batch and semi-continuous cultures treating real wastewater. Biores Technol 101(1):58–64. https://doi.org/10.1016/j.biortech.2009.02.076
Santos-Rosa F, Galván F (1989) Ammonium photoproduction by free and immobilized cells of Chlamydomonas reinhardtii. Appl Microbiol Biot 31(1):55–58. https://doi.org/10.1007/bf00252526
Santos-Rosa F, Galván F, Vega JM (1989) Biological viability of Chlamydomonas reinhardtii cells entrapped in alginate beads for ammonium photoproduction. J Biotechnol 9(3):209–219. https://doi.org/10.1016/0168-1656(89)90109-0
Shapiro JA (1985) Scanning electron microscope study of Pseudomonas putida colonies. J Bacteriol 164(3):1171–1181
Shapiro JA (1987) Organization of developing Escherichia coli colonies viewed by scanning electron microscopy. J Bacteriol 169(1):142–156. https://doi.org/10.1128/jb.169.1.142-156.1987
Strasser BJ, Strasser RJ (1995) Measuring fast fluorescence transients to address environmental questions: the JIP-test. In: Mathis P (ed) Photosynthesis: From Light to Biosphere. Kluwer Academic Press, Dordrecht
Sun J, Tan H (2013) Alginate-based biomaterials for regenerative medicine applications. Materials 6(4):1285–1309. https://doi.org/10.3390/ma6041285
Tannin-Spitz T, Bergman M, Van-Moppes D, Grossman S, Arad S (2005) Antioxidant activity of the polysaccharide of the red microalga Porphyridium sp. J Appl Phycol 17(3):215–222. https://doi.org/10.1007/s10811-005-0679-7
Touloupakis E, Rontogiannis G, Silva Benavides AM, Cicchi B, Ghanotakis DF, Torzillo G (2016) Hydrogen production by immobilized Synechocystis sp. PCC 6803. Int J Hydrogen Energy 41(34):15181–15186. https://doi.org/10.1016/j.ijhydene.2016.07.075
Uemoto H, Saiki H (2000) Distribution of Nitrosomonas europaea and Paracoccus denitrificans immobilized in tubular polymeric gel for nitrogen removal. Appl Environ Microbiol 66(2):816–819. https://doi.org/10.1128/aem.66.2.816-819.2000
Valsami EA, Pateraki A, Melis A, Ghanotakis DF (2021) Heterologous β-phellandrene production by alginate immobilized Synechocystis sp. PCC 6803. J Appl Phycol. https://doi.org/10.1007/s10811-021-02451-x
Vidali M (2001) Bioremediation. An overview. Pure Appl Chem 73(7):1163–1172
Vílchez MJ, Vigara J, Garbayo I, Vílchez C (1997) Electron microscopic studies on immobilized growing Chlamydomonas reinhardtii cells. Enzyme MicrobTech 21(1):45–47. https://doi.org/10.1016/S0141-0229(96)00223-2
Wee S, Gombotz WR (1998) Protein release from alginate matrices. Adv Drug Deliv Rev 31(3):267–285. https://doi.org/10.1016/s0169-409x(97)00124-5
Xie B, Gong W, Yu H, Tang X, Yan Z, Luo X, Gan Z, Wang T, Li G, Liang H (2018) Immobilized microalgae for anaerobic digestion effluent treatment in a photobioreactor-ultrafiltration system: Algal harvest and membrane fouling control. Biores Technol 268:139–148. https://doi.org/10.1016/j.biortech.2018.07.110
Zhang E, Wang B, Wang Q, Zhang S, Zhao B (2008) Ammonia–nitrogen and orthophosphate removal by immobilized Scenedesmus sp. isolated from municipal wastewater for potential use in tertiary treatment. Bioresour Techol 99(9):3787–3793. https://doi.org/10.1016/j.biortech.2007.07.011
Zur J, Wojcieszynska D, Guzik U (2016) Metabolic responses of bacterial cells to immobilization. Molecules. https://doi.org/10.3390/molecules21070958
Acknowledgements
The authors would like to thank Mr. Stefanos Papadakis from the Electron Microscopy Laboratory “Vasileios Galanopoulos” at the University of Crete for his assistance in the preparation and observation of alginate beads samples by Scanning Electron Microscopy.
Funding
This research was co-financed by Greece and the European Union (European Social Fund—ESF) through the Operational Programme “Human Resources Development, Education and Lifelong Learning” in the context of the project “Scholarships programme for post-graduate studies -2nd Study Cycle” (MIS-5003404), implemented by the State Scholarships Foundation (IKY).
Author information
Authors and Affiliations
Contributions
TN: Conceptualization, methodology, investigation, validation, visualization, writing—original draft, writing—review and editing; DG: conceptualization, validation, writing—review and editing, supervision, project administration.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Human and animal rights statement
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent for publication
Both the authors read and approved the manuscript and agreed for its submission in Archives of Microbiology.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nazos, T.T., Ghanotakis, D.F. Biodegradation of phenol by alginate immobilized Chlamydomonas reinhardtii cells. Arch Microbiol 203, 5805–5816 (2021). https://doi.org/10.1007/s00203-021-02570-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-021-02570-6