Abstract
Fracture risk is increased in patients with type 2 diabetes mellitus (T2DM). In addition, these patients sustain fractures despite having higher levels of areal bone mineral density, as measured by dual-energy X-ray absorptiometry, than individuals without T2DM. Thus, additional factors such as alterations in bone quality could have important roles in mediating skeletal fragility in patients with T2DM. Although the pathogenesis of increased fracture risk in T2DM is multifactorial, impairments in bone material properties and increases in cortical porosity have emerged as two key skeletal abnormalities that contribute to skeletal fragility in patients with T2DM. In addition, indices of bone formation are uniformly reduced in patients with T2DM, with evidence from mouse studies published over the past few years linking this abnormality to accelerated skeletal ageing, specifically cellular senescence. In this Review, we highlight the latest advances in our understanding of the mechanisms of skeletal fragility in patients with T2DM and suggest potential novel therapeutic approaches to address this problem.
Key points
-
Fracture risk is increased in patients with type 2 diabetes mellitus (T2DM) despite normal, or even increased, bone mineral density.
-
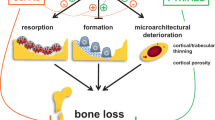
Clinical studies have revealed that the two most consistent alterations in bone quality in patients with T2DM are impaired bone material properties and increased cortical porosity.
-
These abnormalities seem to be linked, at least in part, to accumulation of advanced glycation end products (leading to impaired bone material properties) and microvascular disease (leading to increased cortical porosity).
-
Evidence from the past few years also indicates that T2DM, at least in mice, is associated with accelerated skeletal ageing and increased accumulation of senescent cells, in bone as well as in other tissues.
-
Current strategies for fracture prevention in patients with T2DM include minimizing exposure to diabetes mellitus drugs that increase fracture risk and use of osteoporosis medications shown to be effective in patients without diabetes mellitus.
-
Further studies are needed to evaluate the efficacy of osteoporosis medications specifically in patients with T2DM and to develop new drugs targeting the mechanisms potentially driving skeletal fragility in patients with T2DM.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Zimmet, P. Z. Diabetes and its drivers: the largest epidemic in human history? Clin. Diabetes Endocrinol. 3, 1 (2017).
Napoli, N. et al. Mechanisms of diabetes mellitus-induced bone fragility. Nat. Rev. Endocrinol. 13, 208–219 (2017). A comprehensive review of the mechanisms of bone fragility in T2DM; the current review aims largely to update new information since this publication.
Vestergaard, P. Discrepancies in bone mineral density and fracture risk in patients with type 1 and type 2 diabetes–a meta-analysis. Osteoporos. Int. 18, 427–444 (2007).
Schwartz, A. V. et al. Association of BMD and FRAX score with risk of fracture in older adults with type 2 diabetes. JAMA 305, 2184–2192 (2011). A key paper demonstrating that patients with diabetes mellitus experience fractures at higher levels of BMD than individuals without diabetes mellitus.
Koromani, F. et al. Vertebral fractures in individuals with type 2 diabetes: more than skeletal complications alone. Diabetes Care 43, 137–144 (2020).
Kanis, J. A., Johnell, O., Oden, A., Johansson, H. & McCloskey, E. FRAX™ and the assessment of fracture probability in men and women from the UK. Osteoporos. Int. 19, 385–397 (2008).
Ioacara, S. et al. Improvements in life expectancy in adult type 2 diabetes patients in the last six decades. Diabetes Res. Clin. Pract. 92, 400–404 (2011).
Melton, L. J. 3rd, Leibson, C. L., Achenbach, S. J., Therneau, T. M. & Khosla, S. Fracture risk in type 2 diabetes: update of a population-based study. J. Bone Miner. Res. 23, 1334–1342 (2008).
Vilaca, T. et al. The risk of hip and non-vertebral fractures in type 1 and type 2 diabetes: a systematic review and meta-analysis update. Bone 137, 115457 (2020).
Janghorbani, M., Van Dam, R. M., Willett, W. C. & Hu, F. B. Systematic review of type 1 and type 2 diabetes mellitus and risk of fracture. Am. J. Epidemiol. 166, 495–505 (2007).
Fan, Y., Wei, F., Lang, Y. & Liu, Y. Diabetes mellitus and risk of hip fractures: a meta-analysis. Osteoporos. Int 27, 219–228 (2016).
Park, H. Y., Han, K., Kim, Y., Kim, Y. H. & Sur, Y. J. The risk of hip fractures in individuals over 50 years old with prediabetes and type 2 diabetes – a longitudinal nationwide population-based study. Bone 142, 115691 (2020).
Wang, H., Ba, Y., Xing, Q. & Du, J. L. Diabetes mellitus and the risk of fractures at specific sites: a meta-analysis. BMJ Open 9, e024067 (2019).
Vilaca, T., Walsh, J. & Eastell, R. Discordant pattern of peripheral fractures in diabetes: a meta-analysis on the risk of wrist and ankle fractures. Osteoporos. Int. 30, 135–143 (2019).
Adami, G. et al. Risk of fragility fractures in obesity and diabetes: a retrospective analysis on a nation-wide cohort. Osteoporos. Int. 31, 2113–2122 (2020).
Napoli, N. et al. Vertebral fracture risk in diabetic elderly men: the MrOS study. J. Bone Miner. Res. 33, 63–69 (2018).
Oei, L. et al. High bone mineral density and fracture risk in type 2 diabetes as skeletal complications of inadequate glucose control: the Rotterdam study. Diabetes Care 36, 1619–1628 (2013).
Schneider, A. L. et al. Diabetes and risk of fracture-related hospitalization: the Atherosclerosis Risk in Communities study. Diabetes Care 36, 1153–1158 (2013).
Li, C. I. et al. Glycated hemoglobin level and risk of hip fracture in older people with type 2 diabetes: a competing risk analysis of Taiwan diabetes cohort study. J. Bone Miner. Res. 30, 1338–1346 (2015).
Conway, B. N., Long, D. M., Figaro, M. K. & May, M. E. Glycemic control and fracture risk in elderly patients with diabetes. Diabetes Res. Clin. Pract. 115, 47–53 (2016).
Ntouva, A. et al. Hypoglycaemia is associated with increased risk of fractures in patients with type 2 diabetes mellitus: a cohort study. Eur. J. Endocrinol. 180, 51–58 (2019).
Hung, Y. C. et al. Severe hypoglycemia and hip fracture in patients with type 2 diabetes: a nationwide population-based cohort study. Osteoporos. Int. 28, 2053–2060 (2017).
Johnston, S. S., Conner, C., Aagren, M., Ruiz, K. & Bouchard, J. Association between hypoglycaemic events and fall-related fractures in Medicare-covered patients with type 2 diabetes. Diabetes Obes. Metab. 14, 634–643 (2012).
Komorita, Y. et al. Both hypo- and hyperglycaemia are associated with increased fracture risk in Japanese people with type 2 diabetes: the Fukuoka Diabetes Registry. Diabet. Med. 37, 838–847 (2020). An observational study demonstrating that recurrent hypoglycaemia as well as poorly controlled T2DM are associated with an increase in fracture risk.
Lui, D. T. W. et al. HbA1c variability, in addition to mean HbA1c, predicts incident hip fractures in Chinese people with type 2 diabetes. Osteoporos. Int. 31, 1955–1964 (2020).
Bonds, D. E. et al. Risk of fracture in women with type 2 diabetes: the Women’s Health Initiative Observational study. J. Clin. Endocrinol. Metab. 91, 3404–3410 (2006).
Ma, L. et al. Association between bone mineral density and type 2 diabetes mellitus: a meta-analysis of observational studies. Eur. J. Epidemiol. 27, 319–332 (2012).
Starup-Linde, J. & Vestergaard, P. Biochemical bone turnover markers in diabetes mellitus – a systematic review. Bone 82, 69–78 (2016).
Farr, J. N. et al. In vivo assessment of bone quality in postmenopausal women with type 2 diabetes. J. Bone Miner. Res. 29, 787–795 (2014). The first in vivo demonstration that bone material properties are impaired in patients with T2DM.
Tonks, K. T., White, C. P., Center, J. R., Samocha-Bonet, D. & Greenfield, J. R. Bone turnover is suppressed in insulin resistance, independent of adiposity. J. Clin. Endocrinol. Metab. 102, 1112–1121 (2017). This study demonstrates that the reduced bone turnover in patients with T2DM is linked to insulin resistance and increases in visceral adipose tissue.
Timar, B. et al. The impact of diabetic neuropathy on balance and on the risk of falls in patients with type 2 diabetes mellitus: a cross-sectional study. PLoS ONE 11, e0154654 (2016).
Leslie, W. D., Aubry-Rozier, B., Lamy, O. & Hans, D. TBS (trabecular bone score) and diabetes-related fracture risk. J. Clin. Endocrinol. Metab. 98, 602–609 (2013).
Patsch, J. M. et al. Increased cortical porosity in type 2 diabetic postmenopausal women with fragility fractures. J. Bone Miner. Res. 28, 313–324 (2013).
Heilmeier, U. et al. Cortical bone laminar analysis reveals increased midcortical and periosteal porosity in type 2 diabetic postmenopausal women with history of fragility fractures compared to fracture-free diabetics. Osteoporos. Int. 27, 2791–2802 (2016).
Burghardt, A. J. et al. High-resolution peripheral quantitative computed tomographic imaging of cortical and trabecular bone microarchitecture in patients with type 2 diabetes mellitus. J. Clin. Endocrinol. Metab. 95, 5045–5055 (2010).
Yu, E. W. et al. Defects in cortical microarchitecture among African-American women with type 2 diabetes. Osteoporos. Int. 26, 673–679 (2015).
Samakkarnthai, P. et al. Determinants of bone material strength and cortical porosity in patients with type 2 diabetes mellitus. J. Clin. Endocrinol. Metab. 105, e3718–e372 (2020). This study demonstrates that, in patients with T2DM, impaired bone material properties are associated with accumulation of AGEs, whereas increased cortical porosity is associated with microvascular disease.
Samelson, E. J. et al. Diabetes and deficits in cortical bone density, microarchitecture, and bone size: Framingham HR-pQCT study. J. Bone Miner. Res. 33, 54–62 (2018).
Paccou, J. et al. Bone microarchitecture in men and women with diabetes: the importance of cortical porosity. Calcif. Tissue Int. 98, 465–473 (2016).
Shanbhogue, V. V. et al. Compromised cortical bone compartment in type 2 diabetes mellitus patients with microvascular disease. Eur. J. Endocrinol. 174, 115–124 (2016).
Shu, A. et al. Bone structure and turnover in type 2 diabetes mellitus. Osteoporos. Int. 23, 635–641 (2012).
Bala, Y. et al. Cortical porosity identifies women with osteopenia at increased risk for forearm fractures. J. Bone Miner. Res. 29, 1356–1362 (2014).
Bala, Y. et al. Risedronate slows or partly reverses cortical and trabecular microarchitectural deterioration in postmenopausal women. J. Bone Miner. Res. 29, 380–388 (2014).
Furst, J. R. et al. Advanced glycation endproducts and bone material strength in type 2 diabetes. J. Clin. Endocrinol. Metab. 101, 2502–2510 (2016).
Nilsson, A. G. et al. Type 2 diabetes mellitus is associated with better bone microarchitecture but lower bone material strength and poorer physical function in elderly women: a population-based study. J. Bone Miner. Res. 32, 1062–1071 (2017).
Dawson-Hughes, B., Bouxsein, M. & Shea, K. Bone material strength in normoglycemic and hyperglycemic black and white older adults. Osteoporos. Int. 30, 2429–2435 (2019).
Meerwaldt, R. et al. Simple non-invasive assessment of advanced glycation endproduct accumulation. Diabetologia 47, 1324–1330 (2004).
Litwinoff, E., Hurtado Del Pozo, C., Ramasamy, R. & Schmidt, A. M. Emerging targets for therapeutic development in diabetes and its complications: the RAGE signaling pathway. Clin. Pharmacol. Ther. 98, 135–144 (2015).
Fournet, M., Bonté, F. & Desmoulière, A. Glycation damage: a possible hub for major pathophysiological disorders and aging. Aging Dis. 9, 880–900 (2018).
Byun, K. et al. Advanced glycation end-products produced systemically and by macrophages: a common contributor to inflammation and degenerative diseases. Pharmacol. Ther. 177, 44–55 (2017).
Desai, C. S., Blumenthal, R. S. & Greenland, P. Screening low-risk individuals for coronary artery disease. Curr. Atheroscler. Rep. 16, 402 (2014).
Bacharach, J. M., Rooke, T. W., Osmundson, P. J. & Gloviczki, P. Predictive value of transcutaneous oxygen pressure and amputation success by use of supine and elevation measurements. J. Vasc. Surg. 15, 558–563 (1992).
Thrailkill, K. M., Lumpkin, C. K. Jr., Bunn, R. C., Kemp, S. F. & Fowlkes, J. L. Is insulin an anabolic agent in bone? Dissecting the diabetic bone for clues. Am. J. Physiol. Endocrinol. Metab. 289, E735–745 (2005).
Stolk, R. P. et al. Hyperinsulinemia and bone mineral density in an elderly population: the Rotterdam study. Bone 18, 545–549 (1996).
Johnson, K. C. et al. The effect of intentional weight loss on fracture risk in persons with diabetes: results from the look AHEAD randomized clinical trial. J. Bone Miner. Res. 32, 2278–2287 (2017).
DiGirolamo, D. J., Clemens, T. L. & Kousteni, S. The skeleton as an endocrine organ. Nat. Rev. Rheumatol. 8, 674–683 (2012).
Liu, J. M., Rosen, C. J., Ducy, P., Kousteni, S. & Karsenty, G. Regulation of glucose handling by the skeleton: insights from mouse and human studies. Diabetes 65, 3225–3232 (2016).
Dirckx, N., Moorer, M. C., Clemens, T. L. & Riddle, R. C. The role of osteoblasts in energy homeostasis. Nat. Rev. Endocrinol. 15, 651–665 (2019).
Lecka-Czernik, B. & Rosen, C. J. Energy excess, glucose utilization, and skeletal remodeling: new insights. J. Bone Miner. Res. 30, 1356–1361 (2015).
Rendina-Ruedy, E. & Rosen, C. J. Lipids in the bone marrow: an evolving perspective. Cell Metab. 31, 219–231 (2020).
Shanbhogue, V. V., Mitchell, D. M., Rosen, C. J. & Bouxsein, M. L. Type 2 diabetes and the skeleton: new insights into sweet bones. Lancet Diabetes Endocrinol. 4, 159–173 (2016).
Eckhardt, B. A. et al. Accelerated osteocyte senescence and skeletal fragility in mice with type 2 diabetes. JCI Insight 5, e135236 (2020). This work demonstrates that at least in a mouse model, the combination of obesity and hyperglycaemia is associated with increased cellular senescence in bone.
Starr, J. F. et al. Robust trabecular microstructure in type 2 diabetes revealed by individual trabecula segmentation analysis of HR-pQCT Images. J. Bone Miner. Res. 33, 1665–1675 (2018).
Ogata, N. et al. Insulin receptor substrate-1 in osteoblast is indispensable for maintaining bone turnover. J. Clin. Invest. 105, 935–943 (2000).
Abrahamsen, B., Rohold, A., Henriksen, J. E. & Beck-Nielsen, H. Correlations between insulin sensitivity and bone mineral density in non-diabetic men. Diabet. Med. 17, 124–129 (2000).
Fontana, L., Eagon, J. C., Trujillo, M. E., Scherer, P. E. & Klein, S. Visceral fat adipokine secretion is associated with systemic inflammation in obese humans. Diabetes 56, 1010–1013 (2007).
Martin, T. J. & Sims, N. A. RANKL/OPG; critical role in bone physiology. Rev. Endocr. Metab. Disord. 16, 131–139 (2015).
Krings, A. et al. Bone marrow fat has brown adipose tissue characteristics, which are attenuated with aging and diabetes. Bone 50, 546–552 (2012).
Sheu, Y. et al. Vertebral bone marrow fat, bone mineral density and diabetes: the osteoporotic fractures in men (MrOS) study. Bone 97, 299–305 (2017).
Baum, T. et al. Does vertebral bone marrow fat content correlate with abdominal adipose tissue, lumbar spine bone mineral density, and blood biomarkers in women with type 2 diabetes mellitus? J. Magn. Reson. Imaging 35, 117–124 (2012).
Fazeli, P. K. et al. Marrow fat and bone–new perspectives. J. Clin. Endocrinol. Metab. 98, 935–945 (2013).
Nuche-Berenguer, B. et al. Exendin-4 exerts osteogenic actions in insulin-resistant and type 2 diabetic states. Regul. Pept. 159, 61–66 (2010).
Gennari, L. et al. Circulating sclerostin levels and bone turnover in type 1 and type 2 diabetes. J. Clin. Endocrinol. Metab. 97, 1737–1744 (2012).
Piccoli, A. et al. Sclerostin regulation, microarchitecture, and advanced glycation end-products in the bone of elderly women with type 2 diabetes. J. Bone Miner. Res. 35, 2415–2422 (2020).
Baron, R. & Kneissel, M. WNT signaling in bone homeostasis and disease: from human mutations to treatments. Nat. Med. 19, 179–192 (2013).
Ma, Y. H. et al. Circulating sclerostin associated with vertebral bone marrow fat in older men but not women. J. Clin. Endocrinol. Metab. 99, E2584–2590 (2014).
Khosla, S., Farr, J. N., Tchkonia, T. & Kirkland, J. L. The role of cellular senescence in ageing and endocrine disease. Nat. Rev. Endocrinol. 16, 263–275 (2020).
Alcorta, D. A. et al. Involvement of the cyclin-dependent kinase inhibitor p16 (INK4a) in replicative senescence of normal human fibroblasts. Proc. Natl Acad. Sci. USA 93, 13742–13747 (1996).
Beauséjour, C. M. et al. Reversal of human cellular senescence: roles of the p53 and p16 pathways. EMBO J. 22, 4212–4222 (2003).
Wissler Gerdes, E. O., Zhu, Y., Tchkonia, T. & Kirkland, J. L. Discovery, development, and future application of senolytics: theories and predictions. FEBS J. 287, 2418–2427 (2020).
Coppé, J. P., Desprez, P. Y., Krtolica, A. & Campisi, J. The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu. Rev. Pathol. 5, 99–118 (2010).
Acosta, J. C. et al. A complex secretory program orchestrated by the inflammasome controls paracrine senescence. Nat. Cell Biol. 15, 978–990 (2013).
Palmer, A. K. et al. Cellular senescence in type 2 diabetes: a therapeutic opportunity. Diabetes 64, 2289–2298 (2015).
Palmer, A. K., Gustafson, B., Kirkland, J. L. & Smith, U. Cellular senescence: at the nexus between ageing and diabetes. Diabetologia 62, 1835–1841 (2019).
Prata, L., Ovsyannikova, I. G., Tchkonia, T. & Kirkland, J. L. Senescent cell clearance by the immune system: emerging therapeutic opportunities. Semin. Immunol. 40, 101275 (2018).
Wang, E. Senescent human fibroblasts resist programmed cell death, and failure to suppress bcl2 is involved. Cancer Res. 55, 2284–2292 (1995).
Tchkonia, T. et al. Fat tissue, aging, and cellular senescence. Aging Cell 9, 667–684 (2010).
Palmer, A. K. et al. Targeting senescent cells alleviates obesity-induced metabolic dysfunction. Aging Cell 18, e12950 (2019).
Aguayo-Mazzucato, C. et al. Acceleration of β cell aging determines diabetes and senolysis improves disease outcomes. Cell Metab. 30, 129–142.e124 (2019).
Ogrodnik, M. et al. Cellular senescence drives age-dependent hepatic steatosis. Nat. Commun. 8, 15691 (2017).
Ogrodnik, M. et al. Obesity-induced cellular senescence drives anxiety and impairs neurogenesis. Cell Metab. 29, 1061–1077.e8 (2019).
Xu, M. et al. Targeting senescent cells enhances adipogenesis and metabolic function in old age. eLife 4, e12997 (2015).
Zaragosi, L. E. et al. Activin A plays a critical role in proliferation and differentiation of human adipose progenitors. Diabetes 59, 2513–2521 (2010).
Nelson, G. et al. A senescent cell bystander effect: senescence-induced senescence. Aging Cell 11, 345–349 (2012).
da Silva, P. F. L. et al. The bystander effect contributes to the accumulation of senescent cells in vivo. Aging Cell 18, e12848 (2019).
Manavalan, J. S. et al. Circulating osteogenic precursor cells in type 2 diabetes mellitus. J. Clin. Endocrinol. Metab. 97, 3240–3250 (2012).
Gorgoulis, V. et al. Cellular senescence: defining a path forward. Cell 179, 813–827 (2019).
Swanson, E. C., Manning, B., Zhang, H. & Lawrence, J. B. Higher-order unfolding of satellite heterochromatin is a consistent and early event in cell senescence. J. Cell Biol. 203, 929–942 (2013).
Coppé, J. P. et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 6, 2853–2868 (2008).
Ramasamy, R., Shekhtman, A. & Schmidt, A. M. The multiple faces of RAGE–opportunities for therapeutic intervention in aging and chronic disease. Expert. Opin. Ther. Targets 20, 431–446 (2016).
Poundarik, A. A. et al. A direct role of collagen glycation in bone fracture. J. Mech. Behav. Biomed. Mater. 52, 120–130 (2015).
Zhou, Z. et al. Regulation of osteoclast function and bone mass by RAGE. J. Exp. Med. 203, 1067–1080 (2006).
Franke, S. et al. Advanced glycation end products affect growth and function of osteoblasts. Clin. Exp. Rheumatol. 29, 650–660 (2011).
Ahmed, N. & Thornalley, P. J. Quantitative screening of protein biomarkers of early glycation, advanced glycation, oxidation and nitrosation in cellular and extracellular proteins by tandem mass spectrometry multiple reaction monitoring. Biochem. Soc. Trans. 31, 1417–1422 (2003).
O’Grady, K. L. et al. Development and application of mass spectroscopy assays for Nε-(1-carboxymethyl)-L-lysine and pentosidine in renal failure and diabetes. J. Appl. Lab. Med. 5, 558–568 (2020).
Karim, L. et al. Bone microarchitecture, biomechanical properties, and advanced glycation end-products in the proximal femur of adults with type 2 diabetes. Bone 114, 32–39 (2018).
Schwartz, A. V. et al. Pentosidine and increased fracture risk in older adults with type 2 diabetes. J. Clin. Endocrinol. Metab. 94, 2380–2386 (2009).
Wang, J., Wang, H., Shi, J. & Ding, Y. Effects of bone marrow MSCs transfected with sRAGE on the intervention of HMGB1 induced immuno-inflammatory reaction. Int. J. Clin. Exp. Pathol. 8, 12028–12040 (2015).
Lalla, E. et al. Blockade of RAGE suppresses periodontitis-associated bone loss in diabetic mice. J. Clin. Invest. 105, 1117–1124 (2000).
Walker, D., Lue, L. F., Paul, G., Patel, A. & Sabbagh, M. N. Receptor for advanced glycation endproduct modulators: a new therapeutic target in Alzheimer’s disease. Expert. Opin. Investig. Drugs 24, 393–399 (2015).
Galasko, D. et al. Clinical trial of an inhibitor of RAGE-Aβ interactions in Alzheimer disease. Neurology 82, 1536–1542 (2014).
Mao, Y. X. et al. RAGE-dependent mitochondria pathway: a novel target of silibinin against apoptosis of osteoblastic cells induced by advanced glycation end products. Cell Death Dis. 9, 674 (2018).
Chen, H. et al. Advanced glycation end products induced IL-6 and VEGF-A production and apoptosis in osteocyte-like MLO-Y4 cells by activating RAGE and ERK1/2, P38 and STAT3 signalling pathways. Int. Immunopharmacol. 52, 143–149 (2017).
Lafage-Proust, M. H. et al. Assessment of bone vascularization and its role in bone remodeling. Bonekey Rep. 4, 662 (2015).
Andersen, T. L. et al. A physical mechanism for coupling bone resorption and formation in adult human bone. Am. J. Pathol. 174, 239–247 (2009).
Rehman, J., Li, J., Orschell, C. M. & March, K. L. Peripheral blood “endothelial progenitor cells” are derived from monocyte/macrophages and secrete angiogenic growth factors. Circulation 107, 1164–1169 (2003).
Guo, P. et al. Platelet-derived growth factor-B enhances glioma angiogenesis by stimulating vascular endothelial growth factor expression in tumor endothelia and by promoting pericyte recruitment. Am. J. Pathol. 162, 1083–1093 (2003).
Colnot, C. Skeletal cell fate decisions within periosteum and bone marrow during bone regeneration. J. Bone Miner. Res. 24, 274–282 (2009).
Liao, Y. H. et al. Osteogenic differentiation of adipose-derived stem cells and calvarial defect repair using baculovirus-mediated co-expression of BMP-2 and miR-148b. Biomaterials 35, 4901–4910 (2014).
Divya, M. S. et al. Umbilical cord blood-derived mesenchymal stem cells consist of a unique population of progenitors co-expressing mesenchymal stem cell and neuronal markers capable of instantaneous neuronal differentiation. Stem Cell Res. Ther. 3, 57 (2012).
Wynn, T. A. & Vannella, K. M. Macrophages in tissue repair, regeneration, and fibrosis. Immunity 44, 450–462 (2016).
Doherty, M. J. et al. Vascular pericytes express osteogenic potential in vitro and in vivo. J. Bone Miner. Res. 13, 828–838 (1998).
Rask-Madsen, C. & King, G. L. Vascular complications of diabetes: mechanisms of injury and protective factors. Cell Metab. 17, 20–33 (2013).
Farr, J. N. & Khosla, S. Determinants of bone strength and quality in diabetes mellitus in humans. Bone 82, 28–34 (2016).
Cosman, F. et al. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos. Int. 25, 2359–2381 (2014).
Roman de Mettelinge, T., Cambier, D., Calders, P., Van Den Noortgate, N. & Delbaere, K. Understanding the relationship between type 2 diabetes mellitus and falls in older adults: a prospective cohort study. PLoS ONE 8, e67055 (2013).
Schwartz, A. V. et al. Older women with diabetes have a higher risk of falls: a prospective study. Diabetes Care 25, 1749–1754 (2002).
Chapman, A., Meyer, C., Renehan, E., Hill, K. D. & Browning, C. J. Exercise interventions for the improvement of falls-related outcomes among older adults with diabetes mellitus: a systematic review and meta-analyses. J. Diabetes Complicat. 31, 631–645 (2017).
Gu, Y. & Dennis, S. M. Are falls prevention programs effective at reducing the risk factors for falls in people with type-2 diabetes mellitus and peripheral neuropathy: a systematic review with narrative synthesis. J. Diabetes Complicat. 31, 504–516 (2017).
Napoli, N. et al. Fracture risk in diabetic elderly men: the MrOS study. Diabetologia 57, 2057–2065 (2014).
Pscherer, S., Kostev, K., Dippel, F. W. & Rathmann, W. Fracture risk in patients with type 2 diabetes under different antidiabetic treatment regimens: a retrospective database analysis in primary care. Diabetes Metab. Syndr. Obes. 9, 17–23 (2016).
Losada-Grande, E. et al. Insulin use and excess fracture risk in patients with type 2 diabetes: a propensity-matched cohort analysis. Sci. Rep. 7, 3781 (2017).
Palermo, A. et al. Oral anti-diabetic drugs and fracture risk, cut to the bone: safe or dangerous? A narrative review. Osteoporos. Int. 26, 2073–2089 (2015).
Cheng, L. et al. Glucagon-like peptide-1 receptor agonists and risk of bone fracture in patients with type 2 diabetes: a meta-analysis of randomized controlled trials. Diabetes Metab. Res. Rev. 35, e3168 (2019).
Zhu, Z. N., Jiang, Y. F. & Ding, T. Risk of fracture with thiazolidinediones: an updated meta-analysis of randomized clinical trials. Bone 68, 115–123 (2014).
Watts, N. B. et al. Effects of canagliflozin on fracture risk in patients with type 2 diabetes mellitus. J. Clin. Endocrinol. Metab. 101, 157–166 (2016).
Li, X. et al. Effects of SGLT2 inhibitors on fractures and bone mineral density in type 2 diabetes: an updated meta-analysis. Diabetes Metab. Res. Rev. 35, e3170 (2019).
Barzilay, J. I. et al. The impact of antihypertensive medications on bone mineral density and fracture risk. Curr. Cardiol. Rep. 19, 76 (2017).
Bokrantz, T. et al. Antihypertensive drug classes and the risk of hip fracture: results from the Swedish primary care cardiovascular database. J. Hypertens. 38, 167–175 (2020).
Shi, R., Mei, Z., Zhang, Z. & Zhu, Z. Effects of statins on relative risk of fractures for older adults: an updated systematic review with meta-analysis. J. Am. Med. Dir. Assoc. 20, 1566–1578.e3 (2019).
Wu, Q., Bencaz, A. F., Hentz, J. G. & Crowell, M. D. Selective serotonin reuptake inhibitor treatment and risk of fractures: a meta-analysis of cohort and case-control studies. Osteoporos. Int. 23, 365–375 (2012).
Wu, Q., Qu, W., Crowell, M. D., Hentz, J. G. & Frey, K. A. Tricyclic antidepressant use and risk of fractures: a meta-analysis of cohort and case-control studies. J. Bone Miner. Res. 28, 753–763 (2013).
Anagnostis, P. et al. Efficacy of anti-osteoporotic medications in patients with type 1 and 2 diabetes mellitus: a systematic review. Endocrine 60, 373–383 (2018).
Napoli, N. et al. Effect of denosumab on fasting glucose in women with diabetes or prediabetes from the FREEDOM trial. Diabetes Metab. Res. Rev. 34, e2991 (2018).
Kondegowda, N. G. et al. Osteoprotegerin and denosumab stimulate human beta cell proliferation through inhibition of the receptor activator of NF-κB ligand pathway. Cell Metab. 22, 77–85 (2015).
Schmitz, F., Roscioni, S. & Lickert, H. Repurposing an osteoporosis drug for β cell regeneration in diabetic patients. Cell Metab. 22, 58–59 (2015).
Weivoda, M. M. et al. Identification of osteoclast-osteoblast coupling factors in humans reveals links between bone and energy metabolism. Nat. Commun. 11, 87 (2020).
Dhaliwal, R. et al. Abaloparatide in postmenopausal women with osteoporosis and type 2 diabetes: a post hoc analysis of the ACTIVE study. JBMR 4, e10346 (2020).
Schmidt, A. M. Soluble RAGEs - prospects for treating & tracking metabolic and inflammatory disease. Vasc. Pharmacol. 72, 1–8 (2015).
Ding, K. H. et al. Disordered osteoclast formation in RAGE-deficient mouse establishes an essential role for RAGE in diabetes related bone loss. Biochem. Biophys. Res. Commun. 340, 1091–1097 (2006).
Egawa, T. et al. Potential involvement of dietary advanced glycation end products in impairment of skeletal muscle growth and muscle contractile function in mice. Br. J. Nutr. 117, 21–29 (2017).
Davis, H. M. et al. Short-term pharmacologic RAGE inhibition differentially affects bone and skeletal muscle in middle-aged mice. Bone 124, 89–102 (2019).
Farr, J. N. et al. Targeting cellular senescence prevents age-related bone loss in mice. Nat. Med. 23, 1072–1079 (2017). A study demonstrating that reducing the burden of senescent cells ameliorates age-related bone loss in mice, which raises the possibility that a similar approach might be useful in alleviating the skeletal fragility associated with T2DM.
Monami, M. et al. Bone fractures and hypoglycemic treatment in type 2 diabetic patients: a case-control study. Diabetes Care 31, 199–203 (2008).
Vestergaard, P., Rejnmark, L. & Mosekilde, L. Relative fracture risk in patients with diabetes mellitus, and the impact of insulin and oral antidiabetic medication on relative fracture risk. Diabetologia 48, 1292–1299 (2005).
Hidayat, K., Du, X., Wu, M. J. & Shi, B. M. The use of metformin, insulin, sulphonylureas, and thiazolidinediones and the risk of fracture: systematic review and meta-analysis of observational studies. Obes. Rev. 20, 1494–1503 (2019).
Josse, R. G. et al. Sitagliptin and risk of fractures in type 2 diabetes: results from the TECOS trial. Diabetes Obes. Metab. 19, 78–86 (2017).
Schwartz, A. V. et al. Effects of TZD use and discontinuation on fracture rates in ACCORD bone study. J. Clin. Endocrinol. Metab. 100, 4059–4066 (2015).
Lapane, K. L., Jesdale, B. M., Dubé, C. E., Pimentel, C. B. & Rajpathak, S. N. Sulfonylureas and risk of falls and fractures among nursing home residents with type 2 diabetes mellitus. Diabetes Res. Clin. Pract. 109, 411–419 (2015).
Hidayat, K., Du, X. & Shi, B. M. Risk of fracture with dipeptidyl peptidase-4 inhibitors, glucagon-like peptide-1 receptor agonists, or sodium-glucose cotransporter-2 inhibitors in real-world use: systematic review and meta-analysis of observational studies. Osteoporos. Int. 30, 1923–1940 (2019).
Monami, M., Dicembrini, I., Antenore, A. & Mannucci, E. Dipeptidyl peptidase-4 inhibitors and bone fractures: a meta-analysis of randomized clinical trials. Diabetes Care 34, 2474–2476 (2011).
Mosenzon, O. et al. Incidence of fractures in patients with type 2 diabetes in the SAVOR-TIMI 53 trial. Diabetes Care 38, 2142–2150 (2015).
Kohan, D. E., Fioretto, P., Tang, W. & List, J. F. Long-term study of patients with type 2 diabetes and moderate renal impairment shows that dapagliflozin reduces weight and blood pressure but does not improve glycemic control. Kidney Int. 85, 962–971 (2014).
Acknowledgements
The authors acknowledge the support of NIH grants AG062413 (S.K., J.N.F.), AG004875 (S.K., D.G.M.), AR027065 (S.K.), AR070241 (J.N.F.), AG065868 (J.N.F., S.K.), AG063707 (D.G.M.) and AR068275 (D.G.M.).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Endocrinology thanks P. Vestergaard, who co-reviewed with R. Viggers, A. Schwartz and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khosla, S., Samakkarnthai, P., Monroe, D.G. et al. Update on the pathogenesis and treatment of skeletal fragility in type 2 diabetes mellitus. Nat Rev Endocrinol 17, 685–697 (2021). https://doi.org/10.1038/s41574-021-00555-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41574-021-00555-5
This article is cited by
-
A DNA tetrahedron-based ferroptosis-suppressing nanoparticle: superior delivery of curcumin and alleviation of diabetic osteoporosis
Bone Research (2024)
-
Rescuing SERCA2 pump deficiency improves bone mechano-responsiveness in type 2 diabetes by shaping osteocyte calcium dynamics
Nature Communications (2024)
-
Establishment and validation of a nomogram clinical prediction model for osteoporosis in senile patients with type 2 diabetes mellitus
Scientific Reports (2024)
-
Insights and implications of sexual dimorphism in osteoporosis
Bone Research (2024)
-
A study on the association between gut microbiota, inflammation, and type 2 diabetes
Applied Microbiology and Biotechnology (2024)