Abstract
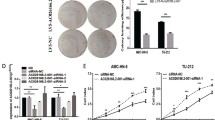
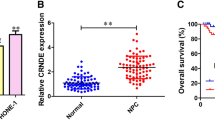
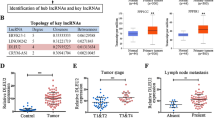
Emerging evidence suggests that long non-coding RNA (lncRNA) is closely associated with numerous human diseases, including cancer. However, the functional relevance of lncRNA in human laryngeal squamous cell carcinoma (LSCC) is largely unknown. In the current study, we described CCAT2, a previously unappreciated oncogenic lncRNA in LSCC. CCAT2 was significantly upregulated in human LSCC tissue and serum samples, associated with larger tumor volume, higher clinical stage, and poorer differentiation status. Lentivirus-mediated CCAT2 knockdown notably repressed the cell viability, colony formation, and DNA synthesis rate of LSCC. Screening of transcription factors revealed that YAP/TEAD activity was affected by CCAT2 in LSCC cells. Further, CCAT2 directly binds to YAP protein and blocks the phosphorylation of YAP induced by LATS1, resulting in the nuclear translocation of YAP and the activation of YAP oncogenic targets, such as CTGF, CYR61 and AMOTL2. Importantly, we also confirmed the regulation of CCAT2 on YAP activity in vivo based on nude mice model. Altogether, we identified a novel lncRNA that controls YAP nucleocytoplasmic shuttling and promotes LSCC cell proliferation. Given the importance of YAP in tumorigenesis and progression, our results provide insights to intervene LSCC by targeting the CCAT2/YAP axis.





Similar content being viewed by others
References
Leemans CR, Snijders P, Brakenhoff RH. The molecular landscape of head and neck cancer. Nat Rev Cancer. 2018;18:269–82.
Mur T, Jaleel Z, Agarwal P, Edwards H, Levi JR. Paediatric laryngeal squamous cell carcinoma: systematic review and pooled analysis. Clin Otolaryngol. 2021;46:494–500.
Dietz A, Wiegand S, Kuhnt T, Wichmann G. Laryngeal preservation approaches: considerations for new selection criteria based on the DeLOS-II trial. Front Oncol. 2019;9:625.
Forsyth AM, Camilon PR, Tracy L, Levi JR. Pediatric laryngeal tumors and demographics, management, and survival in laryngeal squamous cell carcinoma. Int J Pediatr Otorhinolaryngol. 2021;140:110507.
Gregoire V, Grau C, Lapeyre M, Maingon P. Target volume selection and delineation (T and N) for primary radiation treatment of oral cavity, oropharyngeal, hypopharyngeal and laryngeal squamous cell carcinoma. Oral Oncol. 2018;87:131–7.
Kyurkchiyan SG, Popov TM, Mitev VI, Kaneva RP. The role of miRNAs and lncRNAs in laryngeal squamous cell carcinoma—a mini-review. Folia Med. 2020;62:244–52.
Beermann J, Piccoli MT, Viereck J, Thum T. Non-coding RNAs in development and disease: background, mechanisms, and therapeutic approaches. Physiol Rev. 2016;96:1297–325.
Nair L, Chung H, Basu U. Regulation of long non-coding RNAs and genome dynamics by the RNA surveillance machinery. Nat Rev Mol Cell Biol. 2020;21:123–36.
Boon RA, Jae N, Holdt L, Dimmeler S. Long noncoding RNAs: from clinical genetics to therapeutic targets? J Am Coll Cardiol. 2016;67:1214–26.
Lin YH, Wu MH, Liu YC, Lyu PC, Yeh CT, Lin KH. LINC01348 suppresses hepatocellular carcinoma metastasis through inhibition of SF3B3-mediated EZH2 pre-mRNA splicing. Oncogene. 2021. https://doi.org/10.1038/s41388-021-01905-3.
Huang Y, Zheng Y, Shao X, Shi L, Li G, Huang P. Long non-coding RNA TPT1-AS1 sensitizes breast cancer cell to paclitaxel and inhibits cell proliferation by miR-3156-5p/caspase 2 axis. Hum Cell. 2021;34:1244–54.
Yi Q, Liu Z, Zhang K, et al. The role of long non-coding RNA BCAR4 in human cancers. Hum Cell. 2021. https://doi.org/10.1007/s13577-021-00556-6.
Zang Y, Li J, Wan B, Tai Y. circRNA circ-CCND1 promotes the proliferation of laryngeal squamous cell carcinoma through elevating CCND1 expression via interacting with HuR and miR-646. J Cell Mol Med. 2020;24:2423–33.
Ren X. Genome-wide analysis reveals the emerging roles of long non-coding RNAs in cancer. Oncol Lett. 2020;19:588–94.
Qian X, Zhao J, Yeung PY, Zhang QC, Kwok CK. Revealing lncRNA structures and interactions by sequencing-based approaches. Trends Biochem Sci. 2019;44:33–52.
Ling H, Spizzo R, Atlasi Y, et al. CCAT2, a novel noncoding RNA mapping to 8q24, underlies metastatic progression and chromosomal instability in colon cancer. Genome Res. 2013;23:1446–61.
Xin Y, Li Z, Zheng H, Chan M, Ka KWW. CCAT2: a novel oncogenic long non-coding RNA in human cancers. Cell Prolif. 2017;50:e12342.
Ghafouri-Fard S, Hajiesmaeili M, Shoorei H, Bahroudi Z, Taheri M, Sharifi G. The Impact of lncRNAs and miRNAs in regulation of function of cancer stem cells and progression of cancer. Front Cell Dev Biol. 2021;9:696820.
Kan RL, Chen J, Sallam T. Crosstalk between epitranscriptomic and epigenetic mechanisms in gene regulation. Trends Genet. 2021. https://doi.org/10.1016/j.tig.2021.06.014.
Chowdhary A, Satagopam V, Schneider R. Long non-coding RNAs: mechanisms, experimental, and computational approaches in identification, characterization, and their biomarker potential in cancer. Front Genet. 2021;12:649619.
Tian Y, Wang Y, Li F, Yang J, Xu Y, Ouyang M. LncRNA TUG1 regulates the balance of HuR and miR-29b-3p and inhibits intestinal epithelial cell apoptosis in a mouse model of ulcerative colitis. Hum Cell. 2021;34:37–48.
Liang Y, Chen X, Wu Y, et al. LncRNA CASC9 promotes esophageal squamous cell carcinoma metastasis through upregulating LAMC2 expression by interacting with the CREB-binding protein. Cell Death Differ. 2018;25:1980–95.
Piccolo S, Dupont S, Cordenonsi M. The biology of YAP/TAZ: hippo signaling and beyond. Physiol Rev. 2014;94:1287–312.
Rausch V, Hansen CG. The Hippo Pathway, YAP/TAZ, and the Plasma Membrane. Trends Cell Biol. 2020;30:32–48.
Masliantsev K, Karayan-Tapon L, Guichet PO. Hippo Signaling Pathway in Gliomas. Cells-Basel. 2021;10:184.
Dey A, Varelas X, Guan KL. Targeting the Hippo pathway in cancer, fibrosis, wound healing and regenerative medicine. Nat Rev Drug Discov. 2020;19:480–94.
Aljabal G, Yap BK. 14-3-3sigma and Its Modulators in Cancer. Pharmaceuticals. 2020;13:441.
Funding
The work is funded by Henan Province Science and Technology Tackling Project (201201702196702196).
Author information
Authors and Affiliations
Contributions
Contributions: (I) Conception and design: HJ Liu; (II) Administrative support: HJ Liu; (III) Provision of study materials or patients: YZ Zang; (IV) Collection and assembly of data: J Li, BL Wan, Y Tai, Q Li, YZ Ji; (V) Data analysis and interpretation: YZ Zang; (VI) Manuscript writing: YZ Zang; (VII) Final approval of manuscript: all authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
The Ethics Committee of the People's Hospital of Henan Province approved the use of human tissue samples (No.YYNH-00368X), and the experimental steps were conducted in accordance with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The Institutional Animal Care and Use Committee of People's Hospital of Henan Province approved all animal care, use, and euthanasia (No.YYNH-00368X), which were handled in strict accordance with the “Guide for the Care and Use of Laboratory Animals” and the “Principles for the Utilization and Care of Vertebrate Animals”.
Informed consent
All the patients provided signed informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zang, Y., Li, J., Wan, B. et al. Long non-coding RNA CCAT2 drives the growth of laryngeal squamous cell carcinoma via regulating YAP activity. Human Cell 34, 1878–1887 (2021). https://doi.org/10.1007/s13577-021-00606-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13577-021-00606-z