Abstract
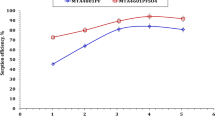
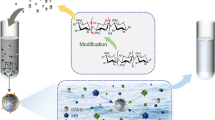
The recovery of thorium from acidic chloride medium with amine grafted cellulose resins was studied. Mesoporous polymers were modified with ethylenediamine (R1) and diethylenetriamine (R2) after grafting with glycidyl methacrylate. The sorbents obtained were characterized by Fourier transform IR spectroscopy, thermogravimetric analysis, scanning electron microscopy, and surface area measurements. Modified cellulose resins showed high capacity with relative fast kinetics in batch sorption of Th(IV) ions from chloride aqueous solutions. The maximum uptake capacity for Th(IV) ions on R1 and R2 was found to be 80 and 94.6 mg g–1 at pH 3.0 at 25°C within 45 min. The adsorption reaction followed the pseudo-second-order model. The data were fitted well by Langmuir isotherm. The mean adsorption energy calculated using DR isotherm showed that the adsorption followed the chemisorption mechanism. The process was exothermic. The performance of the sorbents in the recovery of Th(IV) ions from a standard rock sample and a granite sample from Gattar area, Egypt, was studied. Desorption was performed using different acids. The maximum Th(IV) desorption, 95 ± 2.8%, was reached with 1 M HNO3.
















Similar content being viewed by others
REFERENCES
Kadioglu, S. and Tellioglu, Z., UCTEA 1 Energy Symp., Ankara, 1996.
Gado, M. and Zaki, S., Int. J. Waste Resources, 2015, vol. 6, p. 1. https://doi.org/10.4172/2252-5211.1000194
Kursun, I., Terzi, M., and Tombal, T.D., Russ. J. Non-Ferrous Met., 2016, vol. 57, no. 3, pp. 187–194. https://doi.org/10.3103/S106782121603010X
Radchenko, V., Mastren, T., Meyer, C.A.L., Ivanov, A.S., Bryantsev, V.S., Copping, R., Denton, D., Engle, J.W., Griswold, J.R., Murphy, K., Wilson, J.J., Owens, A., Wyant, L., Birnbaum, E.R., Fitzsimmons, J., Medvedev, D., Cutler, C.S., Mausner, L.F., Nortier, F.M., John, K.D., Mirzadeh, S., and Fassbender, M.E., Talanta, 2017, vol. 175, p. 318. https://doi.org/10.1016/j.talanta.2017.07.057
Mastren, T., Radchenko, V., Owens, A., Copping, R., Bollm, R., Griswold, J.R., Mirzadeh, S., Wyant, L.E., Brugh, M., Engle, J.W., Nortier, F.M., Birnbaum, E.R., John, K.D., and Fassbender, M.E., Sci. Rep., 2017, vol. 7, p. 8216. https://doi.org/10.1038/s41598-017-08506-9
Mastren, T., Radchenko, V., Engle, J.W., Owens, A., Weidner, J.W., Owens, A., Wyant, L.E., Copping, R., Brugh, M., Nortier, F.M., Birnbaum, E.R., John, K.D., and Fassbender, M.E., Anal. Chim. Acta, 2018, vol. 998, pp. 75–82. https://doi.org/10.1016/j.aca.2017.10.020
Huang, H., Ding, S., Su, D., Liu, N., Wang, J., and Tan, M., Sep. Purif. Technol., 2014, vol. 138, p. 65. https://doi.org/10.1016/j.seppur.2014.10.008
Kul, M., Topkaya, Y., and Karakaya, I., Hydrometallurgy, 2008, vol. 93, no. 3, p. 129. https://doi.org/10.1016/j.hydromet.2007.11.008
Hu, C., Liu, H.J., Peng, L., Sun, Y.K., and Long, W., J. Radioanal. Nucl. Chem., 2016, vol. 308, no. 1, p. 251. https://doi.org/10.1007/s10967-015-4306-z
Chellam, S. and Clifford, D.A., J. Environ. Eng., 2002, vol. 128, no. 10, pp. 942–952. https://doi.org/10.1061/(ASCE)0733-9372(2002)128:10(942)
Kuruc, J., Strisovska, J., Galanda, D., Dulanska, S., Matel, L., Jerigova, M., and Velic, D., J. Radioanal. Nucl. Chem., 2012, vol. 292, pp. 973–981. https://doi.org/10.1007/s10967-012-1670-9
Sharma, S. and Balasubramanian, K., RSC Adv., 2015, vol.5, pp. 31732–31741. https://doi.org/10.1039/C5RA02861B
Thirumanavalan, M., Lai, Y., Lin, L., and Lee, J., J. Chem. Eng. Data, 2010, vol. 55, pp. 1186–1192. https://doi.org/10.1021/je900585t
Anirudhan, T.S., Sreekumari, S.S., and Jalajamony, S., J. Environ. Radioact., 2013, vol. 116, no. 1, p. 141. https://doi.org/10.1016/j.jenvrad.2012.10.001
Horsfall, M., Abia, A.A., and Spiff, A.I., Afr. J. Biotechnol., 2003, vol. 2, pp. 360–364.
Yousif, A.M., El-Afandy, A.H., Abdel Wahab, G.M., Mubark, A.E., and Ibrahim, I.A., J. Radioanal. Nucl. Chem., 2015, vol. 303, no. 3, pp. 1821–1833. https://doi.org/10.1007/s10967-014-3688-7
Anirudhan, T.S., Jalajamony, S., and Divya, L., Ind. Eng. Chem. Res., 2009, vol. 48, pp. 2118–2124. https://doi.org/10.1021/ie8000869
Marczenko, Z., Separation and Spectrophotometric Determination of Elements, Chichester: Horwood, 1986.
Yousif, A., El-Afandy, A., Dabbour, G., and Mubark, A.E., J. Disp. Sci. Technol., 2020. https://doi.org/10.1080/01932691.2020.1844018
Dahou, W., Ghemati, D., Oudia, A., and Aliouche, D., Biochem. Eng. J., 2010, vol. 48, no. 2, pp. 187–194. https://doi.org/10.1016/j.bej.2009.10.006
Anirudhan, T.S., Jalajamony, S., and Suchihra, P.S., Colloids Surf. A, 2009, vol. 335, pp. 107–113. https://doi.org/10.1016/j.colsurfa.2008.10.035
Abd El-Magied, M.O., Elshehy, E., Manaa, E.A., Tolba, A.A., and Atia, A.A., Ind. Eng. Chem. Res., 2016, vol. 55, pp. 11338–11345. https://doi.org/10.1021/acs.iecr.6b02977
Ho, Y.S. and McKay, G.A., Inst. Chem. Eng., 1998, vol. 76, no. 4, pp. 332–340. https://doi.org/10.1205/095758298529696
Luca, V. and Hanna, J.V., Hydrometallurgy, 2015, vol. 154, pp. 118–128. https://doi.org/10.1016/j.hydromet.2015.04.002
Dolatyari, L., Yaftian, M.R., and Rostamnia, S., J. Taiwan Inst. Chem. Eng., 2015, pp. 1–11. https://doi.org/10.1016/j.jtice.2015.11.004checkpages
Kütahyal, C. and Eral, M., J. Nucl. Mater., 2010, vol. 396, pp. 251–256. https://doi.org/10.1016/j.jnucmat.2009.11.018
Mirzabe, G.H. and Keshtkar, A.R., J. Ind. Eng. Chem., 2015, vol. 26, pp. 277–285. https://doi.org/10.1016/j.jiec.2014.11.040
Khalili, F. and Al-Banna, G., J. Environ. Radioact., 2015, vol. 146, pp. 16–26. https://doi.org/10.1016/j.jenvrad.2015.03.035
Bandegharaei, A.H., Rastegar, A., Shoar, R.H., Allahabadi, A., Khamirchi, P., Sani, Z., and Mehrpouyan, M., J. Radioanal. Nucl. Chem., 2016, vol. 309, no. 2, pp. 1–16. https://doi.org/10.1007/s10967-015-4689-x
Mpofu, V.P., Addai-Mensah, J., and Ralston, J., J. Colloid Interface Sci., 2004, vol. 271, pp. 145–156. https://doi.org/10.1016/j.jcis.2003.09.042
Ji, B., Shao, F., Hu, G., Zheng, S., Zhang, Q., and Xu, Z., J. Hazard. Mater., 2009, vol. 161, pp. 81–87. https://doi.org/10.1016/j.jhazmat.2008.03.052
Salem, H., Int. J. Environ. Anal. Chem., 2020. https://doi.org/10.1080/03067319.2020.184566
Khalil, M.M.H., Atrees, M.S., Abd El Fatah, A.I.L., Salem, H., and Roshdi, R., J. Disp. Sci. Technol., 2017. https://doi.org/10.1080/01932691.2017.1370674
Zhu, X., Li, W., Zhang, Q., Zhang, C., and Chen, L., Hydrometallurgy, 2018, vol. 176, pp. 42–48. https://doi.org/10.1016/j.hydromet.2018.01.009
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Mubark, A.E., Falila, N.I. & Salem, H.M. Use of Modified Cellulose Sorbents for the Extraction of Th(IV) Ions From Chloride Solutions. Radiochemistry 63, 484–497 (2021). https://doi.org/10.1134/S1066362221040123
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1066362221040123