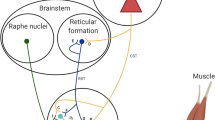
Abstract
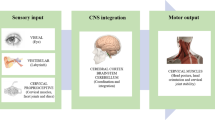
The nociceptive withdrawal reflex (NWR) threshold is commonly employed in the lower limb to assess clinical and experimentally induced pain. However, no studies to date have investigated changes in spinal nociception in the upper limb, via the NWR threshold, following experimentally induced central sensitization (CS). We tested the hypothesis that experimentally induced CS of the C5–C6 spinal segment significantly reduces NWR thresholds in muscles of the upper limb. Upper limb NWR thresholds from 20 young, healthy adults were assessed by applying noxious electrical stimuli to the right index finger and recording muscle activity from the biceps brachii (BI), triceps brachii (TRI), flexor carpi ulnaris (WF), and extensor carpi radialis longus (WE) muscles via surface electromyography. Topical cream (either 0.075% capsaicin, or control) was applied to the C5–C6 dermatome of the lateral forearm (50 cm2). NWR thresholds were compared at baseline, and four 10-min intervals after topical application. WF muscle NWR thresholds were significantly reduced in the capsaicin session compared to control, while TRI muscle NWR thresholds were significantly reduced 40 min after capsaicin application only (p < 0.05). There were no significant differences for BI or WE muscle NWR thresholds. We observed poor to moderate test–retest reliability for all upper limb NWR thresholds, a key contributor to the selective reduction in NWR thresholds among muscles. Accordingly, while our findings demonstrate some comparability to previously reported lower limb NWR studies, we concurrently report limitations of the upper limb NWR technique. Further exploration of optimal parameters for upper limb NWR acquisition is needed.





Similar content being viewed by others
Availability of data and material
Summary data are available upon request to the corresponding author.
Code availability
Matlab and R code used for data and statistical analysis can be requested through the corresponding author’s GitHub Repository.
References
Andersen OK, Gracely RH, Arendt-Nielsen L (1995) Facilitation of the human nociceptive reflex by stimulation of A beta-fibres in a secondary hyperalgesic area sustained by nociceptive input from the primary hyperalgesic area. Acta Physiol Scand 155:87–97. https://doi.org/10.1111/j.1748-1716.1995.tb09951.x
Andersen OK, Sonnenborg FA, Arendt-Nielsen L (2001) Reflex receptive fields for human withdrawal reflexes elicited by non-painful and painful electrical stimulation of the foot sole. Clin Neurophysiol 112:641–649
Arendt-Nielsen L, Morlion B, Perrot S et al (2018) Assessment and manifestation of central sensitisation across different chronic pain conditions. Eur J Pain 22:216–241. https://doi.org/10.1002/ejp.1140
Atkinson G, Nevill AM (1998) Statistical methods for assessing measurement error (reliability) in variables relevant to sports medicine. Sports Med 26:217–238. https://doi.org/10.2165/00007256-199826040-00002
Banic B, Petersen-Felix S, Andersen OK et al (2004) Evidence for spinal cord hypersensitivity in chronic pain after whiplash injury and in fibromyalgia. Pain 107:7–15. https://doi.org/10.1016/j.pain.2003.05.001
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Baumgartner U, Cruccu G, Iannetti GD, Treede RD (2005) Laser guns and hot plates. Pain 116:1–3. https://doi.org/10.1016/j.pain.2005.04.021
Biurrun Manresa JA, Neziri AY, Curatolo M et al (2011) Test–retest reliability of the nociceptive withdrawal reflex and electrical pain thresholds after single and repeated stimulation in patients with chronic low back pain. Eur J Appl Physiol 111:83–92. https://doi.org/10.1007/s00421-010-1634-0
Biurrun Manresa JA, Finnerup NS, Johannesen IL et al (2014) Central sensitization in spinal cord injured humans assessed by reflex receptive fields. Clin Neurophysiol 125:352–362. https://doi.org/10.1016/j.clinph.2013.06.186
Bland JM, Altman DG (1990) A note on the use of the intraclass correlation coefficient in the evaluation of agreement between two methods of measurement. Comput Biol Med 20:337–340. https://doi.org/10.1016/0010-4825(90)90013-f
Cambier J, Dehen H, Bathien N (1974) Upper limb cutaneous polysynaptic reflexes. J Neurol Sci 22:39–49
Culp WJ, Ochoa J, Cline M, Dotson R (1989) Heat and mechanical hyperalgesia induced by capsaicin. Cross modality threshold modulation in human C nociceptors. Brain 112(5):1317–1331
Dahlhamer J (2018) Prevalence of chronic pain and high-impact chronic pain among adults—United States, 2016. MMWR Morb Mortal Wkly Rep. https://doi.org/10.15585/mmwr.mm6736a2
De Luca CJ (1997) The use of surface electromyography in biomechanics. J Appl Biomech 13:135–163
Desmeules JA, Cedraschi C, Rapiti E et al (2003) Neurophysiologic evidence for a central sensitization in patients with fibromyalgia. Arthritis Rheum 48:1420–1429. https://doi.org/10.1002/art.10893
Floeter MK, Gerloff C, Kouri J, Hallett M (1998) Cutaneous withdrawal reflexes of the upper extremity. Muscle Nerve 21:591–598
French DJ, France CR, France JL, Arnott LF (2005) The influence of acute anxiety on assessment of nociceptive flexion reflex thresholds in healthy young adults. Pain 114:358–363. https://doi.org/10.1016/j.pain.2004.12.034
Gregory NS, Harris AL, Robinson CR et al (2013) An overview of animal models of pain: disease models and outcome measures. J Pain 14:1255–1269. https://doi.org/10.1016/j.jpain.2013.06.008
Gronroos M, Pertovaara A (1993) Capsaicin-induced central facilitation of a nociceptive flexion reflex in humans. Neurosci Lett 159:215–218. https://doi.org/10.1016/0304-3940(93)90837-B
Haefeli JS, Blum J, Steeves JD et al (2013) Differences in spinothalamic function of cervical and thoracic dermatomes: insights using contact heat evoked potentials. J Clin Neurophysiol off Publ Am Electroencephalogr Soc 30:291–298. https://doi.org/10.1097/WNP.0b013e31827ed9ee
Halekoh U, Højsgaard S (2014) A Kenward-Roger approximation and parametric bootstrap methods for tests in linear mixed models—the R package pbkrtest. J Stat Softw 1:1–32
Herm C, Silbereisen V, Graf BM, Lassen CL (2019) Long term reliability of nociceptive withdrawal reflex thresholds. J Neurosci Methods 320:44–49. https://doi.org/10.1016/j.jneumeth.2019.03.009
Hermens HJ, Freriks B, Disselhorst-Klug C, Rau G (2000) Development of recommendations for SEMG sensors and sensor placement procedures. J Electromyogr Kinesiol 10:361–374. https://doi.org/10.1016/S1050-6411(00)00027-4
Jutzeler CR, Rosner J, Rinert J et al (2016) Normative data for the segmental acquisition of contact heat evoked potentials in cervical dermatomes. Sci Rep 6:34660. https://doi.org/10.1038/srep34660
Kofler M, Leis AA, Valls-Solé J (2019) Cutaneous silent periods—part 1: update on physiological mechanisms. Clin Neurophysiol 130:588–603. https://doi.org/10.1016/j.clinph.2019.01.002
Koo TK, Li MY (2016) A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med 15:155–163. https://doi.org/10.1016/j.jcm.2016.02.012
Kugelberg E, Eklund K, Grimby L (1960) An electromyographic study of the nociceptive reflexes of the lower limb. Mechanism of the plantar responses. Brain 83:394–410. https://doi.org/10.1093/brain/83.3.394
Latremoliere A, Woolf CJ (2009) Central sensitization: a generator of pain hypersensitivity by central neural plasticity. J Pain 10:895–926. https://doi.org/10.1016/j.jpain.2009.06.012
Lee MW, McPhee RW, Stringer MD (2008) An evidence-based approach to human dermatomes. Clin Anat 21:363–373. https://doi.org/10.1002/ca.20636
Leis AA, Stokic DS, Fuhr P et al (2000) Nociceptive fingertip stimulation inhibits synergistic motoneuron pools in the human upper limb. Neurology 55:1305–1309. https://doi.org/10.1212/WNL.55.9.1305
Linde LD, Srbely JZ (2019) The acute effect of skin pre-heating on capsaicin induced central sensitization in humans. Pain Pr 19:811–820. https://doi.org/10.1111/papr.12811
Linde LD, Duarte FC, Esmaeili H et al (2020) The nociceptive flexion reflex: a scoping review and proposed standardized methodology for acquisition in those affected by chronic pain. Br J Pain. https://doi.org/10.1177/2049463720913289
Loeser JD, Arendt-Nielsen L, Baron R et al (2011) Pain terms, a current list with definitions and notes on usage. Classif Chronic Pain. https://doi.org/10.1016/0304-3959(86)90113-2
Micalos PS, Drinkwater EJ, Cannon J et al (2009) Reliability of the nociceptive flexor reflex (RIII) threshold and association with Pain threshold. Eur J Appl Physiol 105:55–62. https://doi.org/10.1007/s00421-008-0872-x
Peterson CL, Riley ZA, Krepkovich ET et al (2014) Withdrawal reflexes in the upper limb adapt to arm posture and stimulus location. Muscle Nerve 49:716–723. https://doi.org/10.1002/mus.23987
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Rhudy JL, France CR (2011) Reliability and validity of a brief method to assess nociceptive flexion reflex (NFR) threshold. J Pain 12:782–791. https://doi.org/10.1016/j.jpain.2011.01.002
Rice DA, McNair PJ, Lewis GN, Dalbeth N (2015) The effects of joint aspiration and intra-articular corticosteroid injection on flexion reflex excitability, quadriceps strength and pain in individuals with knee synovitis: a prospective observational study. Arthritis Res Ther 17:191. https://doi.org/10.1186/s13075-015-0711-5
Rosner J, Hubli M, Hostettler P et al (2018) Contact heat evoked potentials: reliable acquisition from lower extremities. Clin Neurophysiol 129:584–591. https://doi.org/10.1016/j.clinph.2017.12.034
Serrao M, Ranavolo A, Andersen OK et al (2012) Reorganization of multi-muscle and joint withdrawal reflex during arm movements in post-stroke hemiparetic patients. Clin Neurophysiol 123:527–540. https://doi.org/10.1016/j.clinph.2011.07.031
Shechtman O (2013) The coefficient of variation as an index of measurement reliability. In: Doi SAR, Williams GM (eds) Methods of clinical epidemiology. Springer, Berlin, Heidelberg, pp 39–49
Shrout PE, Fleiss JL (1979) Intraclass correlations: uses in assessing rater reliability. Psychol Bull 86:420–428. https://doi.org/10.1037/0033-2909.86.2.420
Shupler MS, Kramer JK, Cragg JJ et al (2019) Pan-Canadian estimates of chronic pain prevalence from 2000 to 2014: a repeated cross-sectional survey analysis. J Pain 20:557–565. https://doi.org/10.1016/j.jpain.2018.10.010
Simone DA, Baumann TK, LaMotte RH (1989) Dose-dependent pain and mechanical hyperalgesia in humans after intradermal injection of capsaicin. Pain 38:99–107. https://doi.org/10.1016/0304-3959(89)90079-1
Sirucek L, Jutzeler CR, Rosner J et al (2020) The effect of conditioned pain modulation on tonic heat pain assessed using participant-controlled temperature. Pain Med Malden Mass. https://doi.org/10.1093/pm/pnaa041
Smith AD, Jull G, Schneider G et al (2013) A comparison of physical and psychological features of responders and non-responders to cervical facet blocks in chronic whiplash. BMC Musculoskelet Disord 14:313. https://doi.org/10.1186/1471-2474-14-313
Smith AD, Jull G, Schneider G et al (2014) Cervical radiofrequency neurotomy reduces central hyperexcitability and improves neck movement in individuals with chronic whiplash. Pain Med 15:128–141. https://doi.org/10.1111/pme.12262
Sorkin LS, Wallace MS (1999) Acute pain mechanisms. Surg Clin North Am 79:213–229. https://doi.org/10.1016/S0039-6109(05)70380-7
Srbely JZ, Dickey JP, Bent LR et al (2010) Capsaicin-induced central sensitization evokes segmental increases in trigger point sensitivity in humans. J Pain 11:636–643. https://doi.org/10.1016/j.jpain.2009.10.005
Sterling M, Pedler A, Chan C et al (2010) Cervical lateral glide increases nociceptive flexion reflex threshold but not pressure or thermal pain thresholds in chronic whiplash associated disorders: a pilot randomised controlled trial. Man Ther 15:149–153. https://doi.org/10.1016/j.math.2009.09.004
Truini A, Galeotti F, Romaniello A et al (2005) Laser-evoked potentials: normative values. Clin Neurophysiol 116:821–826. https://doi.org/10.1016/j.clinph.2004.10.004
Vernon H (2012) What is different about spinal pain? Chiropr Man Ther 20:22. https://doi.org/10.1186/2045-709X-20-22
von Dincklage F, Olbrich H, Baars JH, Rehberg B (2013) Habituation of the nociceptive flexion reflex is dependent on inter-stimulus interval and stimulus intensity. J Clin Neurosci 20:848–850. https://doi.org/10.1016/j.jocn.2012.07.013
Willer JC (1977) Comparative study of perceived pain and nociceptive flexion reflex in man. Pain 3:69–80
Wilson MG, Lavis JN, Ellen ME (2015) Supporting chronic pain management across provincial and territorial health systems in Canada: Findings from two stakeholder dialogues. Pain Res Manag 20:269–279. https://doi.org/10.1155/2015/918976
Witting N, Svensson P, Gottrup H et al (2000) Intramuscular and intradermal injection of capsaicin: a comparison of local and referred pain. Pain 84:407–412. https://doi.org/10.1016/s0304-3959(99)00231-6
Acknowledgements
We would like to thank all of the individuals who participated in this study.
Funding
JS is supported by a Natural Sciences and Engineering Research Council of Canada Discovery grant, which contributed to this project. No other authors have funding to declare in support of this study.
Author information
Authors and Affiliations
Contributions
LL was responsible for original study inception, data collection, analysis, interpretation, statistical analysis, and manuscript preparation. LB, JD, and DA contributed study inception, the development of methodology, and critically reviewed the manuscript. JS supervised all aspects of the study.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethics approval
The University of Guelph research ethics board approved all study protocols.
Consent to participate
All participants provided informed, written consent prior to study participation.
Consent for publication
All participants consented to inclusion of their data for publication.
Additional information
Communicated by Winston D Byblow.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Linde, L.D., Bent, L.R., Dickey, J.P. et al. Exploring the effect of capsaicin-induced central sensitization on the upper limb nociceptive withdrawal reflex threshold. Exp Brain Res 239, 3405–3415 (2021). https://doi.org/10.1007/s00221-021-06216-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-021-06216-3