Abstract
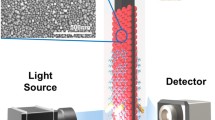
Plasmonic nanosensors for label-free detection of DNA require excellent sensing resolution, which is crucial when monitoring short DNA sequences, as these induce tiny peak shifts, compared to large biomolecules. We report a versatile and simple strategy for plasmonic sensor signal enhancement by assembling multiple (four) plasmonic sensors in series. This approach provided a fourfold signal enhancement, increased signal-to-noise ratio, and improved sensitivity for DNA detection. The response of multiple sensors based on AuNSpheres was also compared with AuNRods, the latter showing better sensing resolution. The amplification system based on AuNR was integrated into a microfluidic sequential injection platform and applied to the monitoring of DNA, specifically from environmental invasive species—zebra mussels. DNA from zebra mussels was log concentration-dependent from 1 to 1 × 106 pM, reaching a detection limit of 2.0 pM. In situ tests were also successfully applied to real samples, within less than 45 min, using DNA extracted from zebra mussel meat. The plasmonic nanosensors’ signal can be used as a binary output (yes/no) to assess the presence of those invasive species. Even though these genosensors were applied to the monitoring of DNA in environmental samples, they potentially offer advantage in a wide range of fields, such as disease diagnostics.
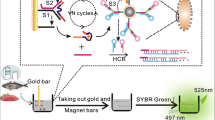
Graphical abstract





Similar content being viewed by others
References
Willets KA, Van Duyne RP (2007) Localized surface plasmon resonance spectroscopy and sensing. Annu Rev Phys Chem 58:267–297. https://doi.org/10.1146/annurev.physchem.58.032806.104607
Guo L, Jackman JA, Yang HH, Chen P, Cho NJ, Kim DH (2015) Strategies for enhancing the sensitivity of plasmonic nanosensors. Nano Today 10:213–239. https://doi.org/10.1016/j.nantod.2015.02.007
Murray WA, Barnes WL (2007) Plasmonic materials. Adv Mater 19:3771–3782. https://doi.org/10.1002/adma.200700678
Scuderi M, Esposito M, Todisco F, Simeone D, Tarantini I, de Marco L, de Giorgi M, Nicotra G, Carbone L, Sanvitto D, Passaseo A, Gigli G, Cuscunà M (2016) Nanoscale study of the tarnishing process in electron beam lithography-fabricated silver nanoparticles for plasmonic applications. J Phys Chem C 120:24314–24323. https://doi.org/10.1021/acs.jpcc.6b03963
Haynes CL, Van Duyne RP (2001) Nanosphere lithography: a versatile nanofabrication tool for studies of size-dependent nanoparticle optics. J Phys Chem B 105:5599–5611. https://doi.org/10.1021/jp010657m
Hanarp P, Kall M, Sutherland DS (2003) Optical properties of short range ordered arrays of nanometer gold disks prepared by colloidal lithography. J Phys Chem B 107:5768–5772. https://doi.org/10.1021/jp027562k
Rahim FA, Dong-Hwan K (2016) Nanoparticle polymer composites on solid substrates for plasmonic sensing applications. Nano Today 11:415–434. https://doi.org/10.1016/j.nantod.2016.07.001
Barsan MM, Brett CMA (2016) Recent advances in layer-by-layer strategies for biosensors incorporating metal nanoparticles. Trac-Trends Anal Chem 79:286–296. https://doi.org/10.1016/j.trac.2015.11.019
Grzelczak M, Pérez-Juste J, Mulvaney P, Liz-Marzán LM (2008) Shape control in gold nanoparticle synthesis. Chem Soc Rev 37:1783–1791. https://doi.org/10.1039/b711490g
Scarabelli L, Sanchez-Iglesias A, Perez-Juste J, Liz-Marzan LM (2015) A “tips and tricks” practical guide to the synthesis of gold nanorods. J Phys Chem Lett 6:4270–4279. https://doi.org/10.1021/acs.jpclett.5b02123
Navarro JRG, Lerouge F (2017) From gold nanoparticles to luminescent nano-objects: experimental aspects for better gold-chromophore interactions. Nanophotonics 6:71–92. https://doi.org/10.1515/nanoph-2015-0143
Guerreiro JRL, Frederiksen M, Bochenkov VE, de Freitas V, Ferreira Sales MG, Sutherland DS (2014) Multifunctional biosensor based on localized surface plasmon resonance for monitoring small molecule-protein interaction. ACS Nano 8:7958–7967. https://doi.org/10.1021/nn501962y
Morsin M, Umar AA, Salleh MM, et al (2012) High sensitivity localized surface plasmon resonance sensor of gold nanoparticles : surface density effect for detection of boric acid. 10th IEEE Int Conf Semicond Electron 352–356. https://doi.org/10.1109/SMElec.2012.6417158
Tu MH, Sun T, Grattan KTV (2012) Optimization of gold-nanoparticle-based optical fibre surface plasmon resonance (SPR)-based sensors. Sensors and Actuators B-Chemical 164:43–53. https://doi.org/10.1016/j.snb.2012.01.060
Lowe S, Browne M, Boudjelas S, De Poorter M (2000) 100 of the world’s worst invasive alien species a selection from the global invasive species database. Invasive Species Spec Gr
Carvalho J, Garrido-Maestu A, Azinheiro S, Fuciños P, Barros-Velázquez J, de Miguel RJ, Gros V, Prado M (2021) Faster monitoring of the invasive alien species (IAS) Dreissena polymorpha in river basins through isothermal amplification. Sci Rep 11:10175. https://doi.org/10.1038/s41598-021-89574-w
EU (2014) Regulation (EU) No 1143/2014 of the European Parliament and of the Council of 22 October 2014 on the prevention and management of the introduction and spread of invasive alien species. 35–55
Wang YY, Tang L (2013) Chemisorption assembly of Au nanorods on mercaptosilanized glass substrate for label-free nanoplasmon biochip. Anal Chim Acta 796:122–129. https://doi.org/10.1016/j.aca.2013.08.024
Chen HJ, Shao L, Li Q, Wang JF (2013) Gold nanorods and their plasmonic properties. Chem Soc Rev 42:2679–2724. https://doi.org/10.1039/c2cs35367a
Malinsky DM, Kelly KL, Schatz GC, Van Duyne RP (2001) Nanosphere lithography: effect of substrate on the localized surface plasmon resonance spectrum of silver nanoparticles. J Phys Chem B 105:2343–2350. https://doi.org/10.1021/jp002906x
Ray PC, Fan Z, Crouch RA, Sinha SS, Pramanik A (2014) Nanoscopic optical rulers beyond the FRET distance limit: fundamentals and applications. Chem Soc Rev 43:6370–6404. https://doi.org/10.1039/C3CS60476D
Chen H, Kou X, Yang Z, Ni W, Wang J (2008) Shape- and size-dependent refractive index sensitivity of gold nanoparticles. Langmuir 24:5233–5237. https://doi.org/10.1021/la800305j
Nehl CL, Liao HW, Hafner JH (2006) Optical properties of star-shaped gold nanoparticles. Nano Lett 6:683–688. https://doi.org/10.1021/nl052409y
Lee KS, El-Sayed MA (2006) Gold and silver nanoparticles in sensing and imaging: sensitivity of plasmon response to size, shape, and metal composition. J Phys Chem B 110:19220–19225. https://doi.org/10.1021/jp062536y
Murray WA, Auguie B, Barnes WL (2009) Sensitivity of localized surface plasmon resonances to bulk and local changes in the optical environment. J Phys Chem C 113:5120–5125. https://doi.org/10.1021/jp810322q
Steel AB, Levicky RL, Herne TM, Tarlov MJ (2000) Immobilization of nucleic acids at solid surfaces : effect of oligonucleotide length on layer assembly. Biophys J 79:975–981. https://doi.org/10.1016/S0006-3495(00)76351-X
Sohreiner SM, Shudy DF, Hatoh AL et al (2010) Controlled and efficient hybridization achieved with DNA probes immobilized solely through preferential DNA-substrate interactions. Anal Chem 82:2803–2810. https://doi.org/10.1021/ac902765g
Lu W, Wang L, Li J, Zhao Y, Zhou Z, Shi J, Zuo X, Pan D (2015) Quantitative investigation of the poly-adenine DNA dissociation from the surface of gold nanoparticles. Sci Rep 5:1–9. https://doi.org/10.1038/srep10158
Herne TM, Tarlov MJ (1997) Characterization of DNA probes immobilized on gold surfaces. J Am Chem Soc 119:8916–8920. https://doi.org/10.1021/ja9719586
Bonya A (2020) Large scale fabrication of ordered gold nanoparticle − epoxy surface nanocomposites and their application as label-free plasmonic DNA biosensors. ACS Appl Mater Interfaces 12:4804–4814. https://doi.org/10.1021/acsami.9b20907
Guerreiro JRL, Teixeira N, De Freitas V et al (2017) A saliva molecular imprinted localized surface plasmon resonance biosensor for wine astringency estimation. Food Chem 233:457–466. https://doi.org/10.1016/j.foodchem.2017.04.051
Masterson AN, Liyanage T, Kaimakliotis H, Gholami Derami H, Deiss F, Sardar R (2020) Bottom-up fabrication of plasmonic nanoantenna-based high-throughput multiplexing biosensors for ultrasensitive detection of microRNAs directly from cancer patients’ plasma. Anal Chem 92:9295–9304. https://doi.org/10.1021/acs.analchem.0c01639
Joshi GK, Deitz-Mcelyea S, Johnson M et al (2014) Highly specific plasmonic biosensors for ultrasensitive microRNA detection in plasma from pancreatic cancer patients. Nano Lett 14:6955–6963. https://doi.org/10.1021/nl503220s
Roether J, Chu KY, Willenbacher N, Shen AQ, Bhalla N (2019) Real-time monitoring of DNA immobilization and detection of DNA polymerase activity by a microfluidic nanoplasmonic platform. Biosens Bioelectron 142:111528. https://doi.org/10.1016/j.bios.2019.111528
Zhu S, Li H, Yang M, Pang SW (2018) Label-free detection of live cancer cells and DNA hybridization using 3D multilayered plasmonic biosensor. Nanotechnology 29:365503. https://doi.org/10.1088/1361-6528/aac8fb
Miti A, Thamm S, Müller P, Csáki A, Fritzsche W, Zuccheri G (2020) A miRNA biosensor based on localized surface plasmon resonance enhanced by surface-bound hybridization chain reaction. Biosens Bioelectron 167:112465. https://doi.org/10.1016/j.bios.2020.112465
Nguyen AH, Sim SJ (2015) Nanoplasmonic biosensor: detection and amplification of dual bio-signatures of circulating tumor DNA. Biosens &. Bioelectron 67:443–449. https://doi.org/10.1016/j.bios.2014.09.003
Funding
This work was supported by project Nanotechnology Based Functional Solutions (NORTE-01-0145-FEDER-000019), supported by Norte Portugal Regional Operational Programme (NORTE2020), under the PORTUGAL 2020 Partnership Agreement, through the European Regional Development Fund, and by the Partnership Agreement with Confederación Hidrográfica del Guadalquivir (Spain) for the development of a system of early detection of the zebra mussel through analysis of environmental DNA.
Author information
Authors and Affiliations
Contributions
Experiments were conducted by J.R. Guerreiro and A. Toldrà. Flow cell design and fabrication, as well as microfluidic assembly and software development, were conducted by A. Ipatov. Zebra mussel-specific DNA sequence identification and extraction/preparation of real samples were conducted by J. Carvalho. Conceptualization and method development were designed by J.R. Guerreiro and M. Prado. Writing—original draft preparation, was conducted by J.R. Guerreiro. M. Prado participated on project administration and funding acquisition. Writing—reviewing and editing, was contributed by J.R. Guerreiro, A. Ipatov, J. Carvalho, A. Toldrà, and M. Prado.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 5390 kb)
Rights and permissions
About this article
Cite this article
Guerreiro, J.R.L., Ipatov, A., Carvalho, J. et al. Amplified plasmonic and microfluidic setup for DNA monitoring. Microchim Acta 188, 326 (2021). https://doi.org/10.1007/s00604-021-04983-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-021-04983-y