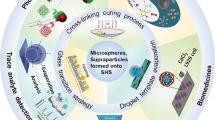
Abstract
Cationic polymers with primary amine groups that can easily be functionalized or coupled with substrates by complexation or hydrogen bonding are especially advantageous in preparing particles for biomedical applications. Poly(vinyl amine) (PVAm) is a cationic polyelectrolyte containing the highest number of primary amine groups among any other polymers. Here, we introduce a general method in synthesizing PVAm microparticles via a surfactant-free water-in-oil emulsion technique using cyclohexane as the oil phase and aqueous PVAm solution as the dispersed phase. PVAm particles were prepared to employ two different bifunctional chemical crosslinkers, divinyl sulfone (DVS) and poly(ethylene glycol) diglycidyl ether (PEGGE). The prepared particles were further treated with HCl to protonate the amine groups of PVAm within particles. The effect of crosslinker types and pH on the hydrolytic degradation of PVAm particles were also investigated at three different solution pHs, 5.4, 7.4, and 9, to simulate the skin, blood, and intestinal pH environments, respectively. The blood compatibility of the PVAm particles was evaluated by in vitro hemolysis and blood clotting assays. Furthermore, antifungal and antibacterial efficacy of PVAm-based particles and their protonated forms were tested against C. albicans yeast and E. coli, S. aureus, B. subtilis, and P. aeruginosa bacterial strains.






Similar content being viewed by others
References
Yamamoto K, Imamura Y, Nagatomo E et al (2003) Synthesis and functionalities of poly(N-vinylalkylamide). XIV. Polyvinylamine produced by hydrolysis of poly(N-vinylformamide) and its functionalization. J Appl Polym Sci 89:1277–1283. https://doi.org/10.1002/app.12230
Pinschmidt RK (2010) Polyvinylamine at last. J Polym Sci Part A Polym Chem 48:2257–2283. https://doi.org/10.1002/pola.23992
Martel B, Morcellet M (1995) Cyclodextrin-poly(vinylamine) systems—II Catalytic hydrolysis of p-nitrophenyl acetate. Eur Polym J 31:1089–1093
Illergård J, Römling U, Wågberg L, Ek M (2012) Biointeractive antibacterial fibres using polyelectrolyte multilayer modification. Cellulose 19:1731–1741. https://doi.org/10.1007/s10570-012-9742-0
Seifert S, Simon F, Baumann G et al (2011) Adsorption of poly(vinyl formamide- co -vinyl amine) (PVFA-co-PVAm) polymers on zinc, zinc oxide, iron, and iron oxide surfaces. Langmuir 27:14279–14289. https://doi.org/10.1021/la203479n
Hollertz R, Durán VL, Larsson PA, Wågberg L (2017) Chemically modified cellulose micro- and nanofibrils as paper-strength additives. Cellulose 24:3883–3899. https://doi.org/10.1007/s10570-017-1387-6
Huang Y, Du J, Zhang Y et al (2016) Batch process of polymer-enhanced ultrafiltration to recover mercury (II) from wastewater. J Memb Sci 514:229–240. https://doi.org/10.1016/j.memsci.2016.04.060
Casadei R, Venturi D, Baschetti MG et al (2019) Polyvinylamine membranes containing graphene-based nanofillers for carbon capture applications. Membranes (Basel) 9:119. https://doi.org/10.3390/membranes9090119
Dréan M, Debuigne A, Goncalves C et al (2017) Use of Primary and Secondary Polyvinylamines for Efficient Gene Transfection. Biomacromol 18:440–451. https://doi.org/10.1021/acs.biomac.6b01526
Khondee S, Yakovleva T, Berkland C (2010) Low charge polyvinylamine nanogels offer sustained, low-level gene expression. J Appl Polym Sci 118:1921–1932. https://doi.org/10.1002/app.32460
Chen Y, Sun P (2019) pH-Sensitive Polyampholyte Microgels of Poly(Acrylic Acid-co-Vinylamine) as Injectable Hydrogel for Controlled Drug Release. Polymers (Basel) 11:285. https://doi.org/10.3390/polym11020285
Henschen J, Larsson PA, Illergård J et al (2017) Bacterial adhesion to polyvinylamine-modified nanocellulose films. Colloids Surfaces B Biointerfaces 151:224–231. https://doi.org/10.1016/j.colsurfb.2016.12.018
Chen C, Petterson T, Illergård J et al (2019) Influence of Cellulose Charge on Bacteria Adhesion and Viability to PVAm/CNF/PVAm-Modified Cellulose Model Surfaces. Biomacromol 20:2075–2083. https://doi.org/10.1021/acs.biomac.9b00297
Marini M, Bondi M, Iseppi R et al (2007) Preparation and antibacterial activity of hybrid materials containing quaternary ammonium salts via sol–gel process. Eur Polym J 43:3621–3628
Yang Y, Cai Z, Huang Z et al (2018) Antimicrobial cationic polymers: From structural design to functional control. Polym J 50:33–44. https://doi.org/10.1038/pj.2017.72
Palermo EF, Kuroda K (2009) Chemical structure of cationic groups in amphiphilic polymethacrylates modulates the antimicrobial and hemolytic activities. Biomacromol 10:1416–1428. https://doi.org/10.1021/bm900044x
Chen Z, Lv Z, Sun Y et al (2020) Recent advancements in polyethyleneimine-based materials and their biomedical, biotechnology, and biomaterial applications. J Mater Chem B 8:2951–2973. https://doi.org/10.1039/c9tb02271f
Thaiboonrod S, Berkland C, Milani AH et al (2013) Poly(vinylamine) microgels: PH-responsive particles with high primary amine contents. Soft Matter 9:3920–3930. https://doi.org/10.1039/c3sm27728c
Thaiboonrod S, Milani AH, Saunders BR (2014) Doubly crosslinked poly(vinyl amine) microgels: Hydrogels of covalently inter-linked cationic microgel particles. J Mater Chem B 2:110–119. https://doi.org/10.1039/c3tb21579b
McCann J, Behrendt JM, Yan J et al (2015) Poly(vinylamine) microgel-dextran composite hydrogels: Characterisation; properties and pH-triggered degradation. J Colloid Interface Sci 449:21–30. https://doi.org/10.1016/j.jcis.2014.09.041
Shi L, Berkland C (2007) Acid-labile polyvinylamine micro- and nanogel capsules. Macromolecules 40:4635–4643. https://doi.org/10.1021/ma070443o
Bonnefond A, Pereira Gomes C, de la Cal JC, Leiza JR (2017) Surfactant-free poly(methyl methacrylate)/poly(vinylamine) (PMMA/PVAm) amphiphilic core-shell polymer particles. Colloid Polym Sci 295:135–144. https://doi.org/10.1007/s00396-016-3985-5
Gu L, Zhu S, Hrymak AN, Pelton RH (2001) Kinetics and modeling of free radical polymerization of N-vinylformamide. Polymer (Guildf) 42:3077–3086. https://doi.org/10.1016/S0032-3861(00)00654-6
Gu L, Zhu S, Hrymak AN (2002) Acidic and basic hydrolysis of poly(N-vinylformamide). J Appl Polym Sci 86:3412–3419. https://doi.org/10.1002/app.11364
Horikoshi S, Akao Y, Ogura T et al (2010) On the stability of surfactant-free water-in-oil emulsions and synthesis of hollow SiO2 nanospheres. Colloids Surfaces A Physicochem Eng Asp 372:55–60. https://doi.org/10.1016/j.colsurfa.2010.09.036
Can M, Ayyala RS, Sahiner N (2019) Crosslinked poly(Lactose) microgels and nanogels for biomedical applications. J Colloid Interface Sci 553:805–812. https://doi.org/10.1016/j.jcis.2019.06.078
Reynolds DD, Kenyon WO (1947) The Preparation of Polyvinylamine, Polyvinylamine Salts, and Related Nitrogenous Resins
Dawson DJ, Gless RD, Wingard RE (1976) Poly(vinylamine hydrochloride). Synthesis and Utilization for the Preparation of Water-Soluble Polymeric Dyes. J Am Chem Soc 98:5996–6000. https://doi.org/10.1021/ja00435a036
Tanaka H, Senju R (1976) Preparation of polyvinylamine by the Hoffmann degradation of polyacrylamide. Bull Chem Soc Jpn 49:2821–2823
Yatabe R, Onodera T, Toko K (2013) Highly sensitive detection of 2,4,6-trinitrotoluene(TNT) using poly(vinylamine-co-N-vinylformamide)based surface plasmon resonance (SPR) immunosensor. Sensors Mater 25:45–56
Pham HQ, Marks MJ (2005) Epoxy Resins. In: Ullmann’s Encyclopedia of Industrial Chemistry. Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, Germany
Demirci S, Sahiner N (2014) PEI-based ionic liquid colloids for versatile use: Biomedical and environmental applications. J Mol Liq 194:85–92. https://doi.org/10.1016/j.molliq.2014.01.015
Sahiner N, Demirci S (2016) Poly ionic liquid cryogel of polyethyleneimine: Synthesis, characterization, and testing in absorption studies. J Appl Polym Sci. https://doi.org/10.1002/app.43478
Pei Y, Zhao L, Du G et al (2016) Investigation of the degradation and stability of acrylamide-based polymers in acid solution: Functional monomer modified polyacrylamide. Petroleum 2:399–407. https://doi.org/10.1016/j.petlm.2016.08.006
Chen Y, Zhang Y (2011) Fluorescent quantification of amino groups on silica nanoparticle surfaces. Anal Bioanal Chem 399:2503–2509. https://doi.org/10.1007/s00216-010-4622-7
Pastor-Pérez L, Chen Y, Shen Z et al (2007) Unprecedented blue intrinsic photoluminescence from hyperbranched and linear polyethylenimines: Polymer architectures and pH-effects. Macromol Rapid Commun 28:1404–1409. https://doi.org/10.1002/marc.200700190
Sütekin SD, Demirci S, Kurt SB et al (2021) Tunable fluorescent and antimicrobial properties of poly(vinyl amine) affected by the acidic or basic hydrolysis of poly(N-vinylformamide). J Appl Polym Sci 51234:1–16. https://doi.org/10.1002/app.51234
Bazan GC, Miao YJ, Renak ML, Sun BJ (1996) Fluorescence quantum yield of oly(p-phenylenevinylene) prepared via the paracyclophene route: Effect of chain length and interchain contacts. J Am Chem Soc 118:2618–2624. https://doi.org/10.1021/ja953716g
Maity S, Shyamal M, Das D et al (2018) Proton triggered emission and selective sensing of 2,4,6-trinitrophenol using a fluorescent hydrosol of 2-phenylquinoline. New J Chem 42:1879–1891. https://doi.org/10.1039/c7nj03861e
Sailema-Palate GP, Vidaurre A, Campillo-Fernández AJ, Castilla-Cortázar I (2016) A comparative study on Poly(ε-caprolactone) film degradation at extreme pH values. Polym Degrad Stab 130:118–125. https://doi.org/10.1016/j.polymdegradstab.2016.06.005
Drozdov AD, Declaville Christiansen J (2015) Modeling the effects of pH and ionic strength on swelling of polyelectrolyte gels. J Chem Phys. https://doi.org/10.1063/1.4914924
de la Harpe KM, Kondiah PPD, Choonara YE et al (2019) The Hemocompatibility of Nanoparticles: A Review of Cell-Nanoparticle Interactions and Hemostasis. Cells 8:1209. https://doi.org/10.3390/cells8101209
Klajnert B, Pikala S, Bryszewska M (2010) Haemolytic activity of polyamidoamine dendrimers and the protective role of human serum albumin. Proc R Soc A Math Phys Eng Sci 466:1527–1534. https://doi.org/10.1098/rspa.2009.0050
Palermo EF, Sovadinova I, Kuroda K (2009) Structural Determinants of Antimicrobial Activity and Biocompatibility in Membrane-Disrupting Methacrylamide Random Copolymers. Biomacromol 10:3098–3107. https://doi.org/10.1021/bm900784x
Ilker MF, Nüsslein K, Tew GN, Coughlin EB (2004) Tuning the Hemolytic and Antibacterial Activities of Amphiphilic Polynorbornene Derivatives. J Am Chem Soc 126:15870–15875. https://doi.org/10.1021/ja045664d
Carmona-Ribeiro A, de Melo CL (2013) Cationic Antimicrobial Polymers and Their Assemblies. Int J Mol Sci 14:9906–9946. https://doi.org/10.3390/ijms14059906
Kennedy DC, Orts-Gil G, Lai C-H et al (2014) Carbohydrate functionalization of silver nanoparticles modulates cytotoxicity and cellular uptake. J Nanobiotechnology 12:59. https://doi.org/10.1186/s12951-014-0059-z
Leigue L, Montiani-Ferreira F, Moore BA (2016) Antimicrobial susceptibility and minimal inhibitory concentration of Pseudomonas aeruginosa isolated from septic ocular surface disease in different animal species. Open Vet J 6:215. https://doi.org/10.4314/ovj.v6i3.9
Palermo EF, Lee D-K, Ramamoorthy A, Kuroda K (2011) Role of Cationic Group Structure in Membrane Binding and Disruption by Amphiphilic Copolymers. J Phys Chem B 115:366–375. https://doi.org/10.1021/jp1083357
Helmy S, Mohamed SS, Mahmoud SS, Talaat M (2015) Preparation and Characterization of DMPC Liposomal-Gentamicin; Antibacterial Time-Kill Study on Escherichia coli ATCC 8739. Int J Innov Sci Eng Technol 3:221–224
Leite AM, Lima EDO, De Souza EL et al (2007) Inhibitory effect of β-pinene, α-pinene and eugenol on the growth of potential infectious endocarditis causing Gram-positive bacteria. Rev Bras Ciencias Farm J Pharm Sci 43:121–126. https://doi.org/10.1590/S1516-93322007000100015
Lu M, Yu C, Cui X et al (2018) Gentamicin synergises with azoles against drug-resistant Candida albicans. Int J Antimicrob Agents 51:107–114. https://doi.org/10.1016/j.ijantimicag.2017.09.012
Adimpong DB, Sørensen KI, Thorsen L et al (2012) Antimicrobial susceptibility of bacillus strains isolated from primary starters for african traditional bread production and characterization of the bacitracin operon and bacitracin biosynthesis. Appl Environ Microbiol 78:7903–7914. https://doi.org/10.1128/AEM.00730-12
Machado I, Graça J, Lopes H et al (2013) Antimicrobial Pressure of Ciprofloxacin and Gentamicin on Biofilm Development by an Endoscope-Isolated Pseudomonas aeruginosa. ISRN Biotechnol 2013:1–10. https://doi.org/10.5402/2013/178646
Yamamoto O (2001) Influence of particle size on the antibacterial activity of zinc oxide. Int J Inorg Mater 3:643–646. https://doi.org/10.1016/S1466-6049(01)00197-0
Westman E-H, Ek M, Enarsson L-E, Wågberg L (2009) Assessment of Antibacterial Properties of Polyvinylamine (PVAm) with Different Charge Densities and Hydrophobic Modifications. Biomacromol 10:1478–1483. https://doi.org/10.1021/bm900088r
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Demirci, S., Sütekin, S.D., Kurt, S.B. et al. Poly(vinyl amine) microparticles derived from N-Vinylformamide and their versatile use. Polym. Bull. 79, 7729–7751 (2022). https://doi.org/10.1007/s00289-021-03874-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-021-03874-9