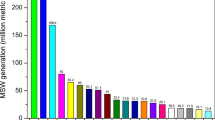
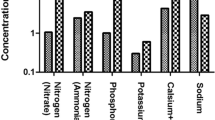
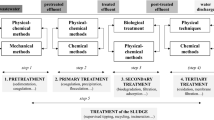
Abstract
Acid mine drainage (AMD) is the biggest environmental problem of water related to the mining industry in many countries. Recently, coal mining and mineral mining industries use various AMD treatment systems. The main problem, however, is the very expensive and long retention times for treatment of AMD. For this reason, biosorption technology has been tested recently as a promising method for AMD treatment. The authors review presently available or potential biosorption methods to remove and detoxify toxic heavy metals and organic pollutants from AMD. Biosorption technology is evolving as an attractive option to supplement conventional AMD treatment. However, a literature review indicates that there is a lack of studies examining biosorption of AMD treatment. This paper examines a range of subjects, including laboratory scale experiments and the commercial application of biosorption for treatment of AMD. Our review paper will help direct future studies on the development of biosorption technology for treating AMD.

Similar content being viewed by others
References
Akcil A, Koldas S (2006) Acid mine drainage (AMD): causes, treatment and case studies. J Clean Prod. 14:1139–1145. https://doi.org/10.1016/j.jclepro.2004.09.006
Aksu Z (2005) Application of biosorption for the removal of organic pollutants: A review. Process Biochem 40:997–1026. https://doi.org/10.1016/j.procbio.2004.04.008
Alcolea A, Vázquez M, Caparrós A, Ibarra I, García C, Linares R, Rodríguez R (2012) Heavy metal removal of intermittent acid mine drainage with an open limestone channel. Miner Eng 26:86–98
Barrera H, Urena-Nunez F, Bilyeu B, Barrera-Diaz C (2006) Removal of chromium and toxic ions present in mine drainage by Ectodermis of Opuntia. J Hazard Mater 136:846–853. https://doi.org/10.1016/j.jhazmat.2006.01.021
Barton CD, Karathanasis AD (1999) Renovation of a failed constructed wetland treating acid mine drainage. Environ Geol 39:39–50
Beni AA, Esmaeili A (2020) Biosorption, an efficient method for removing heavy metals from industrial effluents: A Review. Environ Technol Innov. https://doi.org/10.1016/j.eti.2019.100503
Benner SG, Blowes DW, Gould WD, Herbert RB, Ptacek CJ (1999) Geochemistry of a permeable reactive barrier for metals and acid mine drainage. Environ Sci Technol 33:2793–2799. https://doi.org/10.1021/es981040u
Cantuaria ML, de Almeida Neto AF, Nascimento ES, Vieira MGA (2016) Adsorption of silver from aqueous solution onto pre-treated bentonite clay: complete batch system evaluation. J Clean Prod 112(1):1112–1121. https://doi.org/10.1016/j.jclepro.2015.07.021
Champagne P, Van Geel P, Parker W (2008) Impact of temperature and loading on the mitigation of AMD in peat biofilter columns. Mine Water Environ 27:225. https://doi.org/10.1007/s10230-008-0053-5
Chen T, Yan B, Lei C, Xiao X (2014) Pollution control and metal resource recovery for acid mine drainage. Hydrometallurgy 147–148:112–119. https://doi.org/10.1016/j.hydromet.2014.04.024
Chockalingam E, Subramanian S (2009) Utility of Eucalyptus tereticornis (Smith) bark and Desulfotomaculum nigrificans for the remediation of acid mine drainage. Bioresource Technol. 100:615–621. https://doi.org/10.1016/j.biortech.2008.07.004
Chockalingam E, Subramanian S (2006) Studies on removal of metal ions and sulphate reduction using rice husk and Desulfotomaculum nigrificans with reference to remediation of acid mine drainage. Chemosphere 62:699–708. https://doi.org/10.1016/j.chemosphere.2005.05.013
Choi HJ, Lee SM (2015) Heavy metal removal from acid mine drainage by calcined eggshell and microalgae hybrid system. Environ Sci Pollut R 22:13404–13411. https://doi.org/10.1007/s11356-015-4623-3
Christoforidis AK, Orfanidis S, Papageorgiou SK, Lazaridou AN, Favvas EP, Mitropoulos AC (2015) Study of Cu(II) removal by Cystoseira crinitophylla biomass in batch and continuous flow biosorption. Chem Eng J. 277:334–340. https://doi.org/10.1016/j.cej.2015.04.138
Côrtes LN, Tanabe EH, Bertuol DA, Dotto GL (2015) Biosorption of gold from computer microprocessor leachate solutions using chitin. Waste Manage 45:272–279. https://doi.org/10.1016/j.wasman.2015.07.016
Das D, Vimala R, Das N (2015) Removal of Ag(I) and Zn(II) ions from single and binary solution using sulfonated form of gum arabic-powdered mushroom composite hollow semispheres: Equilibrium, kinetic, thermodynamic and ex-situ studies. Ecol Eng. 75:116–122. https://doi.org/10.1016/j.ecoleng.2014.11.037
Daubert LN, Brennan RA (2007) Passive remediation of acid mine drainage using crab shell chitin Environ. Eng Sci 24:1475–1480. https://doi.org/10.1089/ees.2006.0199
Deng S, Ting YP (2005) Characterization of PEI-modified biomass and biosorption of Cu(II), Pb(II) and Ni(II). Water Res. 39:2167–2177. https://doi.org/10.1016/j.watres.2005.03.033
Donia AM, Atia AA, Elwakeel KZ (2007) Recovery of gold(III) and silver(I) on a chemically modified chitosan with magnetic properties. Hydrometallurgy. 87:197–206. https://doi.org/10.1016/j.hydromet.2007.03.007
Fabian D, Younger PL, Aplin AC (2005) Constructed wetlands for the passive treatment of acid mine drainage allow a quantitative appraisal of the biogeochemical removal of iron, sulphur, and other pollutants. Abstr Pap Am Chem S. 230:U1785–U1786
Fathima A, Aravindhan R, Rao JR, Nair BU (2015) Biomass of Termitomyces clypeatus for chromium (III) removal from chrome tanning wastewater. Clean Technol Environ Policy. 17:541–547
Feng G, Ma J, Zhang X, Zhang Q, Xiao Y, Ma Q, Wang S (2019) Magnetic natural composite Fe3O4-chitosan@bentonite for removal of heavy metals from acid mine drainage. J Colloid Interface Sci. 538:132–141
Ferraz AI, Amorim C, Tavares T, Teixeira JA (2015) Chromium(III) biosorption onto spent grains residual from brewing industry: equilibrium, kinetics and column studies. Int J Environ Sci Technol. 12:1591–1602. https://doi.org/10.1007/s13762-014-0539-6
Fu Y, Viraraghavan T (2001) Fungal decolorization of dye wastewaters: a review. Bioresource Technol 79:251–262. https://doi.org/10.1016/S0960-8524(01)00028-1
Fu Y, Viraraghavan T (2002) Dye biosorption sites in Aspergillus niger. Bioresource Technol 82:139–145. https://doi.org/10.1016/S0960-8524(01)00172-9
Fu FL, Wang Q (2011) Removal of heavy metal ions from wastewaters: A review. J Environ Manage 92:407–418. https://doi.org/10.1016/j.jenvman.2010.11.011
Gaikwad RW, Gupta DV (2008) Review on removal of heavy metals from acid mine drainage. Appl Ecol Environ Res 6:79–96. https://doi.org/10.15666/aeer/0603_081098
Gazea B, Adam K, Kontopoulos A (1996) A review of passive systems for the treatment of acid mine drainage. Miner Eng 9:23–42. https://doi.org/10.1016/0892-6875(95)00129-8
Gong QQ, Guo XY, Liang S, Wang C, Tian QH (2016) Study on the adsorption behavior of modified persimmon powder biosorbent on Pt(IV). Int J Environ Sci Technol. 13:47–54. https://doi.org/10.1007/s13762-015-0809-y
Groudev S, Georgiev P, Spasova I, Nicolova M (2008) Bioremediation of acid mine drainage in a uranium deposit. Hydrometallurgy 94:93–99. https://doi.org/10.1016/j.hydromet.2008.05.023
Gutiérrez C, Hansen HK, Hernández P, Pinilla C (2015) Biosorption of cadmium with brown macroalgae. Chemosphere 138:164–169. https://doi.org/10.1016/j.chemosphere.2015.06.002
Hallam L, Papasergio AE, Lessio M, Veliscek-Carolan J (2021) Phosphate functionalised titania for heavy metal removal from acidic sulfate solutions. J Colloid Interface Sci 600:719–728. https://doi.org/10.1016/j.jcis.2021.05.047
Hedin RS, Watzlaf GR, Nairn RW (1994) Passive treatment of acid-mine drainage with limestone. J Environ Qual 23:1338–1345
Holub M, Balintova M, Pavlikova P (2018) Proposal and testing of multi-step process for acid mine drainage treatment. Inz Miner. https://doi.org/10.29227/Im-2018-02-25
Hong H-J, Yu H, Hong S, Hwang JY, Kim SM, Park MS, Jeong HS (2020) Modified tunicate nanocellulose liquid crystalline fiber as closed loop for recycling platinum-group metals. Carbohyd Polym 228:115424. https://doi.org/10.1016/j.carbpol.2019.115424
Hosseinzadeh H, Mohammadi S (2016) Biosorption of anionic dyes from aqueous solutions using a novel magnetic nanocomposite adsorbent based on rice husk ash. Sep Sci Technol (philadelphia) 51:939–953. https://doi.org/10.1080/01496395.2016.1142564
Hu J, Liu J, Li J, Lv X, Yu L, Wu K, Yang Y (2021) Metal contamination, bioaccumulation, ROS generation, and epigenotoxicity influences on zebrafish exposed to river water polluted by mining activities. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2020.124150
Hurtado C, Viedma P, Cotoras D (2018) Design of a bioprocess for metal and sulfate removal from acid mine drainage. Hydrometallurgy 180:72–77. https://doi.org/10.1016/j.hydromet.2018.07.006
Jeen SW, Mattson B (2016) Evaluation of layered and mixed passive treatment systems for acid mine drainage. Environ Technol 37:2835–2851. https://doi.org/10.1080/09593330.2016.1167249
Johnson DB, Hallberg KB (2005) Acid mine drainage remediation options: a review. Sci Total Environ 338:3–14. https://doi.org/10.1016/j.scitotenv.2004.09.002
Kavehei A, Hose GC, Gore DB (2021) History of environmental contamination at Sunny Corner Ag–Pb–Zn mine, eastern Australia: A meta-analysis approach. Environ Pollution. https://doi.org/10.1016/j.envpol.2020.115742
Khosravihaftkhany S, Morad N, Abdullah AZ, Teng TT, Ismail N (2015) Biosorption of Pb(II) and Fe(III) from aqueous co-solutions using chemically pretreated oil palm fronds. RSC Advances. 5:106498–106508. https://doi.org/10.1039/c5ra15325e
Kim SD, Kilbane Ii JJ, Cha DK (1999) Prevention of acid mine drainage by sulfate reducing bacteria: organic substrate addition to mine waste piles Environ. Eng Sci 16:139–145
Kim N, Park M, Park D (2015a) A new efficient forest biowaste as biosorbent for removal of cationic heavy metals. Bioresource Technol 175:629–632. https://doi.org/10.1016/j.biortech.2014.10.092
Kim MS, Kim JG (2020) Adsorption characteristics of spent coffee grounds as an alternative adsorbent for cadmium in solution. Environments. https://doi.org/10.3390/environments7040024
Kim S, Song M-H, Wei W, Yun Y-S (2015) Selective biosorption behavior of Escherichia coli biomass toward Pd(II) in Pt(IV)–Pd(II) binary solution. J Hazard Mater. 283:657–662. https://doi.org/10.1016/j.jhazmat.2014.10.008
Kleinmann RLP (1990) At-source control of acid mine drainage. Int J Mine Water 9:85–96. https://doi.org/10.1007/BF02503685
Kratochvil D, Volesky B (1998) Advances in the biosorption of heavy metals. Trends Biotechnol 16:291–300. https://doi.org/10.1016/S0167-7799(98)01218-9
Kwak IS, Yun YS (2010) Recovery of zero-valent gold from cyanide solution by a combined method of biosorption and incineration. Bioresource Technol 101:8587–8592. https://doi.org/10.1016/j.biortech.2010.06.080
Laus R, Geremias R, Vasconcelos HL, Laranjeira MCM, Favere VT (2007) Reduction of acidity and removal of metal ions from coal mining effluents using chitosan microspheres. J Hazard Mater 149:471–474. https://doi.org/10.1016/j.jhazmat.2007.04.012
Lewis AE (2010) Review of metal sulphide precipitation. Hydrometallurgy 104:222–234. https://doi.org/10.1016/j.hydromet.2010.06.010
Li Z, Ge Y, Wan L (2015b) Fabrication of a green porous lignin-based sphere for the removal of lead ions from aqueous media. J Hazard Mater 285:77–83. https://doi.org/10.1016/j.jhazmat.2014.11.033
Li WC, Law FY, Chan YHM (2015) Biosorption studies on copper (II) and cadmium (II) using pretreated rice straw and rice husk. Environ Sci Pollut R. https://doi.org/10.1007/s11356-015-5081-7
Lin S, Wei W, Wu X, Zhou T, Mao J, Yun Y-S (2015) Selective recovery of Pd(II) from extremely acidic solution using ion-imprinted chitosan fiber: adsorption performance and mechanisms. J Hazard Mater. 299:10–17. https://doi.org/10.1016/j.jhazmat.2015.05.050
Liu X, Gao C, Liang C, Nie S, Wang S (2016) Copper (II) removed from aqueous solutions by aminated bagasse pith. Jo Residuals Sci Technol. https://doi.org/10.12783/issn.1544-8053/13/3/6
Luptakova A, Kusnierova M (2005) Bioremediation of acid mine drainage contaminated by SRB. Hydrometallurgy 77:97–102. https://doi.org/10.1016/j.hydromet.2004.10.019
Mao J, Lee SY, Won SW, Yun Y-S (2010) Surface modified bacterial biosorbent with poly(allylamine hydrochloride): Development using response surface methodology and use for recovery of hexachloroplatinate(IV) from aqueous solution. Water Res. 44:5919–5928. https://doi.org/10.1016/j.watres.2010.07.034
Massocatto CL et al (2015) Biosorption of Pb2+, Cr3+, and Cu2+ by peach palm sheath modified colonized by Agaricus Blazei. Desalination Water Treatment. https://doi.org/10.1080/19443994.2015.1107503
Masukume M, Onyango MS, Maree JP (2014) Sea shell derived adsorbent and its potential for treating acid mine drainage. Int J Miner Process 133:52–59. https://doi.org/10.1016/j.minpro.2014.09.005
Meneghel AP, Gonçalves AC, Rubio F, Dragunski DC, Lindino CA, Strey L (2013) Biosorption of cadmium from water using moringa (Moringa oleifera Lam.) seeds water. Air Soil Pollution 224:1–13. https://doi.org/10.1007/s11270-012-1383-2
Mirmohseni A, Seyed Dorraji MS, Figoli A, Tasselli F (2012) Chitosan hollow fibers as effective biosorbent toward dye: Preparation and modeling. Bioresource Technol 121:212–220. https://doi.org/10.1016/j.biortech.2012.06.067
Moghaddam MR, Fatemi S, Keshtkar A (2013) Adsorption of lead (Pb2+) and uranium cations by brown algae; experimental and thermodynamic modeling. Chem Eng J. 231:294–303. https://doi.org/10.1016/j.cej.2013.07.037
Mohd Bahari Z, Ali Hamood Altowayti W, Ibrahim Z, Jaafar J, Shahir S (2013) Biosorption of As (III) by non-living biomass of an arsenic-hypertolerant Bacillus cereus strain SZ2 isolated from a gold mining environment: equilibrium and kinetic study. Appl Biochem Biotechnol. 171:2247–2261. https://doi.org/10.1007/s12010-013-0490-x
Monier M, Abdel-Latif DA, Abou El-Reash YG (2016) Ion-imprinted modified chitosan resin for selective removal of Pd(II) ions. J Colloi Interface Sci. 469:344–354. https://doi.org/10.1016/j.jcis.2016.01.074
Moses CO, Nordstrom DK, Herman JS, Mills AL (1987) Aqueous pyrite oxidation by dissolved-oxygen and by ferric iron. Geochim Cosmochim Ac 51:1561–1571. https://doi.org/10.1016/0016-7037(87)90337-1
Moyo M, Chirinda A, Nharingo T (2016) Removal of Copper from Aqueous Solution Using Chemically Treated Potato (Solanum tuberosum) Leaf Powder CLEAN–Soil, Air, Water
Naja G, Volesky B (2011) The mechanism of metal cation and anion biosorption. Microbial Biosorption of Metals. https://doi.org/10.1007/978-94-007-0443-5_3
Nascimento MRL, Fukuma HT, Da Costa WC, Quinelato AL, Garcia O Jr, Gomes HA (2006) Removal of radionuclides from acid mine waters by retention on adsorbing materials. J Radioanal Nucl Chem 269:755–759. https://doi.org/10.1007/s10967-006-0296-1
Nordstrom DK (2000) Advances in the hydrogeochemistry and microbiology of acid mine waters. Int Geol Rev 42:499–515
Noreen S, Bhatti HN, Nausheen S, Sadaf S, Ashfaq M (2013) Batch and fixed bed adsorption study for the removal of Drimarine Black CL-B dye from aqueous solution using a lignocellulosic waste: a cost affective adsorbent. Ind Crops Prod 50:568–579. https://doi.org/10.1016/j.indcrop.2013.07.065
Park D, Yun YS, Park JM (2010) The past present, and future trends of biosorption. Biotechnol Bioproc E 15:86–102. https://doi.org/10.1007/s12257-009-0199-4
Park SI et al (2012) Recovery of gold as a type of porous fiber by using biosorption followed by incineration. Bioresource Technol 104:208–214. https://doi.org/10.1016/j.biortech.2011.11.018
Parshetti GK, Chowdhury S, Balasubramanian R (2014) Hydrothermal conversion of urban food waste to chars for removal of textile dyes from contaminated waters. Bioresource Technol 161:310–319. https://doi.org/10.1016/j.biortech.2014.03.087
Pat-Espadas AM, Field JA, Otero-Gonzalez L, Razo-Flores E, Cervantes FJ, Sierra-Alvarez R (2016) Recovery of palladium(II) by methanogenic granular sludge. Chemosphere. 144:745–75. https://doi.org/10.1016/j.chemosphere.2015.09.035
Pinto PX, Al-Abed SR, Reisman DJ (2011) Biosorption of heavy metals from mining influenced water onto chitin products. Chem Eng J 166:1002–1009. https://doi.org/10.1016/j.cej.2010.11.091
Podstawczyk D, Witek-Krowiak A, Chojnacka K, Sadowski Z (2014) Biosorption of malachite green by eggshells: mechanism identification and process optimization. Bioresource Technol 160:161–165. https://doi.org/10.1016/j.biortech.2014.01.015
Puranik PR, Paknikar KM (1999) Biosorption of lead, cadmium, and zinc by Citrobacter strain MCM B-181: characterization studies. Biotechnol Progress. 15:228–237. https://doi.org/10.1021/bp990002r
Rahman MS, Sathasivam KV (2016) Heavy metal biosorption potential of a Malaysian Rhodophyte (Eucheuma denticulatum) from aqueous solutions. Int J Environ Sci Technol. 13:1973–1988. https://doi.org/10.1007/s13762-016-1022-3
Ramakul P, Yanachawakul Y, Leepipatpiboon N, Sunsandee N (2012) Biosorption of palladium(II) and platinum(IV) from aqueous solution using tannin from Indian almond (Terminalia catappa L.) leaf biomass: Kinetic and equilibrium studies. Chem Eng J. 193–194:102–111. https://doi.org/10.1016/j.cej.2012.04.035
Robinson-Lora MA, Brennan RA (2009) Efficient metal removal and neutralization of acid mine drainage by crab-shell chitin under batch and continuous-flow conditions. Biores Technol 100:5063–5071. https://doi.org/10.1016/j.biortech.2008.11.063
Rubio F, Gonçalves AC Jr, Meneghel AP, Teixeira Tarley CR, Schwantes D, Coelho GF (2013) Removal of cadmium from water using by-product Crambe abyssinica Hochst seeds as biosorbent material. Water Sci Technol 68:227–233. https://doi.org/10.2166/wst.2013.233
Salomons W (1995) Environmental-impact of metals derived from mining activities - processes predictions, prevention. J Geochem Explor 52:5–23. https://doi.org/10.1016/0375-6742(94)00039-E
Santos AL, Johnson DB (2017) The effects of temperature and pH on the kinetics of an acidophilic sulfidogenic bioreactor and indigenous microbial communities. Hydrometallurgy 168:116–120
Santos S, Machado R, Correia MJN, Carvalho JR (2004) Treatment of acid mining waters. Minerals Eng 17:225–232. https://doi.org/10.1016/j.mineng.2003.09.015
Sarode S, Upadhyay P, Khosa MA, Mak T, Shakir A, Song S, Ullah A (2019) Overview of wastewater treatment methods with special focus on biopolymer chitin-chitosan. Int J Biol Macromol 121:1086–1100
Schippers A, Breuker A, Blazejak A, Bosecker K, Kock D, Wright TL (2010) The biogeochemistry and microbiology of sulfidic mine waste and bioleaching dumps and heaps, and novel Fe(II)-oxidizing bacteria. Hydrometallurgy. 104:342–350. https://doi.org/10.1016/j.hydromet.2010.01.012
Seo JH, Kim N, Park M, Lee S, Yeon S, Park D (2020) Evaluation of metal removal performance of rod-type biosorbent prepared from sewage-sludge. Environ Eng Res 25:700–706. https://doi.org/10.4491/eer.2019.201
Sheoran AS, Sheoran V (2006) Heavy metal removal mechanism of acid mine drainage in wetlands: a critical review. Miner Eng 19:105–116. https://doi.org/10.1016/j.mineng.2005.08.006
Simate GS, Ndlovu S (2014) Acid mine drainage: challenges and opportunities Journal of Environmental. Chem Eng 2:1785–1803. https://doi.org/10.1016/j.jece.2014.07.021
Song D, Park SJ, Kang HW, Park SB, Han JI (2013) Recovery of lithium(I), strontium(II), and lanthanum(III) using Ca-alginate beads. J Chem Eng Data. 58:2455–2464. https://doi.org/10.1021/je400317v
Stoltz E, Greger M (2006) Influences of wetland plants on weathered acidic mine tailings. Environ Pollut 144:689–694. https://doi.org/10.1016/j.envpol.2005.12.038
Su W, Yang Y, Dai H, Jiang L (2015) Biosorption of heavy metal ions from aqueous solution on chinese fir bark modified by sodium hypochlorite. BioResources 10:6993–7008
Sulaymon AH, Mohammed AA, Al-Musawi TJ (2014) Comparative study of removal of cadmium (II) and chromium (III) ions from aqueous solution using low-cost biosorbent. Int J Chem Reactor Eng. 12:477–486
Sun L, Chen D, Wan S, Yu Z (2015) Performance, kinetics, and equilibrium of methylene blue adsorption on biochar derived from eucalyptus saw dust modified with citric, tartaric, and acetic acids. Bioresource Technol. 198:300–308. https://doi.org/10.1016/j.biortech.2015.09.026
Tan WS, Ting ASY (2014) Alginate-immobilized bentonite clay: Adsorption efficacy and reusability for Cu(II) removal from aqueous solution. Bioresource Technol. 160:115–118. https://doi.org/10.1016/j.biortech.2013.12.056
Utgikar V, Chen BY, Tabak HH, Bishop DF, Govind R (2000) Treatment of acid mine drainage: I equilibrium biosorption of zinc and copper on non-viable activated sludge. Int Biodeter Biodegr 46:19–28. https://doi.org/10.1016/S0964-8305(00)00053-6
Varia J, Zegeye A, Roy S, Yahaya S, Bull S (2014) Shewanella putrefaciens for the remediation of Au3+, Co2+ and Fe3+ metal ions from aqueous systems. Biochem Eng J. 85:101–109. https://doi.org/10.1016/j.bej.2014.02.002
Vijayaraghavan K, Won SW, Mao J, Yun YS (2008) Chemical modification of corynebacterium glutamicum to improve methylene blue biosorption. Chem Eng J 145:1–6. https://doi.org/10.1016/j.cej.2008.02.011
Vijayaraghavan K, Joshi UM (2014) Application of Ulva sp. biomass for single and binary biosorption of chromium (III) and manganese (II) ions: equilibrium modeling. Environ Progress Sustainable Energy. 33:147–153
Volesky B (2007) Biosorption and me. Water Res 41:4017–4029. https://doi.org/10.1016/j.watres.2007.05.062
Wang T, Sun H (2013) Biosorption of heavy metals from aqueous solution by UV-mutant bacillus subtilis. Environ Sci Pollut R 20:7450–7463. https://doi.org/10.1007/s11356-013-1767-x
Wang M-X, Zhang Q-L, Yao S-J (2015) A novel biosorbent formed of marine-derived penicillium janthinellum mycelial pellets for removing dyes from dye-containing wastewater. Chem Eng J 259:837–844. https://doi.org/10.1016/j.cej.2014.08.003
Won SW, Mao J, Kwak I-S, Sathishkumar M, Yun Y-S (2010) Platinum recovery from ICP wastewater by a combined method of biosorption and incineration. Bioresource Technol 101:1135–1140. https://doi.org/10.1016/j.biortech.2009.09.056
Yang J, Volesky B (1999) Removal and concentration of uranium by seaweed biosorbent. doi:https://doi.org/10.1016/S1572-4409(99)80137-0
Yang JS, Kim YS, Park SM, Baek K (2014) Removal of As(III) and As(V) using iron-rich sludge produced from coal mine drainage treatment plant. Environ Sci Pollution Res. 21:10878–10889. https://doi.org/10.1007/s11356-014-3023-4
Zagury GJ, Kulnieks VI, Neculita CM (2006) Characterization and reactivity assessment of organic substrates for sulphate-reducing bacteria in acid mine drainage treatment. Chemosphere 64:944–954. https://doi.org/10.1016/j.chemosphere.2006.01.001
Zhang M, Helleur R, Zhang Y (2015) Ion-imprinted chitosan gel beads for selective adsorption of Ag+ from aqueous solutions. Carbohydrate Polym. 130:206–212. https://doi.org/10.1016/j.carbpol.2015.05.038
Zhao Y, Wang D, Xie H, Won SW, Cui L, Wu G (2015) Adsorption of Ag (I) from aqueous solution by waste yeast: kinetic, equilibrium and mechanism studies. Bioprocess Biosystems Eng. 38:69–77. https://doi.org/10.1007/s00449-014-1244-z
Zhao S, Zhou T (2016) Biosorption of methylene blue from wastewater by an extraction residue of Salvia miltiorrhiza Bge. Bioresource Technol 219:330–337. https://doi.org/10.1016/j.biortech.2016.07.121
Acknowledgements
This research was supported by Basic Science Research Program through the National Research Foundation of Korea(NRF) funded by the Ministry of Education (2019R1A6A3A01096685).
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial responsibility: Maryam Shabani.
Rights and permissions
About this article
Cite this article
Kim, N., Park, D. Biosorptive treatment of acid mine drainage: a review. Int. J. Environ. Sci. Technol. 19, 9115–9128 (2022). https://doi.org/10.1007/s13762-021-03631-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-021-03631-5