Abstract
Purpose
To investigate the association of omentin-1 and inflammatory factors in serum and visceral adipose tissue (VAT) of women with gestational diabetes mellitus (GDM) compared to normal pregnant (NP) subjects. Furthermore, to examine their correlation with maternal clinical characteristics.
Methods
We compared 116 GDM women to 115 NP women, at the time of cesarean section. Circulating omentin-1 and pro-inflammatory (IL-1β, IL-6, TNF-α), and anti-inflammatory cytokines (IL-1RA, IL-10) were examined. Moreover, their mRNA expression in VAT, along with inflammatory factors involved in the NF-κB pathway (TLR2, TLR4, NF-κB, IKκB), were examined.
Results
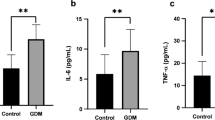
Circulating omentin-1 (p = 0.022) was lower and circulating IL-1-β, IL-1RA, as well as IL-10 (p = 0.005, p = 0.007, and p = 0.015, respectively), were higher in GDM compared to NP women. Omentin-1 correlated negatively with pre-pregnancy and gestational BMI, and HOMA-IR in all women, but was not associated with cytokines. TLR2, TLR4, IL-1β, IL-1RA, IL-6, IL-10 mRNA expression in VAT was lower in GDM compared with controls (p < 0.05 all). In multivariate analysis, BMI at delivery was significantly correlated to omentin-1 concentrations in all and NP subjects. In addition, omentin-1 expression was correlated to inflammatory gene expression in all, GDM and NP, women (p < 0.05 all).
Conclusion
Serum levels and VAT gene expression of omentin-1 are not independently linked to GDM; notwithstanding, GDM women have a VAT-altered inflammatory status. In addition, no systemic association between omentin-1 and inflammatory factors was found, whereas associations between their expression in all women were observed, indicating that expression of these adipokines is linked between them regardless of GDM.


Similar content being viewed by others
Availability of data and material
The data used to support the findings of this study are available from the corresponding author by request.
Code availability
Not applicable for that section.
References
McIntyre HD, Catalano P, Zhang C, Desoye G, Mathiesen ER, Damm P (2019) Gestational diabetes mellitus. Nat Rev Dis Primers 5:47. https://doi.org/10.1038/s41572-019-0098-8
Fasshauer M, Blüher M, Stumvoll M (2014) Adipokines in gestational diabetes. Lancet Diabetes Endocrinol 2:488–499. https://doi.org/10.1016/S2213-8587(13)70176-1
Yang RZ, Lee MJ, Hu H, Pray J, Wu HB, Hansen BC, Shuldiner AR, Fried SK, McLenithan JC, Gong DW (2006) Identification of omentin as a novel depot-specific adipokine in human adipose tissue: possible role in modulating insulin action. Am J Physiol Endocrinol Metab 290:E1253-1261. https://doi.org/10.1152/ajpendo.00572.2004
Watanabe T, Watanabe KK, Takahashi Y, Kojima M, Watanabe R (2017) Adipose tissue-derived omentin-1 function and regulation. Compr Physiol 7:765–781. https://doi.org/10.1002/cphy.c160043
de Souza Batista CM, Yang RZ, Lee MJ, Glynn NM, Yu DZ, Pray J, Ndubuizu K, Patil S, Schwartz A, Kligman M, Fried SK, Gong DW, Shuldiner AR, Pollin TI, McLenithan JC (2007) Omentin plasma levels and gene expression are decreased in obesity. Diabetes 56:1655–1661. https://doi.org/10.2337/db06-1506
Yamawaki H, Kuramoto J, Kameshima S, Usui T, Okada M, Hara Y (2011) Omentin, a novel adipocytokine inhibits TNF-induced vascular inflammation in human endothelial cells. Biochem Biophys Res Commun 408:339–343. https://doi.org/10.1016/j.bbrc.2011.04.039
Kazama K, Usui T, Okada M, Hara Y, Yamawaki H (2012) Omentin plays an anti-inflammatory role through inhibition of TNF-α-induced superoxide production in vascular smooth muscle cells. Eur J Pharmacol 686:116–123. https://doi.org/10.1016/j.ejphar.2012.04.033
Barker G, Lim R, Georgiou HM, Lappas M (2012) Omentin-1 is decreased in maternal plasma, placenta and adipose tissue of women with pre-existing obesity. PLoS One 7:e42943. https://doi.org/10.1371/journal.pone.0042943
Tsiotra PC, Halvatsiotis P, Patsouras K, Maratou E, Salamalekis G, Raptis SA, Dimitriadis G, Boutati E (2018) Circulating adipokines and mRNA expression in adipose tissue and the placenta in women with gestational diabetes mellitus. Peptides 101:157–166. https://doi.org/10.1016/j.peptides.2018.01.005
Pan BL, Ma RM (2016) Correlation of serum omentin-1 and chemerin with gestational diabetes mellitus. Nan Fang Yi Ke Da Xue Xue Bao 36:1231–1236
Souvannavong-Vilivong X, Sitticharoon Ch, Klinjampa R, Keadkraichaiwat I, Sripong Ch, Chatree S, Sririwichitchai R, Lertbunnaphong T (2019) Placental expressions and serum levels of adiponectin, visfatin, and omentin in GDM. Acta Diabetol 56:1121–1131. https://doi.org/10.1007/s00592-019-01355-0
Mierzynski R, Dłuski D, Nowakowski L, Poniedziałek-Czajkowska E, Leszczyńska-Gorzelak B (2018) Adiponectin and omentin levels as predictive biomarkers of preterm birth in patients with gestational diabetes mellitus. Biomed Res Int 2018:7154216. https://doi.org/10.1155/2018/7154216
Abell SK, Shorakae S, Harrison CL, Hiam D, Moreno A, Stepto NK, De Courten B, Teede HJ (2017) The association between dysregulated adipocytokines in early pregnancy and development of gestational diabetes. Diabetes Metab Res Rev 33:e2926. https://doi.org/10.1002/dmrr.2926
Lewandowski K, Nadel I, Lewinski A, Bienkiewicz M, Tan B, Randeva HS, Cypryk K (2010) Positive correlation between serum omentin and thrombospondin-1 in gestational diabetes despite lack of correlation with insulin resistance indices. Ginekol Pol 81:907–912
Franz M, Polterauer M, Springer S, Kuessel L, Haslinger P, Worda C, Worda K (2018) Maternal and neonatal omentin-1 levels in gestational diabetes. Arch Gynecol Obstet 297:885–889. https://doi.org/10.1007/s00404-018-4652-5
Lappas M (2014) Activation of inflammasomes in adipose tissue of women with gestational diabetes. Mol Cell Endocrinol 382:74–83. https://doi.org/10.1016/j.mce.2013.09.011
Hotamisligil GS, Peraldi P, Budavari A, Ellis R, White MF, Spiegelman BM (1996) IRS-1- mediated inhibition of insulin receptor tyrosine kinase activity in TNF-alpha- and obesity-induced insulin resistance. Science 271:665–668. https://doi.org/10.1126/science.271.5249.665
Ballak DB, Stienstra R, Tack CJ, Dinarello CA, van Diepen JA (2015) IL-1 family members in the pathogenesis and treatment of metabolic disease: Focus on adipose tissue inflammation and insulin resistance. Cytokine 75:280–290. https://doi.org/10.1016/j.cyto.2015.05.005
Kirwan JP, Hauguel-De Mouzon S, Lepercq J, Challier JC, Huston PL, Friedman JE, Kalhan SC, Catalano PM (2002) TNF-alpha is a predictor of insulin resistance in human pregnancy. Diabetes 51:2207–2213. https://doi.org/10.2337/diabetes.51.7.2207
Atègbo JM, Grissa O, Yessoufou A, Hichami A, Dramane KL, Moutairou K, Miled A, Grissa A, Jerbi M, Tabka Z, Khan NA (2006) Modulation of adipokines and cytokines in gestational diabetes and macrosomia. J Clin Endocrinol Metab 91:4137–4143. https://doi.org/10.1210/jc.2006-0980
Saucedo R, Valencia J, Moreno-González LE, Peña-Cano MI, Aranda-Martínez A, García Y, Díaz-Velázquez MF, Hernández-Valencia M (2021) Maternal serum adipokines and inflammatory markers at late gestation and newborn weight in mothers with and without gestational diabetes mellitus. Ginekol Pol. https://doi.org/10.5603/GP.a2021.0083
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC (1985) Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28:412–419. https://doi.org/10.1007/BF00280883
Sitticharoon C, Nway NC, Chatree S, Churintaraphan M, Boonpuan P, Maikaew P (2014) Interactions between adiponectin, visfatin, and omentin in subcutaneous and visceral adipose tissues and serum, and correlations with clinical and peripheral metabolic factors. Peptides 62:164–175. https://doi.org/10.1016/j.peptides.2014.10.006
Auguet T, Quintero Y, Riesco D, Morancho B, Terra X, Crescenti A, Broch M, Aguilar C, Olona M, Porras JA, Hernandez M, Sabench F, del Castillo D, Richart C (2011) New adipokines vaspin and omentin. Circulating levels and gene expression in adipose tissue from morbidly obese women. BMC Med Genet 12:60. https://doi.org/10.1186/1471-2350-12-60
Tan BK, Adya R, Farhatullah S, Lewandowski KC, O’Hare P, Lehnert H, Randeva HS (2008) Omentin-1, a novel adipokine, is decreased in overweight insulin-resistant women with polycystic ovary syndrome: ex vivo and in vivo regulation of omentin-1 by insulin and glucose. Diabetes 57:801–808. https://doi.org/10.2337/db07-0990
Wang J, Gao Y, Lin F, Han K, Wang X (2020) Omentin-1 attenuates lipopolysaccharide (LPS)-induced U937 macrophages activation by inhibiting the TLR4/MyD88/NF-κB signaling. Arch Biochem Biophys 679:108187. https://doi.org/10.1016/j.abb.2019.108187
Zhou H, Zhang Z, Qian G, Zhou J (2020) Omentin-1 attenuates adipose tissue inflammation via restoration of TXNIP/NLRP3 signaling in high-fat diet-induced obese mice. Fundam Clin Pharmacol 34:721–735. https://doi.org/10.1111/fcp.12575
Lappas M, Mitton A, Permezel M (2010) In response to oxidative stress, the expression of inflammatory cytokines and antioxidant enzymes are impaired in placenta, but not adipose tissue, of women with gestational diabetes. J Endocrinol 204:75–84. https://doi.org/10.1677/JOE-09-0321
Rojas RR, Lifshitz LM, Bellve KD, Min SY, Pires J, Leung K, Boeras C, Sert A, Draper JT, Corvera S, Moore ST (2015) Human adipose tissue expansion in pregnancy is impaired in gestational diabetes mellitus. Diabetologia 58:2106–2114. https://doi.org/10.1007/s00125-015-3662-0
Kuzmicki M, Telejko B, Zonenberg A, Szamatowicz J, Kretowski A, Nikolajuk A, Laudanski P, Gorska M (2008) Circulating pro- and anti-inflammatory cytokines in Polish women with gestational diabetes. Horm Metab Res 40:556–560. https://doi.org/10.1055/s-2008-1073166
Kanigur Sultuybek G, Soydas T, Yenmis G (2019) NF-κB as the mediator of metformin’s effect on ageing and ageing-related diseases. Clin Exp Pharmacol Physiol 46:413–422. https://doi.org/10.1111/1440-1681.13073
He L, He M, Lv X, Pu D, Su P, Liu Z (2010) NF-κB binding activity and pro-inflammatory cytokines expression correlate with body mass index but not glycosylated hemoglobin in Chinese population. Diabetes Res Clin Pract 90:73–80. https://doi.org/10.1016/j.diabres.2010.06.016
Dasu MR, Devaraj S, Zhao L, Hwang DH, Jialal I (2008) High glucose induces toll-like receptor expression in human monocytes: mechanism of activation. Diabetes 57:3090–3098. https://doi.org/10.2337/db08-0564
Feng H, Su R, Song Y, Wang C, Lin L, Ma J, Yang H (2016) Positive correlation between enhanced expression of TLR4/MyD88/NF-κB with insulin resistance in placentae of gestational diabetes mellitus. PLoS One 11:e0157185. https://doi.org/10.1371/journal.pone.0157185
Kuzmicki M, Telejko B, Wawrusiewicz-Kurylonek N, Lipinska D, Pliszka J, Wilk J, Zielinska A, Skibicka J, Szamatowicz J, Kretowski A, Gorska M (2013) The expression of genes involved in NF-κB activation in peripheral blood mononuclear cells of patients with gestational diabetes. Eur J Endocrinol 168:419–427. https://doi.org/10.1530/EJE-12-0654
Ghanim H, Abuaysheh S, Sia CL, Korzeniewski K, Chaudhuri A, Fernandez-Real JM, Dandona P (2009) Increase in plasma endotoxin concentrations and the expression of toll-like receptors and suppressor of cytokine signaling-3 in mononuclear cells after a high-fat, high-carbohydrate meal implications for insulin resistance. Diabetes Care 32:2281–2287. https://doi.org/10.2337/dc09-0979
Funding
This study was supported by scientific grants from Instituto Mexicano del Seguro Social (FIS/IMSS/PROT/G18/1826). RS, JVO, EMA, and RGD hold a fellowship from the National System of Investigators (Consejo Nacional de Ciencia y Tecnología).
Author information
Authors and Affiliations
Contributions
MIPC, JVO, and RS planned and designed the study. MIPC, and JVO performed laboratory biomarker analysis. EMA, MFDV, RGD, and RS drafted the manuscript. MIPC, and RS performed the data analysis. All authors provided significant intellectual contribution in interpreting data and critical review of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethics approval
The study protocol was approved by the Institutional Review Board of the Instituto Mexicano del Seguro Social (IMSS) in Mexico City (R-2018-785-026).
Consent to participate
All participants gave written informed consent.
Consent for publication
All authors approved the final version of the manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Peña-Cano, M.I., Valencia-Ortega, J., Morales-Ávila, E. et al. Omentin-1 and its relationship with inflammatory factors in maternal plasma and visceral adipose tissue of women with gestational diabetes mellitus. J Endocrinol Invest 45, 453–462 (2022). https://doi.org/10.1007/s40618-021-01671-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-021-01671-9