Abstract
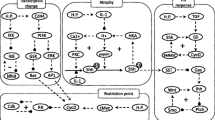
Infection with Helicobacter pylori (H. pylori) leads to a fork in the road situation where it is critical and complex to judge the fate of the cell. We propose for the first time an in silico representation of a protein level network model that can unfold the mystery behind the cell fate decision between inflammation or cell proliferation or cell death. Upon infection TNF inducible protein α (Tip α) is internalised after binding with the cell surface receptor Nucleolin which is overexpressed on the cell surface thereby activating the Ras pathway. Tip α, Nucleolin and Ras decides the cell fate for apoptosis or abnormal cell proliferation along with ulcers in the gastric tract, hence we term it as the “death triad”, which otherwise triggers the inflammatory pathway through downstream signalling of NF-κβ. A series of proteins involved in the signalling cascade are portrayed through compartmentalization of the bacteria and the gut wall. The depicted network works synchronously toward an overarching goal of deciding between apoptosis or inflammation or proliferation. The model has been validated by simulating it with existing transcriptomic data along with clinical findings from patients infected with H. pylori across different regions in India. The results clearly indicate that for a short period of time there is increased binding of Tip α to Nucleolin and the receptor starts to saturate. This increases the tenacity of binding and the cell triggers an inflammatory cascade reaction which involves proinflammatory cytokines such as TNF α thereby progressing to inflammation by activating NF-κβ downstream. On the other hand, Ras involved in interaction with nucleolin can be present both in its activated or inactivated state. Binding of Tip α as a monomer leads to desensitization of Nucleolin leading to cell survival and proliferation.




Similar content being viewed by others
Data Availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Morgner A et al (2001) Helicobacter pylori associated gastric B cell MALT lymphoma: predictive factors for regression. Gut 48(3):290–292
Bakri MM (2013) Prevalence of Helicobacter pylori infection and the incidence of ureA and clarithromycin resistance gene 23S rRNA genotypes status in Saudi Arabia. Saudi J Biol Sci 20(1):75–78
Ferreira AC et al (2008) Helicobacter and gastric malignancies. Helicobacter 13:28–34
Atherton JC et al (1997) Clinical and pathological importance of heterogeneity in vacA, the vacuolating cytotoxin gene of Helicobacter pylori. Gastroenterology 112(1):92–99
Yamaoka Y et al (1999) Relationship between Helicobacter pylori iceA, cagA, and vacA status and clinical outcome: studies in four different countries. J Clin Microbiol 37(7):2274–2279
Suganuma M et al (2008) TNF-α-inducing protein, a carcinogenic factor secreted from H. pylori, enters gastric cancer cells. Int J Cancer 123(1):117–122
Tsuge H et al (2009) Structural basis for the Helicobacter pylori-carcinogenic TNF-α-inducing protein. Biochem Biophys Res Commun 388(2):193–198
Godlewska R et al (2008) Tip-α (hp0596 gene product) is a highly immunogenic Helicobacter pylori protein involved in colonization of mouse gastric mucosa. Curr Microbiol 56(3):279–286
Polk DB, Peek RM (2010) Helicobacter pylori: gastric cancer and beyond. Nat Rev Cancer 10(6):403–414
Censini S et al (1996) Cag, a pathogenicity island of Helicobacter pylori, encodes type I-specific and disease-associated virulence factors. Proc Natl Acad Sci 93(25):14648–14653
Sayed IM et al (2020) Helicobacter pylori infection downregulates the DNA glycosylase NEIL2, resulting in increased genome damage and inflammation in gastric epithelial cells. J Biol Chem 295(32):11082–11098
Derakhshan MH et al (2006) Gastric histology, serological markers and age as predictors of gastric acid secretion in patients infected with Helicobacter pylori. J Clin Pathol 59(12):1293–1299
Ugrinova I et al (2018) Chapter four—multifaceted nucleolin protein and its molecular partners in oncogenesis. In: Donev R (ed) Advances in protein chemistry and structural biology. Academic Press, Cambridge, pp 133–164
Masiuk M (2008) Nucleolin—characteristics of protein and its role in biology of cancers and viral infections. Adv Cell Biol 1:1–19
Srivastava M, Pollard HB (1999) Molecular dissection of nucleolin’s role in growth and cell proliferation: new insights. FASEB J 13(14):1911–1922
Reyes-Reyes EM, Akiyama SK (2008) Cell-surface nucleolin is a signal transducing P-selectin binding protein for human colon carcinoma cells. Exp Cell Res 314(1112):2212–2223
Watanabe T et al (2010) Nucleolin on the cell surface as a new molecular target for gastric cancer treatment. Biol Pharm Bull 33(5):796–803
Schokoroy S et al (2013) Disrupting the oncogenic synergism between nucleolin and ras results in cell growth inhibition and cell death. PLoS ONE 8(9):e75269
Novere NL et al (2005) Minimum information requested in the annotation of biochemical models (MIRIAM). Nat Biotech 23(12):1509–1515
Hucka M et al (2003) The systems biology markup language (SBML): a medium for representation and exchange of biochemical network models. Bioinformatics 19(4):524–531
Farin K et al (2011) Oncogenic synergism between ErbB1, nucleolin, and mutant ras. Can Res 71(6):2140–2151
Merrell DS et al (2003) pH-regulated gene expression of the gastric pathogen Helicobacter pylori. Infect Immun 71(6):3529–3539
Girvan AC et al (2006) AGRO100 inhibits activation of nuclear factor-κB (NF-κB) by forming a complex with NF-κB essential modulator (NEMO) and nucleolin. Mol Cancer Ther 5(7):1790–1799
Mukherjee S, Zhang Y (2011) Protein–protein complex structure predictions by multimeric threading and template recombination. Structure 19(7):955–966
Tamura K et al (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24(8):1596–1599
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucl Acids Res 29:e45
Fernández-Medarde A, Santos E (2011) Ras in cancer and developmental diseases. Genes Cancer 2(3):344–358
Papamichael K, Konstantopoulos P, Mantzaris GJ (2014) Helicobacter pylori infection and inflammatory bowel disease: is there a link? World J Gastroenterol WJG 20(21):6374–6385
Capitani N, Codolo G, Vallese F, Minervini G, Grassi A, Cianchi F, Troilo A, Fischer W, Zanotti G, Baldari CT, de Bernard M, D’Elios MM (2019) The lipoprotein HP1454 of Helicobacter pylori regulates T-cell response by shaping T-cell receptor signalling. Cell Microbiol 21(5):e13006
Tajrishi MM, Tuteja R, Tuteja N (2011) Nucleolin. Commun Integr Biol 4(3):267–275
Takahashi G et al (1993) Effect of granulocyte-macrophage colony-stimulating factor and interleukin-3 on interleukin-8 production by human neutrophils and monocytes. Blood 81(2):357–364
Hovanessian AG et al (2010) Surface expressed nucleolin is constantly induced in tumor cells to mediate calcium-dependent ligand internalization. PLoS ONE 5(12):e15787
Lamb A et al (2009) Helicobacter pylori CagA activates NF-κB by targeting TAK1 for TRAF6-mediated Lys 63 ubiquitination. EMBO Rep 10(11):1242–1249
Acknowledgements
We would like to acknowledge Dr Ashok K. Chauhan, Founder President, Amity University, for providing the infrastructure and support to carry out our research work. We also thank, Mr Pranav Patni of Amity Institute of Biotechnology for his commendable help.
Funding
This study has been funded by ICMR (5/13/16/2015/NCD-III). Dr. Amlan Chakraborty is a postdoctoral research fellow and is funded through an NHMRC grant.
Author information
Authors and Affiliations
Contributions
RD and SB have given the concept of association between Tip α and Nucleolin. SM & AC have performed the experiments, analysed the data and drafted the manuscript. RD and VG have revised the manuscript. AS has done the part of the docking analysis. SM has done the part of the expression analysis. AKM has provided the samples for expression profiling. KD has conceptualised the idea clinically and corrected the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical Approval
This study was approved by institutional ethical committee of Amity Institute of Biotechnology, Noida, U.P, dated 2-12-2015 and National Institute of Cholera and Enteric Diseases, Kolkata (A-1/2016-IEC, dated 22-12-2016).
Informed Consent
Written informed consent was taken.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mahant, S., Chakraborty, A., Som, A. et al. The Synergistic Role of Tip α, Nucleolin and Ras in Helicobacter pylori Infection Regulates the Cell Fate Towards Inflammation or Apoptosis. Curr Microbiol 78, 3720–3732 (2021). https://doi.org/10.1007/s00284-021-02626-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-021-02626-2