Abstract
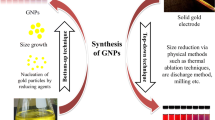
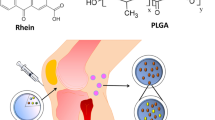
Rheumatoid arthritis (RA) could be a common autoimmune disease that involves severe joint deformation. The main off-target medicines unable to cure RA, that permits associate in nursing infection with the unwellness. The nanomaterials-based RA medical aid is an excellent strategy to enhance the treatment efficaciousness within the inflammatory region. Specifically, metal-based nanomaterials are a wonderful choice for a delivery vehicle for inflammatory disease agents, because of their novel properties. We have got developed a small-sized Polyethylenimine (PEI) coated gold nanoparticles (AuNPs) with an average diameter of 80 nm, that area unit used for top loading of carboxylic acid (MA). Various microscopic and qualitative analysis tools like high-resolution transmission microscopy (HR-TEM), Field-emission scanning microscope (FE-SEM), and Fourier-transform infrared (FTIR) spectroscopic analysis studies were accustomed to make sure as-made PEI-AuNPs. The invented PEI-coated AuNPs (PEI-AuNPs) exhibited higher contrast with an extended expanse that's promising to store giant amounts of MA medicine. MA is attributed to reducing the inflammatory response by inhibiting the pro-inflammatory cytokines and averting undesirable ancient drug aspect effects in Collagen-induced inflammatory disease (CIA). Many organic chemistry parameters like weight, hind paw volume, protein estimation, anti-serum protein analysis, and microscopic anatomy examination were conducted in Collagen-induced arthritis mice treated at a dose (10 µg) of MA packed PEI-AuNPs. The obtained results showed the MA-PEI-AuNPs were used with success within the treatment of Collagen-induced arthritis, relative to PEI-AuNPs and MA. Therefore, MA loaded PEI-AuNPs as a stimulating candidate in future RA applications.









Similar content being viewed by others
References
Q. Cui and Y. Wang (2021). J. Clust. Sci. 2021, 1–8.
M. A. Hussain, I. Shad, I. Malik, F. Amjad, T. Kausar, M. Sher, M. N. Tahir, N. Ullah, M. Ashraf, and S. N. A. Bukhari (2020). Saudi Pharm. J. 28, 869–875.
J. Pulit-Prociak, A. Staroń, O. Długosz, K. Kluz, and M. Banach (2020). J. Clust. Sci. 2020, 1–12.
S. F. El-Amin, M. S. Kwon, T. Starnes, H. R. Allcock, and C. T. Laurencin (2006). J. Inorg. Organomet. Polym. Mater. 16, 387–396.
L. D. Quan, G. M. Thiele, J. Tian, and D. Wang (2008). Expert Opin. Ther. Pat. 18, 723–738.
P. Li, Y. Zheng, and X. Chen (2017). Front. Pharmacol. 8, 460.
T. D. Wilsdon and C. L. Hill (2017). Aust. Prescr. 40 (5), 1.
A. N. Panche, A. D. Diwan, and S. R. Chandra (2016). J. Nutr. Sci. 5, e47.
A. M. Abdel-Azeem, S. M. Zaki, W. F. Khalil, N. A. Makhlouf, and L. M. Farghaly (2016). Front. Microbiol. 7, 1477.
M. H. Pan, C. S. Lai, and C. T. Ho (2010). Food Funct. 1, 15–31.
T. C. Wallace and M. M. Giusti (2015). Adv. Nutr. 6, 620–622.
H. E. Khoo, A. Azlan, S. T. Tang, and S. M. Lim (2017). Food Nutr. Res. 61, 1361779.
M. H. Jeong, H. Ko, H. Jeon, G. J. Sung, S. Y. Park, W. J. Jun, Y. H. Lee, J. Lee, S. W. Lee, H. G. Yoon, and K. C. Choi (2016). Oncotarget 7, 56767.
C. T. Pham (2011). Wiley Interdiscip. Rev. 3, 607–619.
A. Mani, C. Vasanthi, V. Gopal, and D. Chellathai (2016). Int. Immunopharmacol. 41, 17–23.
H. Nah, D. Lee, M. Heo, J. S. Lee, S. J. Lee, D. N. Heo, J. Seong, H. N. Lim, Y. H. Lee, H. J. Moon, and Y. S. Hwang (2019). Sci. Technol. Adv. Mater. 20, 826–836.
S. Taranejoo, J. Liu, P. Verma, and K. Hourigan (2015). J. Appl. Polym. Sci. 2015, 132.
C. Hu, Q. Peng, F. Chen, Z. Zhong, and R. Zhuo (2010). Bioconj. Chem. 21, 836–843.
Q. Xue and Y. Wang (2021). J. Clust. Sci. 2021, 1–7.
M. M. Ansari, A. Ahmad, R. K. Mishra, S. S. Raza, and R. Khan (2019). ACS Biomater. Sci. Eng. 5, 3380–3397.
C. Y. Tsai, A. L. Shiau, S. Y. Chen, Y. H. Chen, P. C. Cheng, M. Y. Chang, D. H. Chen, C. H. Chou, C. R. Wang, and C. L. Wu (2007). Arthr. Rheum. 56, 544–554.
R. Singh and J. W. Lillard Jr. (2009). Exp. Mol. Pathol. 86, 215–223.
H. Singh, J. Du, P. Singh, and T. H. Yi (2018). Artif. Cells Nanomed. Biotechnol. 46, 1163–1170.
D. Jain and R. Banerjee (2008). J. Biomed. Mater. Res. Part B 86, 105–112.
A. S. AbdRaboh, M. S. El-khooly, and M. Y. Hassaan (2021). J. Inorg. Organomet. Polym. Mater. 2021, 1–12.
P. C. Pandey, G. Pandey, and R. J. Narayan (2017). Biointerphases. 12, 011005.
S. Tummala, M. S. Kumar, and S. K. Pindiprolu (2016). Drug Deliv. 23, 3505–3519.
Q. Yuan, Y. Zhao, P. Cai, Z. He, F. Gao, J. Zhang, and X. Gao (2019). ACS Omega 4, 14092–14099.
S. Ueha, F. H. Shand, and K. Matsushima (2012). Front. Immunol. 3, 71.
R. Domingo-Gonzalez, O. Prince, A. Cooper, and S. A. Khader (2017). Tuberc. Tuber. Bacillus. 2017, 33–72.
C. Yang, Z. Daoping, X. Xiaoping, L. Jing, and Z. Chenglong (2019). J. Microencapsul. 37, 77–90.
M. Komiyama, T. Mori, and K. Ariga (2018). Bull. Chem. Soc. Jpn. 91, 1075–1111.
H. Lee, M. Y. Lee, S. H. Bhang, B. S. Kim, Y. S. Kim, J. H. Ju, K. S. Kim, and S. K. Hahn (2014). ACS Nano. 8, 4790–4798.
S. D. Vita, F. Zaja, S. Sacco, A. D. Candia, R. Fanin, and G. Ferraccioli (2002). Arthr. Rheum. 46, 2029–2033.
B. Marston, A. Palanichamy, and J. H. Anolik (2010). Curr. Opin. Rheumatol. 22, 307.
J. Kim, H. Y. Kim, S. Y. Song, S. H. Go, H. S. Sohn, S. Baik, M. Soh, K. Kim, D. Kim, H. C. Kim, and N. Lee (2019). ACS Nano. 13, 3206–3217.
Acknowledgements
This research was supported by a Grant from the Key Research and Development Program of Shaanxi Province (No. 2019-SF-194).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cui, Y., Zhang, J., Liu, Y. et al. Engineering Mucic Acid Loaded Polyethylenimine@GoldNanoparticles for Improving the Treatment of Rheumatoid Arthritis. J Clust Sci 33, 2419–2427 (2022). https://doi.org/10.1007/s10876-021-02159-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-021-02159-5