Abstract
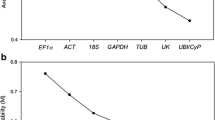
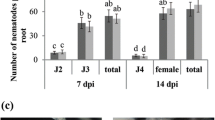
Biochar (BC) is known to enhance plant growth and may activate plant resistance to various soil borne pathogens. In this experiment, the rice husk (RH) BC was combined with biocontrol agents (BCAs), Bacillus subtilis and Trichoderma harzianum against Meloidogyne incognita in tomato and the efficacy of BC and BCAs in boosting overall plant biomass assessed side by side. BCAs along with 3% RH BC were applied to potting soil, tomato plants were transplanted, and then the soil was inoculated with second stage juveniles of Meloidogyne incognita. Bacillus subtilis +3% RH BC improved overall plant growth and reduced M. incognita damage. To unravel the resistance at the molecular level, root samples were taken 6‑ and 12-days post inoculation (dpi) to measure the expression of two resistance genes, PR-1b and JERF3, with the help of quantitative real-time polymerase chain reaction (qRT-PCR). The expression of PR-1b was upregulated 6 dpi for only BCAs while JERF3 remained unchanged except in only Bacillus subtilis treated plants. Similarly, the expression of both genes was found to be upregulated in some combined treatments at 12 dpi. The results suggested that the 3% BC application with BCAs effectively manages root-knot nematode, enhances overall plant biomass, and triggers defense related genes in tomato plants.
Zusammenfassung
Es ist bekannt, dass Biokohle (BC) das Pflanzenwachstum fördert und die Pflanzenresistenz gegen verschiedene bodenbürtige Krankheitserreger aktivieren kann. In diesem Versuch wurde die Reishülsen(RH)-Biokohle mit den Biokontrollmitteln (BCA) Bacillus subtilis und Trichoderma harzianum gegen Meloidogyne incognita in Tomaten kombiniert und die Wirksamkeit von BC und BCAs bei der Steigerung der Gesamtbiomasse der Pflanze nebeneinander geprüft. BCAs wurden zusammen mit 3 % RH BC auf die Blumenerde aufgebracht, die Tomaten verpflanzt und die Erde dann mit Meloidogyne incognita inokuliert. Bacillus subtilis +3 % RH BC verbesserte das gesamte Pflanzenwachstum und verringerte die Schäden durch M. incognita. Um die Resistenz auf molekularer Ebene zu entschlüsseln, wurden 6 und 12 Tage nach der Inokulation (dpi) Wurzelproben entnommen, um die Expression von zwei Resistenzgenen, PR-1b und JERF3, mithilfe der quantitativen Echtzeit-Reverse-Transkriptions-Polymerase-Kettenreaktion (qRT-PCR) zu messen. Die Expression von PR-1b wurde 6 dpi nur bei BCAs hochreguliert, während JERF3 unverändert blieb, außer bei mit Bacillus subtilis behandelten Pflanzen. Ebenso wurde festgestellt, dass die Expression beider Gene in einigen Kombinationsbehandlungen 12 Tage nach der Inokulation hochreguliert war. Die Kohärenz der Ergebnisse deutet darauf hin, dass die Anwendung von 3 % BC mit BCA Wurzelknotennematoden effektiv bekämpft, die gesamte Pflanzenbiomasse erhöht und abwehrbezogene Gene in Tomatenpflanzen aktiviert.



Similar content being viewed by others
References
Ali MA, Shahzadi M, Zahoor A, Dababat AA, Toktay H, Bakhsh A et al (2019) Resistance to cereal cyst nematodes in wheat and barley: an emphasis on classical and modern approaches. Int J Mol Sci 20:432
Ali MA, Azeem F, Abbas A, Joiya FA, Li H, Dababat AA (2017) Transgenic strategies for enhancement of nematode resistance in plants. Front Plant Sci 8:750
Amonette JE (2013) Letter report for characterization of biochar (no. PNNL-22391). Pacific Northwest National Lab. (PNNL), Richland
Anwar SA, McKenry MV (2012) Incidence and population density of plant-parasitic nematodes infecting vegetable crops and associated yield losses in Punjab, Pakistan. Pak J Zoo 44(2):327–333
Arguelles-Arias A, Ongena M, Halimi B, Lara Y, Brans A, Joris B, Fickers P (2009) Bacillus amyloliquefaciens GA1 as a source of potent antibiotics and other secondary metabolites for biocontrol of plant pathogens. Microb Cell Fact 8(1):63
Arshad U, Naveed M, Javed N, Gogi MD, Ali MA (2020) Biochar application from different feedstocks enhances plant growth and resistance against Meloidogyne incognita in tomato. Int J Agri Biol 24(4):961–968
Awad YM, Ok YS, Abrigata J, Beiyuan J, Beckers F, Tsang DC, Rinklebe J (2018) Pine sawdust biomass and biochars at different pyrolysis temperatures change soil redox processes. Sci Total Environ 625:147–154
Basyony AG, Abo-Zaid GA (2018) Biocontrol of the root-knot nematode, Meloidogyne incognita, using an eco-friendly formulation from Bacillus subtilis, lab. and greenhouse studies. Egyp J Biol Pest Control 28(1):1–3
Bhise KK, Dandge PB (2019) Mitigation of salinity stress in plants using plant growth promoting bacteria. Symbiosis 79(3):191–204
Bridge J, Page SLJ (1980) Estimation of root-knot nematode infestation levels on roots using a rating chart. Int J Pest Manag 26(3):296–298
Brugger SD, Baumberger C, Jost M, Jenni W, Brugger U, Mühlemann K (2012) Automated counting of bacterial colony forming units on agar plates. Plos One 7(3). https://doi.org/10.1371/journal.pone.0033695
Cao H, Bowling SA, Gordon AS, Dong X (1994) Characterization of an Arabidopsis mutant that is nonresponsive to inducers of systemic acquired resistance. Plant Cell 6(11):1583–1592
Choudhary DK, Johri BN (2009) Interactions of Bacillus spp. and plants–with special reference to induced systemic resistance (ISR). Microbiol Res 164(5):493–513
De Medeiros HA, de Araújo Filho JV, De Freitas LG, Castillo P, Rubio MB, Hermosa R, Monte E (2017) Tomato progeny inherit resistance to the nematode Meloidogyne javanica linked to plant growth induced by the biocontrol fungus Trichoderma atroviride. Sci Rep 7(1):1–13
Derksen H, Rampitsch C, Daayf F (2013) Signaling cross-talk in plant disease resistance. Plant Sci 207:79–87
Devi G, Yadav RK, Thakur NS (2007) Screening of tomato varieties/lines for resistance against root-knot nematode (Meloidogyne incognita). Ind J Nematol 37(1):83–84
Elad Y, David DR, Harel YM, Borenshtein M, Kalifa HB, Silber A, Graber ER (2010) Induction of systemic resistance in plants by biochar, a soil-applied carbon sequestering agent. Phytopathology 100(9):913–921
Elkelany US, El-Mougy NS, Abdel-Kader MM (2020) Management of root-knot nematode Meloidogyne incognita of eggplant using some growth-promoting rhizobacteria and chitosan under greenhouse conditions. Egyp J Biol Pest Control (1):1–7. https://doi.org/10.1186/s41938-020-00334-w
Eltayeb FME (2017) The effects of Bacillus subtilis bacteria on Meloidogyne javanica (nematode) infection and tomato plant growth. Eur J Adv Res Biol Life Sci 5(2):45–51
Fudali SL, Wang C, Williamson VM (2013) Ethylene signaling pathway modulates attractiveness of host roots to the root-knot nematode Meloidogyne hapla. Mol Plant Microbe Interact 26(1):75–86
Gao S, Wu H, Wang W, Yang Y, Xie S, Xie Y, Gao X (2013) Efficient colonization and harpins mediated enhancement in growth and biocontrol of wilt disease in tomato by B acillus subtilis. Lett Appl Microbiol 57(6):526–533
Ghareeb H, Bozsó Z, Ott PG, Repenning C, Stahl F, Wydra K (2011) Transcriptome of silicon-induced resistance against Ralstonia solanacearum in the silicon non-accumulator tomato implicates priming effect. Physiol Mol Plant Pathol 75(3):83–89
Giovannoni JJ (2004) Genetic regulation of fruit development and ripening. Plant Cell 16(suppl 1):S170–S180
Glazebrook J (2005) Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu Rev Phytopathol 43:205–227
Gouda S, Kerry RG, Das G, Paramithiotis S, Shin HS, Patra JK (2018) Revitalization of plant growth promoting rhizobacteria for sustainable development in agriculture. Microbiol Res 206:131–140
Graber ER, Harel YM, Kolton M, Cytryn E, Silber A, David DR, Elad Y (2010) Biochar impact on development and productivity of pepper and tomato grown in fertigated soilless media. Plant Soil 337(1-2):481–496
Hale LE (2014) Biochar and plant growth promoting Rhizobacteria as soil amendments. Doctoral dissertation, UC Riverside
Hamamouch N, Li C, Seo PJ, Park CM, Davis EL (2011) Expression of Arabidopsis pathogenesis-related genes during nematode infection. Mol Plant Pathol 12(4):355–364
Harel YM, Elad Y, Rav-David D, Borenstein M, Shulchani R, Lew B, Graber ER (2012) Biochar mediates systemic response of strawberry to foliar fungal pathogens. Plant Soil 357(1-2):245–257
Holzapfel NP, Holzapfel BM, Champ S, Feldthusen J, Clements J, Hutmacher DW (2013) The potential role of lycopene for the prevention and therapy of prostate cancer: from molecular mechanisms to clinical evidence. Int J Mol Sci 14(7):14620–14646
Hooks CR, Wang KH, Ploeg A, McSorley R (2010) Using marigold (Tagetes spp.) as a cover crop to protect crops from plant-parasitic nematodes. Appl Soil Ecol 46(3):307–320
Huang WK, Ji HL, Gheysen G, Debode J, Kyndt T (2015) Biochar-amended potting medium reduces the susceptibility of rice to root-knot nematode infections. BMC Plant Biol 15(1):267
Janisiewicz WJ, Korsten L (2002) Biological control of postharvest diseases of fruits. Annu Rev Phytopathol 40(1):411–441
Jepson SB (1987) Identification of root-knot nematodes (Meloidogyne species). CAB Inter, Wallingford
Karami R, Kamgar S, Karparvarfard SH, Rasul M, Khan M (2019) Biodiesel production from tomato seed and its engine emission test and simulation using Artificial Neural Network. J Oil Gas Petrochem Technol 5:41–62
Lahm GP, Desaeger J, Smith BK, Pahutski TF, Rivera MA, Meloro T, Cordova D (2017) The discovery of fluazaindolizine: a new product for the control of plant parasitic nematodes. Bioorg Med Chem Lett 27(7):1572–1575
Leonetti P, Zonno MC, Molinari S, Altomare C (2017) Induction of SA-signaling pathway and ethylene biosynthesis in Trichoderma harzianum-treated tomato plants after infection of the root-knot nematode Meloidogyne incognita. Plant Cell Rep 36(4):621–631
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 25(4):402–408
Macedo NJ, Ferreira TL (2014) Maximizing total RNA yield from TRIzol reagent protocol: a feasibility study. In: ASEE zone I conference
Martínez-Medina A, Fernandez I, Lok GB, Pozo MJ, Pieterse CMJ, Van Wees SCM (2016) Shifting from priming of salicylic acid- to jasmonic acid-regulated defences by Trichoderma protects tomato against the root knot nematode Meloidogyne incognita. New Phytol. https://doi.org/10.1111/nph.14251
Molinari S, Leonetti P (2019) Bio-control agents activate plant immune response and prime susceptible tomato against root-knot nematodes. PLoS ONE 14(12). https://doi.org/10.1371/journal.pone.0213230
Morris S, Nicholls J (1978) An evaluation of optical density to estimate fungal spore concentrations in water suspensions. Strain 1:1240–1242
Munif A, Hallmann J, Sikora RA (2000) Evaluation of the biocontrol activity of endophytic bacteria from tomato against Meloidogyne incognita. Mededelingen Fac Landbouwkundige Toegepaste Biol Wet Univ Gent 65(2b):471–480
Nahar K, Kyndt T, de Vleesschauwer D, Höfte M, Gheysen G (2011) The jasmonate pathway is a key player in systemically induced defense against root knot nematodes in rice. Plant Physiol. 157:305–316
Oladele SO, Adeyemo AJ, Awodun MA (2019) Influence of rice husk biochar and inorganic fertilizer on soil nutrients availability and rain-fed rice yield in two contrasting soils. Geoderma 15(336):1–1
Padilla FM, Peña-Fleitas MT, Gallardo M, Thompson RB (2015) Threshold values of canopy reflectance indices and chlorophyll meter readings for optimal nitrogen nutrition of tomato. Ann Appl Biol 166(2):271–285
Palansooriya KN, Wong JTF, Hashimoto Y, Huang L, Rinklebe J, Chang SX, Bolan N, Wang H, Ok YS (2019) Response of microbial communities to biochar-amended soils: a critical review. Biochar 1(1):3–22
Paul D, Lade H (2014) Plant-growth-promoting rhizobacteria to improve crop growth in saline soils: a review. Agron Sustain Dev 34(4):737–752
Pozo MJ, Azcón-Aguilar C (2007) Unraveling mycorrhiza-induced resistance. Curr Opin Plant Biol 10(4):393–398
Pratiwi EPA, Shinogi Y (2016) Rice husk biochar application to paddy soil and its effects on soil physical properties, plant growth, and methane emission. Paddy Water Environ 14(4):521–532
Salazar-Ordóñez M, Sayadi S (2011) Environmental care in agriculture: a social perspective. J Agric Environ Ethics 24:243–258
Sharma SB, Sikora RA, Greco N, Di Vito M, Caubel G (1993) Screening techniques and sources of resistance to nematodes in cool season food legumes. Euphytica 73(1-2):59–66
Shoresh M, Yedidia I, Chet I (2005) Involvement of jasmonic acid/ethylene signaling pathway in the systemic resistance induced in cucumber by Trichoderma asperellum T203. Phytopathology 95(1):76–84
Steel RGD, Torrie JH, DickeyDA (1997) Principles and procedures of statistics - A Biometrical Approach. 3rd Ed. McGraw Hill, Inc. New York
Sánchez ME, Lindao E, Margaleff D, Martínez O, Morán A (2009) Pyrolysis of agricultural residues from rape and sunflowers: production and characterization of bio-fuels and biochar soil management. J Anal Appl Pyrolysis 85(1-2):142–144
Tao S, Wu Z, Wei M, Liu X, He Y, Ye BC (2019) Bacillus subtilis SL-13 biochar formulation promotes pepper plant growth and soil improvement. Can J Microbiol 65(5):333–342
Tornero P, Gadea J, Conejero V, Vera P (1997) Two PR‑1 genes from tomato are differentially regulated and reveal a novel mode of expression for a pathogenesis-related gene during the hypersensitive response and development. Mol Plant Microbe Interact 10(5):624–634
Uehara T, Sugiyama S, Matsuura H, Arie T, Masuta C (2010) Resistant and susceptible responses in tomato to cyst nematode are differentially regulated by salicylic acid. Plant Cell Physiol 51(9):1524–1536
Van Loon LC, Rep M, Pieterse CM (2006) Significance of inducible defense-related proteins in infected plants. Annu Rev Phytopathol 44:135–162
Viljoen JJ, Labuschagne N, Fourie H, Sikora RA (2019) Biological control of the root-knot nematode Meloidogyne incognita on tomatoes and carrots by plant growth-promoting rhizobacteria. Trop Plant Pathol 44(3):284–291
Visser R, Blok C, Leyh R, Fryda L (2019) Biochar as potting soil constituent and as carrier of Bacillus in the cultivation of Cyclamen. September 15–20 (https://dc.engconfintl.org/biochar_ii/28/)
Vitti A, La Monaca E, Sofo A, Scopa A, Cuypers A, Nuzzaci M (2015) Beneficial effects of Trichoderma harzianum T‑22 in tomato seedlings infected by Cucumber mosaic virus (CMV). BioCont 60(1):135–147
Wang H, Huang Z, Chen Q, Zhang Z, Zhang H, Wu Y et al (2004) Ectopic overexpression of tomato JERF3 in tobacco activates downstream gene expression and enhances salt tolerance. Plant Mol Biol 55:183–192
Whitehead AG, Hemming JR (1965) A comparison of some quantitative methods of extracting small vermiform nematodes from soil. Ann Appl Biol 55(1):25–38
Ye J, Joseph SD, Ji M, Nielsen S, Mitchell DR, Donne S, Horvat J, Wang J, Munroe P, Thomas T (2017) Chemolithotrophic processes in the bacterial communities on the surface of mineral-enriched biochars. ISME J 11:1087
Zhao D, Zhao H, Zhao D, Zhu X, Wang Y, Duan Y, Xuan Y, Chen L (2018) Isolation and identification of bacteria from rhizosphere soil and their effect on plant growth promotion and root-knot nematode disease. Biol Contr 119:12–19
Zwart DC, Kim SH (2012) Biochar amendment increases resistance to stem lesions caused by Phytophthora spp. in tree seedlings. Hort Sci 47(12):1736–1740
Acknowledgements
The authors are highly thankful to Higher Education Commission of Pakistan for supporting this research through Pak-Turk Researcher’s Mobility Grant No. 9‑5(Ph-1-MG-2) Pak-Turk-R&D-HEC-2017 and NRPU project number 8097.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
U. Arshad, F. Azeem, G. Mustafa, A. Bakhsh, H. Toktay, M. McGiffen, M.A. Nawaz, M. Naveed and M.A. Ali declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Arshad, U., Azeem, F., Mustafa, G. et al. Combined Application of Biochar and Biocontrol Agents Enhances Plant Growth and Activates Resistance Against Meloidogyne incognita in Tomato. Gesunde Pflanzen 73, 591–601 (2021). https://doi.org/10.1007/s10343-021-00580-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10343-021-00580-4