Abstract
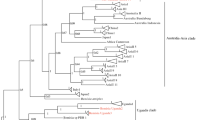
The whitefly, Bemisia tabaci (Gennadius), is responsible for significant yield losses in many crops, including potato, by sucking the phloem sap and transmitting a number of plant viruses. B. tabaci is a complex of cryptic species which is commonly designated as genetic groups. The B. tabaci genetic groups differ biologically with respect to host plant preference, insecticidal resistance, reproduction capacity, and ability to transmit begomoviruses. Therefore, understanding genetic variation among populations is important for establishing crop-specific distribution profile and management. We sequenced the mitochondrial cytochrome oxidase I (mtCOI) gene of B. tabaci collected from major potato growing areas of India. BLAST analysis of the 24 mtCOI sequences with reference Gene Bank sequences revealed four B. tabaci genetic groups prevailing in this region. mtCOI analysis exhibited the presence of Asia II 1, Asia II 5, Asia 1, and MEAM1 B. tabaci genetic groups. Our study highlighted that a new genetic group Asia II 5 has been detected in Indo-Gangetic Plains. Further virus–vector relationship study of ToLCNDV with Asia II 5 B. tabaci revealed that females are efficient vector of this virus as compared to males. This behavior of females might be due to their ability to acquire more virus titer than males. This study will help in better understanding of whitefly genetic group mediated virus diseases.




Similar content being viewed by others
References
Ashwathappa KV, Venkataravanappa V, Reddy CNL, Reddy MK (2020) Association of tomato leaf curl New Delhi virus with mosaic and leaf curl disease of Chrysanthemum and its whitefly cryptic species. Indian Phytopathol 73:533–542. https://doi.org/10.1007/s42360-020-00214-1
Banks GK, Colvin J, Reddy RVC et al (2001) First report of the Bemisia tabaci B biotype in India and an associated tomato leaf curl virus disease Epidemic. Plant Dis 85:231–231. https://doi.org/10.1094/pdis.2001.85.2.231c
Bedford ID, Briddon RW, Brown JK et al (1994) Geminivirus transmission and biological characterisation of Bemisia tabaci (Gennadius) biotypes from different geographic regions. Ann Appl Biol 125:311–325. https://doi.org/10.1111/j.1744-7348.1994.tb04972.x
Boykin LM, De Barro PJ (2014) A practical guide to identifying members of the Bemisia tabaci species complex: and other morphologically identical species. Front Ecol Evol 2:45. https://doi.org/10.3389/FEVO.2014.00045
Boykin LM, Shatters RG, Rosell RC et al (2007) Global relationships of Bemisia tabaci (Hemiptera: Aleyrodidae) revealed using Bayesian analysis of mitochondrial COI DNA sequences. Mol Phylogenet Evol 44:1306–1319. https://doi.org/10.1016/J.YMPEV.2007.04.020
Briddon RW, Bull SE, Amin I et al (2003) Diversity of DNA β, a satellite molecule associated with some monopartite begomoviruses. Virology 312:106–121. https://doi.org/10.1016/S0042-6822(03)00200-9
Brown JK (1991) An update on the whitefly-transmitted geminiviruses in the Americas and the Caribbean basin. FAO Plant Prot Bull 39:5–23
Brown JK, Coats SA, Bedford ID et al (1995) Characterization and distribution of esterase electromorphs in the whitefly, Bemisia tabaci (Genn.) (Homoptera: Aleyrodidae). Biochem Genet 33:205–214. https://doi.org/10.1007/BF02401851
Byrne DN, Bellows TS (1991) Whitefly biology. Annu Rev Entomol 36(1):431–457. https://doi.org/10.1146/annurev.ento.36.1.431
Chandel RS, Banyal DK, Singh BP et al (2010) Integrated management of whitefly, Bemisia tabaci (Gennadius) and potato apical leaf curl virus in India. Potato Res 53:129–139. https://doi.org/10.1007/s11540-010-9152-3
Chowda-Reddy RV, Kirankumar M, Seal SE et al (2012) Bemisia tabaci phylogenetic groups in India and the relative transmission efficacy of tomato leaf curl Bangalore virus by an indigenous and an exotic population. J Integr Agric 11:235–248. https://doi.org/10.1016/S2095-3119(12)60008-2
Czosnek H, Hariton-Shalev A, Sobol I et al (2017) The incredible journey of begomoviruses in their whitefly vector. Viruses 9(10):273. https://doi.org/10.3390/v9100273
De Barro PJ (2005) Genetic structure of the whitefly Bemisia tabaci in the Asia-Pacific region revealed using microsatellite markers. Mol Ecol 14:3695–3718. https://doi.org/10.1111/j.1365-294X.2005.02700.x
De Barro PJ, Liu S-S, Boykin LM, Dinsdale AB (2011) Bemisia tabaci : A statement of species status. Annu Rev Entomol 56:1–19. https://doi.org/10.1146/annurev-ento-112408-085504
den Heuvel V, Johannes FJM, Verbeek M, Van der Wilk F (1994) Endosymbiotic bacteria associated with circulative transmission of potato leafroll virus by Myzus persicae. J Gen Virol 75:2559–2565. https://doi.org/10.1099/0022-1317-75-10-2559
Dinsdale A, Cook L, Riginos C et al (2010) Refined global analysis of Bemisia tabaci (Hemiptera: Sternorrhyncha: Aleyrodoidea: Aleyrodidae) mitochondrial cytochrome oxidase 1 to identify species level genetic boundaries. Ann Entomol Soc Am 103:196–208. https://doi.org/10.1603/an09061
Drost YC, van Lenteren JC, van Roermund HJW (1998) Life-history parameters of different biotypes of Bemisia tabaci (Hemiptera: Aleyrodidae) in relation to temperature and host plant: a selective review. Bull Entomol Res 88(3):219–230. https://doi.org/10.1017/s0007485300025840
Ellango R, Singh ST, Rana VS et al (2015) Distribution of Bemisia tabaci genetic groups in India. Environ Entomol 44:1258–1264. https://doi.org/10.1093/ee/nvv062
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution (n y) 39:783–791. https://doi.org/10.1111/j.1558-5646.1985.tb00420.x
Garg ID, Khurana SP, Shiv K, Lakra BS (2001) Association of a geminivirus with potato apical leaf curl in India and its immuno-electron microscopic detection. J Indian Potato Assoc 28:227–232
Gottlieb Y, Zchori-Fein E, Mozes-Daube N et al (2010) The transmission efficiency of tomato yellow leaf curl virus by the whitefly, Bemisia tabaci is correlated with the presence of a specific symbiotic bacterium species. J Virol 84:9310–9317. https://doi.org/10.1128/jvi.00423-10
Guo T, Zhao J, Pan LL et al (2018) The level of midgut penetration of two begomoviruses affects their acquisition and transmission by two species of Bemisia tabaci. Virology 515:66–73. https://doi.org/10.1016/j.virol.2017.12.004
Hogenhout SA, Ammar ED, Whitfield AE, Redinbaugh MG (2008) Insect vector interactions with persistently transmitted viruses. Annu Rev Phytopathol 46:327–359. https://doi.org/10.1146/annurev.phyto.022508.092135
Hussain S, Farooq M, Malik HJ et al (2019) Whole genome sequencing of Asia II 1 species of whitefly reveals that genes involved in virus transmission and insecticide resistance have genetic variances between Asia II 1 and MEAM1 species. BMC Genom 20(1):1–13. https://doi.org/10.1186/s12864-019-5877-9
Jeevalatha A, Kaundal P, Venkatasalam EP et al (2013) Uniplex and duplex pcr detection of geminivirus associated with potato apical leaf curl disease in India. J Virol Methods 193:62–67. https://doi.org/10.1016/j.jviromet.2013.05.012
Jeevalatha A, Singh BP, Kaundal P et al (2014) RCA-PCR: A robust technique for the detection of tomato leaf curl New Delhi virus-potato at ultra low virus titre. Potato J 41:76–80
Jiang YX, De Blas C, Barrios L, Fereres A (2000) Correlation between whitefly (Homoptera: Aleyrodidae) feeding behavior and transmission of tomato yellow leaf curl virus. Ann Entomol Soc Am 93:573–579. https://doi.org/10.1603/0013-8746(2000)093[0573:CBWHAF]2.0.CO;2
Kanakala S, Ghanim M (2019) Global genetic diversity and geographical distribution of Bemisia tabaci and its bacterial endosymbionts. PLoS ONE 14(3):e0213946. https://doi.org/10.1371/journal.pone.0213946
Kliot A, Ghanim M (2013) The role of bacterial chaperones in the circulative transmission of plant viruses by insect vectors. Viruses 5:1516–1535. https://doi.org/10.3390/v5061516
Kreuze JF, Souza-Dias JAC, Jeevalatha A, Figueira AR, Valkonen JP, Jones RA (2020) Viral diseases in potato. In: Campos H, Ortiz O (eds) The potato crop. Springer, Cham, pp 389–430. https://doi.org/10.1007/978-3-030-28683-5_11
Lakra BS (2003) Potato apical leaf curl begomovirus-symptoms, appraisal of a scale and losses in potato crop. Potato J 30:119–120
Lee W, Park J, Lee GS et al (2013) Taxonomic status of the Bemisia tabaci complex (Hemiptera: Aleyrodidae) and reassessment of the number of its constituent species. PLoS One 8(5):e63817. https://doi.org/10.1371/journal.pone.0063817
Muniyappa V, Venkatesh HM, Ramappa HK et al (2000) Tomato leaf curl virus from Bangalore (ToLCV-Ban4): sequence comparison with Indian ToLCV isolates, detection in plants and insects, and vector relationships. Arch Virol 145:1583–1598. https://doi.org/10.1007/s007050070078
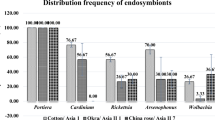
Naga KC, Subhash S, Bhatnagar A, Tiwari RK et al (2020) Influence of host plants on virus acquisition and endosymbionts of whitefly, Bemisia tabaci (Gennadius). Indian J Entomol 83:1–5. https://doi.org/10.5958/0974-8172.2020.00206.0
Naranjo SE, Ellsworth PC (2001) Challenges and opportunities for pest management of Bemisia tabaci in the new century. Crop Prot 9(20):707
Narayana YD, Muniyappa V, Narayana YD (1996) Virus vector relationships of a planthopper (Peregrinus maidis) borne sorghum stripe tenuivirus. Int J Pest Manag 42:165–170. https://doi.org/10.1080/09670879609371990
Navas-Castillo J, Fiallo-Olivé E, Sánchez-Campos S (2011) Emerging virus diseases transmitted by whiteflies. Annu Rev Phytopathol 49:219–248. https://doi.org/10.1146/annurev-phyto-072910-095235
Naveen NC, Chaubey R, Kumar D et al (2017) Insecticide resistance status in the whitefly, Bemisia tabaci genetic groups Asia-I, Asia-II-1 and Asia-II-7 on the Indian subcontinent. Sci Rep 7:1–15. https://doi.org/10.1038/srep40634
Ning W, Shi X, Liu B et al (2015) Transmission of tomato yellow leaf curl virus by Bemisia tabaci as affected by whitefly sex and biotype. Sci Rep 5:1–8. https://doi.org/10.1038/srep10744
Paredes-Montero JR, Hameed U, Zia-Ur-Rehman M et al (2019) Demographic expansion of the predominant Bemisia tabaci (Gennadius) (Hemiptera: Aleyrodidae) mitotypes associated with the cotton leaf curl virus epidemic in Pakistan. Ann Entomol Soc Am 112(3):265–280. https://doi.org/10.1093/aesa/saz002
Peng J, Xie G, Zhang S et al (2020) Higher ramie mosaic virus transmission efficiency by females than by males of Bemisia tabaci MED. Sci Rep 10:5–10. https://doi.org/10.1038/s41598-019-57343-5
Polston JE (1990) Association of the nucleic acid of squash leaf curl Geminivirus with the whitefly, Bemisia Tabaci. Phytopathology 80(9):850–856. https://doi.org/10.1094/Phyto-80-850
Rana VS, Popli S, Saurav GK et al (2016) A Bemisia tabaci midgut protein interacts with begomoviruses and plays a role in virus transmission. Cell Microbiol 18(5):663–678. https://doi.org/10.1111/cmi.12538
Rekha AR, Maruthi MN, Muniyappa V, Colvin J (2005) Occurrence of three genotypic clusters of Bemisia tabaci and the rapid spread of the B biotype in south India. Entomol Exp Appl 117:221–233. https://doi.org/10.1111/J.1570-7458.2005.00352.X
Rotenberg D, Kumar NKK, Ullman DE et al (2009) Variation in tomato spotted wilt virus titer in Frankliniella occidentalis and its association with frequency of transmission. Phytopathology 99:404–410. https://doi.org/10.1094/PHYTO-99-4-0404
Saeed ST, Kumar B, Shasany AK, Samad A (2017) Molecular identification of chilli leaf curl india virus along with betasatellite molecule causing leaf curl disease of menthol mint (Mentha arvensis var. Kosi) in India. J Gen Plant Pathol 83:333–336. https://doi.org/10.1007/s10327-017-0730-y
Saha A, Saha D (2014) Molecular detection and partial characterization of a begomovirus causing leaf curl disease of potato in sub-Himalayan West Bengal, India. Artic J Environ Biol 35:601–606
Segev L, Wintermantel WM, Polston JE, Lapidot M (2004) First report of tomato chlorosis virus in Israel. Plant Dis 88:1160–1160. https://doi.org/10.1094/pdis.2004.88.10.1160a
Shah MA, Malik K, Bhatnagar A et al (2019) Effect of temperature and cropping sequence on the infestation pattern of Bemisia tabaci in potato. Indian J Agric Sci 89:1802–1807
Shi X, Tang X, Zhang X et al (2018) Transmission efficiency, preference and behavior of Bemisia tabaci MEAM1 and MED under the influence of tomato chlorosis virus. Front Plant Sci 8:1–9. https://doi.org/10.3389/fpls.2017.02271
Simon C, Frati F, Beckenbach A et al (1994) Evolution, weighting, and phylogenetic utility of mitochondrial gene sequences and a compilation of conserved polymerase chain reaction primers. Ann Entomol Soc Am 87:651–701. https://doi.org/10.1093/aesa/87.6.651
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22(22):4673–4680
Usharani KS, Surendranath B, Paul-Khurana SM et al (2004) Potato leaf curl—a new disease of potato in northern India caused by a strain of tomato leaf curl New Delhi virus. Plant Pathol 53(2):235–235. https://doi.org/10.1111/j.0032-0862.2004.00959.x
Van De Wetering F, Van Der Hoek M, Goldbach R, Peters D (1999) Differences in tomato spotted wilt virus vector competency between males and females of Frankliniella occidentalis. Entomol Exp Appl 93:105–112. https://doi.org/10.1046/j.1570-7458.1999.00567.x
Venkatasalam EP, Singh S, Sivalingam PN et al (2011) Polymerase chain reaction and nucleic acid spot hybridisation detection of begomovirus(es) associated with apical leaf curl disease of potato. Arch Phytopathol Plant Prot 44:987–992. https://doi.org/10.1080/03235401003633741
Wintermantel WM, Gilbertson RL, McCreight JD, Natwick ET (2016) Host-specific relationship between virus titer and whitefly transmission of cucurbit yellow stunting disorder virus. Plant Dis 100:92–98. https://doi.org/10.1094/PDIS-11-14-1119-RE
Xie W, Xu YX, Jiao XG, Zhang YJ (2012) High efficient of females of B-type Bemisia tabaci as males in transmitting the whitefly-borne tomato yellow leaf curl virus to tomato plant with Q-PCR method confirmation. Commun Integr Biol 5:543–545. https://doi.org/10.4161/cib.21692
Acknowledgements
This study was supported by grants from Indian Council of Agricultural Research, New Delhi, India to ICAR-CPRI, Shimla.
Author information
Authors and Affiliations
Contributions
KCN and SSH conceptualization. KCN, SK, RMS, GMB, AS and GKP methodology. KCN, SSI, RK, TB experimentation. GV software analysis. KCN, RKT, SS, AB writing-original draft.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Rights and permissions
About this article
Cite this article
Naga, K.C., Siddappa, S., Kumar, R. et al. A new record of Asia II 5 genetic group of Bemisia tabaci (Gennadius) in the major potato growing areas of India and its relationship with tomato leaf curl New Delhi virus infecting potato. 3 Biotech 11, 421 (2021). https://doi.org/10.1007/s13205-021-02966-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-02966-7