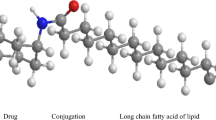
Abstract
Lipophilic conjugates (LCs) of small molecule drugs have been used widely in clinical and pre-clinical studies to achieve a number of pharmacokinetic and therapeutic benefits. For example, lipophilic derivatives of drugs are employed in several long acting injectable products to provide sustained drug exposure for hormone replacement therapy and to treat conditions such as neuropsychiatric diseases. LCs can also be used to modulate drug metabolism, and to enhance drug permeation across membranes, either by increasing lipophilicity to enhance passive diffusion or by increasing protein-mediated active transport. Furthermore, such conjugation strategies have been employed to promote drug association with endogenous macromolecular carriers (e.g. albumin and lipoproteins), and this in turn results in altered drug distribution and pharmacokinetic profiles, where the changes can be ‘general’ (e.g. prolonged plasma half-life) or ‘specific’ (e.g. enhanced delivery to specific tissues in parallel with the macromolecular carriers). Another utility of LCs is to enhance the encapsulation of drugs within engineered nanoscale drug delivery systems, in order to best take advantage of the targeting and pharmacokinetic benefits of nanomedicines. The current review provides a summary of the mechanisms by which lipophilic conjugates, including in combination with delivery vehicles, can be used to control drug delivery, distribution and therapeutic profiles. The article is structured into sections which highlight a specific benefit of LCs and then demonstrate this benefit with case studies. The review attempts to provide a toolbox to assist researchers to design and optimise drug candidates, including consideration of drug-formulation compatibility.
























Similar content being viewed by others
References
Rautio J, Meanwell NA, Di L, Hageman MJ. The expanding role of prodrugs in contemporary drug design and development. Nat Rev Drug Discov. 2018;17(8):559–87.
Rautio J, Kumpulainen H, Heimbach T, Oliyai R, Oh D, Jarvinen T, Savolainen J. Prodrugs: design and clinical applications. Nat Rev Drug Discov. 2008;7(3):255–70.
Huttunen KM, Raunio H, Rautio J. Prodrugs--from serendipity to rational design. Pharmacol Rev. 2011;63(3):750–71.
Sofia MJ, Bao D, Chang W, Du J, Nagarathnam D, Rachakonda S, Reddy PG, Ross BS, Wang P, Zhang H-R, Bansal S, Espiritu C, Keilman M, Lam AM, Steuer HMM, Niu C, Otto MJ, Furman PA. Discovery of a β-d-2′-Deoxy-2′-α-fluoro-2′-β-C-methyluridine Nucleotide Prodrug (PSI-7977) for the Treatment of Hepatitis C Virus. Journal of Medicinal Chemistry. 2010;53(19):7202–18.
Babusis D, Curry MP, Kirby B, Park Y, Murakami E, Wang T, Mathias A, Afdhal N, McHutchison JG, Ray AS. Sofosbuvir and Ribavirin Liver Pharmacokinetics in Patients Infected with Hepatitis C Virus. Antimicrobial Agents and Chemotherapy. 2018;62(5)
Prodrugs: Challenges and Rewards, Part 1. New York: Springer; 2007.
Prodrugs: Challenges and Rewards, Part 2; New York: Springer; 2007.
Long JZ, Cravatt BF. The Metabolic Serine Hydrolases and Their Functions in Mammalian Physiology and Disease. Chemical Reviews. 2011;111(10):6022–63.
Bachovchin DA, Cravatt BF. The pharmacological landscape and therapeutic potential of serine hydrolases. Nature Reviews Drug Discovery. 2012;11(1):52–68.
Liederer BM, Borchardt RT. Enzymes involved in the bioconversion of ester-based prodrugs. Journal of Pharmaceutical Sciences. 2006;95(6):1177–95.
Knudsen LB, Lau J. The Discovery and Development of Liraglutide and Semaglutide. Frontiers in Endocrinology. 2019;10.00155.
Bech EM, Pedersen SL, Jensen KJ. Chemical Strategies for Half-Life Extension of Biopharmaceuticals: Lipidation and Its Alternatives. ACS Med Chem Lett. 2018;9(7):577–80.
Abdallah M, Mullertz OO, Styles IK, Morsdorf A, Quinn JF, Whittaker MR, Trevaskis NL. Lymphatic targeting by albumin-hitchhiking: Applications and optimisation. Journal of Controlled Release. 2020;327:117–28.
Liu HP, Moynihan KD, Zheng YR, Szeto GL, Li AV, Huang B, Van Egeren DS, Park C, Irvine DJ. Structure-based programming of lymph-node targeting in molecular vaccines. Nature. 2014;507(7493):519–22.
Liu Z, Chen X. Simple bioconjugate chemistry serves great clinical advances: albumin as a versatile platform for diagnosis and precision therapy. Chem Soc Rev. 2016;45(5):1432–56.
Siegel RA, Rathbone MJ. Overview of Controlled Release Mechanisms. In: Siepmann J, Siegel RA, Rathbone MJ, editors. Fundamentals and Applications of Controlled Release Drug Delivery: Springer US; 2012. p. 19–43.
Nkanga CI, Fisch A, Rad-Malekshahi M, Romic MD, Kittel B, Ullrich T, Wang J, Krause RWM, Adler S, Lammers T, Hennink WE, Ramazani F. Clinically established biodegradable long acting injectables: An industry perspective. Adv Drug Deliv Rev. 2020;167:19–46.
Remenar JF. Making the leap from daily oral dosing to long-acting injectables: lessons from the antipsychotics. Mol Pharm. 2014;11(6):1739–49.
Sanrame CN, Remenar JF, Blumberg LC, Waters J, Dean RL, Dong N, Kriksciukaite K, Cao P, Almarsson O. Prodrugs of pioglitazone for extended-release (XR) injectable formulations. Mol Pharm. 2014;11(10):3617–23.
Sillman B, Bade AN, Dash PK, Bhargavan B, Kocher T, Mathews S, Su H, Kanmogne GD, Poluektova LY, Gorantla S, McMillan J, Gautam N, Alnouti Y, Edagwa B, Gendelman HE. Creation of a long-acting nanoformulated dolutegravir. Nat Commun. 2018;9(1):443.
Mihajlovic T, Kachrimanis K, Graovac A, Djuric Z, Ibric S. Improvement of Aripiprazole Solubility by Complexation with (2-Hydroxy)propyl-β-cyclodextrin Using Spray Drying Technique. AAPS PharmSciTech. 2012;13(2):623–31.
Ray AS, Fordyce MW, Hitchcock MJM. Tenofovir alafenamide: A novel prodrug of tenofovir for the treatment of Human Immunodeficiency Virus. Antiviral Research. 2016;125:63–70.
Beaumont K, Webster R, Gardner I, Dack K. Design of ester prodrugs to enhance oral absorption of poorly permeable compounds: challenges to the discovery scientist. Curr Drug Metab. 2003;4(6):461–85.
Wiemer AJ, Wiemer DF. Prodrugs of phosphonates and phosphates: crossing the membrane barrier. Top Curr Chem. 2015;360:115–60.
USFDA. Approval Package for NDA 22-512/S-04 Pradaxa (dabigatran etexilate mesylate). https://wwwaccessdatafdagov/drugsatfda_docs/nda/2011/022512Orig1s004pdf. 2012.
Stinchcomb AL, Swaan PW, Ekabo O, Harris KK, Browe J, Hammell DC, Cooperman TA, Pearsall M. Straight-chain naltrexone ester prodrugs: Diffusion and concurrent esterase biotransformation in human skin. Journal of Pharmaceutical Sciences. 2002;91(12):2571–8.
Fukano Y, Kawazu K. Disposition and Metabolism of a Novel Prostanoid Antiglaucoma Medication, Tafluprost, Following Ocular Administration to Rats. Drug Metabolism and Disposition. 2009;37(8):1622–34.
Doh H-J, Cho W-J, Yong C-S, Choi H-G, Kim JS, Lee C-H, Kim D-D. Synthesis and evaluation of Ketorolac ester prodrugs for transdermal delivery. Journal of Pharmaceutical Sciences. 2003;92(5):1008–17.
Fang J-Y, Leu Y-L. Prodrug Strategy for Enhancing Drug Delivery via Skin. CDDT. 2006;3(3):211–24.
Gote V, Ansong M, Pal D. Prodrugs and nanomicelles to overcome ocular barriers for drug penetration. Expert Opinion on Drug Metabolism & Toxicology. 2020;16(10):885–906.
Liu KS, Sung KC, Al-Suwayeh SA, Ku MC, Chu CC, Wang JJ, Fang JY. Enhancement of transdermal apomorphine delivery with a diester prodrug strategy. Eur J Pharm Biopharm. 2011;78(3):422–31.
Mandell AI, Stentz F, Kitabchi AE. Dipivalyl Epinephrine: A New Pro-Drug in the Treatment of Glaucoma. Ophthalmology. 1978;85(3):268–75.
Mills KB, Jacobs NA. A single-blind randomised trial comparing adrenaline 1.0% with dipivalyl epinephrine (propine) 0.1% in the treatment of open-angle glaucoma and ocular hypertension. British Journal of Ophthalmology. 1988;72(6):465–8.
Kass MA, Mandell AI, Goldberg I, Paine JM, Becker B. Dipivefrin and Epinephrine Treatment of Elevated Intraocular Pressure: A Comparative Study. Archives of Ophthalmology. 1979;97(10):1865–6.
Daneman R, Prat A. The blood-brain barrier. Cold Spring Harb Perspect Biol. 2015;7(1):a020412–2.
Pardridge WM. The Blood-Brain Barrier: Bottleneck in Brain Drug Development. NeuroRX. 2005;2(1):3–14.
Pandit R, Chen LY, Gotz J. The blood-brain barrier: Physiology and strategies for drug delivery. Advanced Drug Delivery Reviews. 2020;165-166:1-14.
Banks WA. From blood-brain barrier to blood-brain interface: new opportunities for CNS drug delivery. Nature Reviews Drug Discovery. 2016;15(4):275–92.
Chen Y, Liu LH. Modern methods for delivery of drugs across the blood-brain barrier. Advanced Drug Delivery Reviews. 2012;64(7):640–65.
Greig NH, Genka S, Daly EM, Sweeney DJ, Rapoport SI. Physicochemical and pharmacokinetic parameters of seven lipophilic chlorambucil esters designed for brain penetration. Cancer Chemotherapy and Pharmacology. 1990;25(5):311–9.
Oldendorf WH, Hyman S, Braun L, Oldendorf SZ. Blood-brain barrier: penetration of morphine, codeine, heroin, and methadone after carotid injection. Science. 1972;178(4064):984–6.
Ettmayer P, Amidon GL, Clement B, Testa B. Lessons Learned from Marketed and Investigational Prodrugs. Journal of Medicinal Chemistry. 2004;47(10):2393–404.
Murakami T. A Minireview: Usefulness of Transporter-Targeted Prodrugs in Enhancing Membrane Permeability. Journal of Pharmaceutical Sciences. 2016;105(9):2515–26.
Burke AC, Giles FJ. Elacytarabine - lipid vector technology overcoming drug resistance in acute myeloid leukemia. Expert Opin Investig Drugs. 2011;20(12):1707–15.
Tanino T, Nawa A, Kondo E, Kikkawa F, Daikoku T, Tsurumi T, Luo C, Nishiyama Y, Takayanagi Y, Nishimori K, Ichida S, Wada T, Miki Y, Iwaki M. Paclitaxel-2′-Ethylcarbonate Prodrug Can Circumvent P-glycoprotein-mediated Cellular Efflux to Increase Drug Cytotoxicity. Pharmaceutical Research. 2007;24(3):555–65.
Benes J, Parada A, Figueiredo AA, Alves PC, Freitas AP, Learmonth DA, Cunha RA, Garrett J, Soares-da-Silva P. Anticonvulsant and Sodium Channel-Blocking Properties of Novel 10,11-Dihydro-5H-dibenz[b,f]azepine-5-carboxamide Derivatives. Journal of Medicinal Chemistry. 1999;42(14):2582–7.
Almeida L, Soares-da-Silva P. Eslicarbazepine acetate (BIA 2-093). Neurotherapeutics. 2007;4(1):88–96.
Cundy KC, Branch R, Chernov-Rogan T, Dias T, Estrada T, Hold K, Koller K, Liu XL, Mann A, Panuwat M, Raillard SP, Upadhyay S, Wu QQ, Xiang JN, Yan H, Zerangue N, Zhou CX, Barrett RW, Gallop MA. XP13512 (+/-)-1-( (alpha-isobutanoyloxyethoxy)carbonyl aminomethyl)-1-cyclohexane acetic acid , a novel gabapentin prodrug: I. Design, synthesis, enzymatic conversion to gabapentin, and transport by intestinal solute transporters. Journal of Pharmacology and Experimental Therapeutics. 2004;311(1):315–23.
Cundy KC, Annamalai T, Bu L, De Vera J, Estrela J, Luo W, Shirsat P, Torneros A, Yao FM, Zou J, Barrett RW, Gallop MA. XP13512 (+/-)-1-( (alpha-isobutanoyloxyethoxy)carbonyl aminomethyl)-1-cyclohexane acetic acid , a novel gabapentin prodrug: II. Improved oral bioavailability, dose proportionality, and colonic absorption compared with gabapentin in rats and monkeys. Journal of Pharmacology and Experimental Therapeutics. 2004;311(1):324–33.
Cundy KC, Sastry S, Luo WD, Zou JA, Moors TL, Canafax DM. Clinical Pharmacokinetics of XP13512, a Novel Transported Prodrug of Gabapentin. Journal of Clinical Pharmacology. 2008;48(12):1378–88.
Rais R, Fletcher S, Polli JE. Synthesis and In Vitro Evaluation of Gabapentin Prodrugs that Target the Human Apical Sodium-Dependent Bile Acid Transporter (hASBT). Journal of Pharmaceutical Sciences. 2011;100(3):1184–95.
Gokulgandhi MR, Barot M, Bagui M, Pal D, Mitra AK. Transporter-Targeted Lipid Prodrugs of Cyclic Cidofovir: A Potential Approach for the Treatment of Cytomegalovirus Retinitis. Journal of Pharmaceutical Sciences. 2012;101(9):3249–63.
Vadlapudi AD, Vadlapatla RK, Earla R, Sirimulla S, Bailey JB, Pal D, Mitra AK. Novel Biotinylated Lipid Prodrugs of Acyclovir for the Treatment of Herpetic Keratitis (HK): Transporter Recognition, Tissue Stability and Antiviral Activity. Pharmaceutical Research. 2013;30(8):2063–76.
Bhutia YD, Hung SW, Patel B, Lovin D, Govindarajan R. CNT1 expression influences proliferation and chemosensitivity in drug-resistant pancreatic cancer cells. Cancer Res. 2011;71(5):1825–35.
Bergman AM, Kuiper CM, Voorn DA, Comijn EM, Myhren F, Sandvold ML, Hendriks HR, Peters GJ. Antiproliferative activity and mechanism of action of fatty acid derivatives of arabinofuranosylcytosine in leukemia and solid tumor cell lines. Biochem Pharmacol. 2004;67(3):503–11.
Hidalgo M, Villalona-Calero MA, Eckhardt SG, Drengler RL, Rodriguez G, Hammond LA, Diab SG, Weiss G, Garner AM, Campbell E, Davidson K, Louie A, O'Neil JD, von Borstel R, Von Hoff DD, Rowinsky EK. Phase I and pharmacologic study of PN401 and fluorouracil in patients with advanced solid malignancies. J Clin Oncol. 2000;18(1):167–77.
Irby D, Du C, Li F. Lipid-Drug Conjugate for Enhancing Drug Delivery. Mol Pharm. 2017;14(5):1325–38.
Zaro JL. Lipid-Based Drug Carriers for Prodrugs to Enhance Drug Delivery. The AAPS Journal. 2015;17(1):83–92.
Chhikara BS, Mandal D, Parang K. Synthesis, anticancer activities, and cellular uptake studies of lipophilic derivatives of doxorubicin succinate. J Med Chem. 2012;55(4):1500–10.
Osborne O, Peyravian N, Nair M, Daunert S, Toborek M. The Paradox of HIV Blood-Brain Barrier Penetrance and Antiretroviral Drug Delivery Deficiencies. Trends in Neurosciences. 2020;43(9):695–708.
Dalpiaz A, Paganetto G, Pavan B, Fogagnolo M, Medici A, Beggiato S, Perrone D. Zidovudine and ursodeoxycholic acid conjugation: design of a new prodrug potentially able to bypass the active efflux transport systems of the central nervous system. Mol Pharm. 2012;9(4):957–68.
Peters GJ, Adema AD, Bijnsdorp IV, Sandvold ML. Lipophilic prodrugs and formulations of conventional (deoxy)nucleoside and fluoropyrimidine analogs in cancer. Nucleosides Nucleotides Nucleic Acids. 2011;30(12):1168–80.
Liu J, Liu J, Zhao D, Ma N, Luan Y. Highly enhanced leukemia therapy and oral bioavailability from a novel amphiphilic prodrug of cytarabine. RSC Advances. 2016;6(42):35991–9.
Schwendener RA, Schott H. Treatment of l1210 murine leukemia with liposome-incorporated N4-hexadecyl-1-β-D-arabinofuranosyl cytosine. International Journal of Cancer. 1992;51(3):466–9.
Bohnert T, Gan LS. Plasma Protein Binding: From Discovery to Development. Journal of Pharmaceutical Sciences. 2013;102(9):2953–94.
Polo L, Valduga G, Jori G, Reddi E. Low-density lipoprotein receptors in the uptake of tumour photosensitizers by human and rat transformed fibroblasts. The International Journal of Biochemistry & Cell Biology. 2002;34(1):10–23.
Tanaka T, Shiramoto S, Miyashita M, Fujishima Y, Kaneo Y. Tumor targeting based on the effect of enhanced permeability and retention (EPR) and the mechanism of receptor-mediated endocytosis (RME). International Journal of Pharmaceutics. 2004;277(1-2):39–61.
Maeda H. The enhanced permeability and retention (EPR) effect in tumor vasculature: The key role of tumor-selective macromolecular drug targeting. Advances in Enzyme Regulation, 2001; 41:189-207.
van der Vusse GJ. Albumin as Fatty Acid Transporter. Drug Metabolism and Pharmacokinetics. 2009;24(4):300–7.
Kratz F. Albumin as a drug carrier: design of prodrugs, drug conjugates and nanoparticles. J Control Release. 2008;132(3):171–83.
Elsadek B, Kratz F. Impact of albumin on drug delivery--new applications on the horizon. J Control Release. 2012;157(1):4–28.
Sparreboom A, Wolff AC, Verweij J, Zabelina Y, van Zomeren DM, McIntire GL, Swindell CS, Donehower RC, Baker SD. Disposition of docosahexaenoic acid-paclitaxel, a novel taxane, in blood: In vitro and clinical pharmacokinetic studies. Clinical Cancer Research. 2003;9(1):151–9.
Sparreboom A, van Zuylen L, Brouwer E, Loos WJ, de Bruijn P, Gelderblom B, Pillay M, Nooter K, Stoter G, Verweij J. Cremophor EL-mediated alteration of paclitaxel distribution in human blood: Clinical pharmacokinetic implications. Cancer Research. 1999;59(7):1454–7.
Homsi J, Bedikian AY, Kim KB, Papadopoulos NE, Hwu W-J, Mahoney SL, Hwu P. Phase 2 open-label study of weekly docosahexaenoic acid-paclitaxel in cutaneous and mucosal metastatic melanoma patients. Melanoma Res. 2009;19(4):238–42.
Zheng YR, Suntharalingam K, Johnstone TC, Yoo H, Lin W, Brooks JG, Lippard SJ. Pt(IV) prodrugs designed to bind non-covalently to human serum albumin for drug delivery. J Am Chem Soc. 2014;136(24):8790–8.
Boros E, Caravan P. Structure–Relaxivity Relationships of Serum Albumin Targeted MRI Probes Based on a Single Amino Acid Gd Complex. Journal of Medicinal Chemistry. 2013;56(4):1782–6.
Caravan P, Cloutier NJ, Greenfield MT, McDermid SA, Dunham SU, Bulte JWM, Amedio JC, Looby RJ, Supkowski RM, Horrocks WD, McMurry TJ, Lauffer RB. The Interaction of MS-325 with Human Serum Albumin and Its Effect on Proton Relaxation Rates. Journal of the American Chemical Society. 2002;124(12):3152–62.
de Haën C, Anelli PL, Lorusso V, Morisetti A, Maggioni F, Zheng J, Uggeri F, Cavagna FM. Gadocoletic acid trisodium salt (b22956/1): a new blood pool magnetic resonance contrast agent with application in coronary angiography. Invest Radiol. 2006;41(3):279–91.
Meloni MM, Barton S, Xu L, Kaski JC, Song W, He T. Contrast agents for cardiovascular magnetic resonance imaging: an overview. J Mater Chem B. 2017;5(29):5714–25.
Spada A, Emami J, Tuszynski JA, Lavasanifar A. The Uniqueness of Albumin as a Carrier in Nanodrug Delivery. Molecular Pharmaceutics. 2021;18(5):1862–94.
Wahsner J, Gale EM, Rodríguez-Rodríguez A, Caravan P. Chemistry of MRI Contrast Agents: Current Challenges and New Frontiers. Chemical Reviews. 2019;119(2):957–1057.
Gershkovich P, Hoffman A. Uptake of lipophilic drugs by plasma derived isolated chylomicrons: Linear correlation with intestinal lymphatic bioavailability. European Journal of Pharmaceutical Sciences. 2005;26(5):394–404.
Trevaskis NL, Charman WN, Porter CJH. Lipid-based delivery systems and intestinal lymphatic drug transport: A mechanistic update. Advanced Drug Delivery Reviews. 2008;60(6):702–16.
Trevaskis NL, Shanker RM, Charman WN, Porter CJH. The Mechanism of Lymphatic Access of Two Cholesteryl Ester Transfer Protein Inhibitors (CP524,515 and CP532,623) and Evaluation of Their Impact on Lymph Lipoprotein Profiles. Pharmaceutical Research. 2010;27(9):1949–64.
Wasan KM, Brocks DR, Lee SD, Sachs-Barrable K, Thornton SJ. Impact of lipoproteins on the biological activity and disposition of hydrophobic drugs: implications for drug discovery. Nature Reviews Drug Discovery. 2008;7(1):84–99.
Humberstone AJ, Porter CJ, Edwards GA, Charman WN. Association of halofantrine with postprandially derived plasma lipoproteins decreases its clearance relative to administration in the fasted state. J Pharm Sci. 1998;87(8):936–42.
Shayeganpour A, Jun AS, Brocks DR. Pharmacokinetics of Amiodarone in hyperlipidemic and simulated high fat-meal rat models. Biopharm Drug Dispos. 2005;26(6):249–57.
Wasan KM, Brocks DR, Lee SD, Sachs-Barrable K, Thornton SJ. Impact of lipoproteins on the biological activity and disposition of hydrophobic drugs: implications for drug discovery. Nat Rev Drug Discov. 2008;7(1):84–99.
Koziolek M, Alcaro S, Augustijns P, Basit AW, Grimm M, Hens B, Hoad CL, Jedamzik P, Madla CM, Maliepaard M, Marciani L, Maruca A, Parrott N, Pávek P, Porter CJH, Reppas C, van Riet-Nales D, Rubbens J, Statelova M, et al. The mechanisms of pharmacokinetic food-drug interactions – A perspective from the UNGAP group. European Journal of Pharmaceutical Sciences. 2019;134:31–59.
Busatto S, Walker SA, Grayson W, Pham A, Tian M, Nesto N, Barklund J, Wolfram J. Lipoprotein-based drug delivery. Advanced Drug Delivery Reviews. 2020;159:377–90.
Thaxton CS, Rink JS, Naha PC, Cormode DP. Lipoproteins and lipoprotein mimetics for imaging and drug delivery. Advanced Drug Delivery Reviews. 2016;106:116–31.
Trevaskis NL, Kaminskas LM, Porter CJH. From sewer to saviour - targeting the lymphatic system to promote drug exposure and activity. Nature Reviews Drug Discovery. 2015;14(11):781–803.
Tabas I, Williams KJ, Boren J. Subendothelial lipoprotein retention as the initiating process in atherosclerosis - Update and therapeutic implications. Circulation. 2007;116(16):1832–44.
Schwartz EA, Reaven PD. Lipolysis of triglyceride-rich lipoproteins, vascular inflammation, and atherosclerosis. Biochimica Et Biophysica Acta-Molecular and Cell Biology of Lipids. 2012;1821(5):858–66.
Vitols S, Peterson C, Larsson O, Holm P, Åberg B. Elevated Uptake of Low Density Lipoproteins by Human Lung Cancer Tissue in Vivo. Cancer Research. 1992;52(22):6244–7.
Masquelier M, Vitols S, Peterson C. Low-Density Lipoprotein as a Carrier of Antitumoral Drugs: In Vivo Fate of Drug-Human Low-Density Lipoprotein Complexes in Mice. Cancer Research. 1986;46(8):3842–7.
Samadi-Baboli M, Favre G, Canal P, Soula G. Low density lipoprotein for cytotoxic drug targeting: improved activity of elliptinium derivative against B16 melanoma in mice. British Journal of Cancer. 1993;68(2):319–26.
Radwan AA, Alanazi FK. Design and Synthesis of New Cholesterol-Conjugated 5-Fluorouracil: A Novel Potential Delivery System for Cancer Treatment. Molecules. 2014;19(9):13177–87.
Pussinen PJ, Lindner H, Glatter O, Reicher H, Kostner GM, Wintersperger A, Malle E, Sattler W. Lipoprotein-associated α-tocopheryl-succinate inhibits cell growth and induces apoptosis in human MCF-7 and HBL-100 breast cancer cells. Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids. 2000;1485(2):129–44.
Dahan A, Duvdevani R, Shapiro I, Elmann A, Finkelstein E, Hoffman A. The oral absorption of phospholipid prodrugs: In vivo and in vitro mechanistic investigation of trafficking of a lecithin-valproic acid conjugate following oral administration. Journal of Controlled Release. 2008;126(1):1–9.
Tu B, Bazan NG. Hippocampal kindling epilepto,genesis upregulates neuronal cyclooxygenase-2 expression in neocortex. Experimental Neurology. 2003;179(2):167–75.
Mula M. Emerging drugs for focal epilepsy. Expert Opinion on Emerging Drugs. 2018;23(3):243–9.
Li Y, Zhan HZ, Fan YX, Zhang J, Cao GY, Yu JC, Chen YC, Guo BN. Determination of DP-VPA and its active metabolite, VPA, in human plasma, urine, and feces by UPLC-MS/MS: A clinical pharmacokinetics and excretion study. Drug Test Anal. 2019;11(7):1035–47.
Tauchi Y, Zushida I, Chono S, Sato J, Ito K, Morimoto K. Effect of dexamethasone palmitate-low density lipoprotein complex on cholesterol ester accumulation in aorta of atherogenic model mice. Biological & Pharmaceutical Bulletin. 2001;24(8):925–9.
Mansbach CM, Siddiqi SA. The Biogenesis of Chylomicrons. Annual Review of Physiology. 2010;72:315–33.
Borkar N, Li B, Holm R, Håkansson AE, Müllertz A, Yang M, Mu H. Lipophilic prodrugs of apomorphine I: Preparation, characterisation, and in vitro enzymatic hydrolysis in biorelevant media. European Journal of Pharmaceutics and Biopharmaceutics. 2015;89:216–23.
Bala V, Rao S, Li P, Wang S, Prestidge CA. Lipophilic Prodrugs of SN38: Synthesis and in Vitro Characterization toward Oral Chemotherapy. Mol Pharm. 2016;13(1):287–94.
Qin C, Chu Y, Feng W, Fromont C, He S, Ali J, Lee JB, Zgair A, Berton M, Bettonte S, Liu R, Yang L, Monmaturapoj T, Medrano-Padial C, Ugalde AAR, Vetrugno D, Ee SY, Sheriston C, Wu Y, et al. Targeted delivery of lopinavir to HIV reservoirs in the mesenteric lymphatic system by lipophilic ester prodrug approach. Journal of Controlled Release. 2021;329:1077–89.
Lee JB, Zgair A, Malec J, Kim TH, Kim MG, Ali J, Qin CL, Feng WS, Chiang MT, Gao XZ, Voronin G, Garces AE, Lau CL, Chan TH, Hume A, McIntosh TM, Soukarieh F, Al-Hayali M, Cipolla E, et al. Lipophilic activated ester prodrug approach for drug delivery to the intestinal lymphatic system. Journal of Controlled Release. 2018;286:10–9.
Zgair A, Dawood Y, Ibrahem SM, Back HM, Kagan L, Gershkovich P, Lee JB. Predicting Intestinal and Hepatic First-Pass Metabolism of Orally Administered Testosterone Undecanoate. Applied Sciences-Basel. 2020;10(20).
Tauber U, Schroder K, Dusterberg B, Matthes H. Absolute bioavailability of testosterone after oral administration of testosterone-undecanoate and testosterone. Eur J Drug Metabol Pharmacokinet. 1986;11(2):145–9.
Shackleford DM, Faassen WA, Houwing N, Lass H, Edwards GA, Porter CJH, Charman WN. Contribution of lymphatically transported testosterone undecanoate to the systemic exposure of testosterone after oral administration of two andriol formulations in conscious lymph duct-cannulated dogs. Journal of Pharmacology and Experimental Therapeutics. 2003;306(3):925–33.
Kohn FM, Schill WB. A new oral testosterone undecanoate formulation. World Journal of Urology. 2003;21(5):311–5.
Gershkovich P, Hoffman A. Effect of a high-fat meal on absorption and disposition of lipophilic compounds: The importance of degree of association with triglyceride-rich lipoproteins. European Journal of Pharmaceutical Sciences. 2007;32(1):24–32.
Han S, Hu L, Quach T, Simpson JS, Trevaskis NL, Porter CJH. Constitutive Triglyceride Turn-Over into the Mesenteric Lymph is Unable to Support Efficient Lymphatic Transport of a Bio-mimetic Triglyceride Prodrug. Journal of Pharmaceutical Sciences. 2016;105:786–96.
Hu L, Quach T, Han S, Lim SF, Yadav P, Senyschyn D, Trevaskis NL, Simpson JS, Porter CJ. Glyceride-Mimetic Prodrugs Incorporating Self-Immolative Spacers Promote Lymphatic Transport, Avoid First-Pass Metabolism, and Enhance Oral Bioavailability. Angew Chem Int Ed. 2016;55:13700–5.
Scriba GKE. Synthesis and in-vitro degradation of testosterone-lipid conjugates. Archiv Der Pharmazie. 1995;328(3):271–6.
Kochappan R, Cao E, Han S, Hu L, Quach T, Senyschyn D, Ferreira VI, Lee G, Leong N, Sharma G, Lim SF, Nowell CJ, Chen Z, von Andrian UH, Bonner D, Mintern JD, Simpson JS, Trevaskis NL, Porter CJH. Targeted delivery of mycophenolic acid to the mesenteric lymph node using a triglyceride mimetic prodrug approach enhances gut-specific immunomodulation in mice. Journal of Controlled Release. 2021;332:636–51.
Lalanne M, Paci A, Andrieux K, Dereuddre-Bosquet N, Clayette P, Deroussent A, Re M, Vassal G, Couvreur P, Desmaele D. Synthesis and biological evaluation of two glycerolipidic prodrugs of didanosine for direct lymphatic delivery against HIV. Bioorganic & Medicinal Chemistry Letters. 2007;17(8):2237–40.
Han S, Quach T, Hu L, Wahab A, Charman WN, Stella VJ, Trevaskis NL, Simpson JS, Porter CJH. Targeted delivery of a model immunomodulator to the lymphatic system: Comparison of alkyl ester versus triglyceride mimetic lipid prodrug strategies. Journal of Controlled Release. 2014;177:1–10.
Han SF, Hu LJ. Gracia, Quach T, Simpson JS, Edwards GA, Trevaskis NL, Porter CJH. Lymphatic Transport and Lymphocyte Targeting of a Triglyceride Mimetic Prodrug Is Enhanced in a Large Animal Model: Studies in Greyhound Dogs. Mol Pharm. 2016;13(10):3351–61.
Sun B, Luo C, Cui W, Sun J, He Z. Chemotherapy agent-unsaturated fatty acid prodrugs and prodrug-nanoplatforms for cancer chemotherapy. J Control Release. 2017;264:145–59.
Caliph SM, Cao E, Bulitta JB, Hu L, Han S, Porter CJH, Trevaskis NL. The impact of lymphatic transport on the systemic disposition of lipophilic drugs. Journal of Pharmaceutical Sciences. 2013;102(7):2395–408.
Van Eck M, Oost J, Goudriaan JR, Hoekstra M, Hildebrand RB, Bos IST, van Dijk KW, Van Berkel TJC. Role of the macrophage very-low-density lipoprotein receptor in atherosclerotic lesion development. Atherosclerosis. 2005;183(2):230–7.
Mead JR, Ramji DP. The pivotal role of lipoprotein lipase in atherosclerosis. Cardiovascular Research. 2002;55(2):261–9.
Kuemmerle NB, Rysman E, Lombardo PS, Flanagan AJ, Lipe BC, Wells WA, Pettus JR, Froehlich HM, Memoli VA, Morganelli PM, Swinnen JV, Timmerman LA, Chaychi L, Fricano CJ, Eisenberg BL, Coleman WB, Kinlaw WB. Lipoprotein Lipase Links Dietary Fat to Solid Tumor Cell Proliferation. Molecular Cancer Therapeutics. 2011;10(3):427–36.
Liu X, Hartman CL, Li L, Albert CJ, Si F, Gao A, Huang L, Zhao Y, Lin W, Hsueh EC, Shen L, Shao Q, Hoft DF, Ford DA, Peng G. Reprogramming lipid metabolism prevents effector T cell senescence and enhances tumor immunotherapy. Science Translational Medicine. 2021;13(587):eaaz6314.
Dahan A, Markovic M, Epstein S, Cohen N, Zimmermann EM, Aponick A, Ben-Shabat S. Phospholipid-drug conjugates as a novel oral drug targeting approach for the treatment of inflammatory bowel disease. European Journal of Pharmaceutical Sciences. 2017;108:78–85.
Radwan AA, Alanazi FK. Targeting cancer using cholesterol conjugates. Saudi Pharmaceutical Journal. 2014;22(1):3–16.
Markovic M, Ben-Shabat S, Keinan S, Aponick A, Zimmermann EM, Dahan A. Prospects and Challenges of Phospholipid-Based Prodrugs. Pharmaceutics. 2018;10(4).
Dahan A, Markovic M, Aponick A, Zimmermann EM, Ben-Shabat S. The prospects of lipidic prodrugs: an old approach with an emerging future. Future Medicinal Chemistry. 2019;11(19):2563–71.
Shi JJ, Kantoff PW, Wooster R, Farokhzad OC. Cancer nanomedicine: progress, challenges and opportunities. Nature Reviews Cancer. 2017;17(1):20–37.
Mitchell MJ, Billingsley MM, Haley RM, Wechsler ME, Peppas NA, Langer R. Engineering precision nanoparticles for drug delivery. Nat Rev Drug Discov. 2021;20(2):101–24.
Mura S, Bui DT, Couvreur P, Nicolas J. Lipid prodrug nanocarriers in cancer therapy. J Control Release. 2015;208:25–41.
Fattahi N, Shahbazi MA, Maleki A, Hamidi M, Ramazani A, Santos HA. Emerging insights on drug delivery by fatty acid mediated synthesis of lipophilic prodrugs as novel nanomedicines. Journal of Controlled Release. 2020;326:556–98.
Tretiakova DS, Khaidukov SV, Babayants AA, Frolova IS, Shcheglovitova ON, Onishchenko NR, Vodovozova EL. Lipophilic Prodrug of Methotrexate in the Membrane of Liposomes Promotes Their Uptake by Human Blood Phagocytes. Acta Naturae. 2020;12(1):99–109.
Xing J, Zhang XQ, Wang Z, Zhang HQ, Chen P, Zhou GX, Sun CL, Gu N, Ji M. Novel lipophilic SN38 prodrug forming stable liposomes for colorectal carcinoma therapy. International Journal of Nanomedicine. 2019;14:5201–13.
Tretiakova D, Onishchenko N, Boldyrev I, Mikhalyov I, Tuzikov A, Bovin N, Evtushenko E, Vodovozova E. Influence of stabilizing components on the integrity of antitumor liposomes loaded with lipophilic prodrug in the bilayer. Colloids and Surfaces B-Biointerfaces. 2018;166:45–53.
Lee J, Goh U, Lee HJ, Kim J, Jeong M, Park JH. Effective Retinal Penetration of Lipophilic and Lipid-Conjugated Hydrophilic Agents Delivered by Engineered Liposomes. Molecular Pharmaceutics. 2017;14(2):423–30.
Mikhalin AA, Evdokimov NM, Frolova LV, Magedov IV, Kornienko A, Johnston R, Rogelj S, Tartis MS. Lipophilic prodrug conjugates allow facile and rapid synthesis of high-loading capacity liposomes without the need for post-assembly purification. Journal of Liposome Research. 2015;25(3):232–60.
Gabizon A, Shmeeda H, Tahover E, Kornev G, Patil Y, Amitay Y, Ohana P, Sapir E, Zalipsky S. Development of Promitil(R), a lipidic prodrug of mitomycin c in PEGylated liposomes: From bench to bedside. Adv Drug Deliv Rev. 2020;154-155:13-26.
Dragovich T, Mendelson D, Kurtin S, Richardson K, Von Hoff D, Hoos A. A Phase 2 trial of the liposomal DACH platinum L-NDDP in patients with therapy-refractory advanced colorectal cancer. Cancer Chemother Pharmacol. 2006;58(6):759–64.
PerezSoler R, Shin DM, Siddik ZH, Murphy WK, Huber M, Lee JS, Khokhar AR, Hong WK. Phase I clinical and pharmacological study of liposome-entrapped NDDP administered intrapleurally in patients with malignant pleural effusions. Clinical Cancer Research. 1997;3(3):373–9.
Arouri A, Mouritsen OG. Anticancer double lipid prodrugs: liposomal preparation and characterization. J Liposome Res. 2011;21(4):296–305.
Signorell RD, Luciani P, Brambilla D, Leroux JC. Pharmacokinetics of lipid-drug conjugates loaded into liposomes. Eur J Pharm Biopharm. 2018;128:188–99.
Nakamura T, Harashima H. Dawn of lipid nanoparticles in lymph node targeting: Potential in cancer immunotherapy. Adv Drug Deliv Rev. 2020;167:78–88.
Swaminathan G, Thoryk EA, Cox KS, Meschino S, Dubey SA, Vora KA, Celano R, Gindy M, Casimiro DR, Bett AJ. A novel lipid nanoparticle adjuvant significantly enhances B cell and T cell responses to sub-unit vaccine antigens. Vaccine. 2016;34(1):110–9.
Tenchov R, Bird R, Curtze AE, Zhou Q. Lipid Nanoparticles—From Liposomes to mRNA Vaccine Delivery, a Landscape of Research Diversity and Advancement. ACS Nano. 2021;In press, https://doi.org/10.1021/acsnano.1c04996.
Pilkington EH, Suys EJA, Trevaskis NL, Wheatley AK, Zukancic D, Algarni A, Al-Wassiti H, Davis TP, Pouton CW, Kent SJ, Truong NP. From influenza to COVID-19: Lipid nanoparticle mRNA vaccines at the frontiers of infectious diseases. Acta Biomater. 2021. In press, https://doi.org/10.1016/j.actbio.2021.06.023
Pedersbaek D, Simonsen JB. A systematic review of the biodistribution of biomimetic high-density lipoproteins in mice. J Control Release. 2020;328:792–804.
Di L, Maiseyeu A. Low-density lipoprotein nanomedicines: mechanisms of targeting, biology, and theranostic potential. Drug Deliv. 2021;28(1):408–21.
Duivenvoorden R, Tang J, Cormode DP, Mieszawska AJ, Izquierdo-Garcia D, Ozcan C, Otten MJ, Zaidi N, Lobatto ME, van Rijs SM, Priem B, Kuan EL, Martel C, Hewing B, Sager H, Nahrendorf M, Randolph GJ, Stroes ESG, Fuster V, et al. A statin-loaded reconstituted high-density lipoprotein nanoparticle inhibits atherosclerotic plaque inflammation. Nat Commun. 2014;5:3065–5.
Duivenvoorden R, Tang J, Cormode DP, Mieszawska AJ, Izquierdo-Garcia D, Ozcan C, Otten MJ, Zaidi N, Lobatto ME, van Rijs SM, Priem B, Kuan EL, Martel C, Hewing B, Sager H, Nahrendorf M, Randolph GJ, Stroes ESG, Fuster V, et al. A statin-loaded reconstituted high-density lipoprotein nanoparticle inhibits atherosclerotic plaque inflammation. Nat Commun. 2014;5:3065.
Rajora MA, Ding L, Valic M, Jiang W, Overchuk M, Chen J, Zheng G. Tailored theranostic apolipoprotein E3 porphyrin-lipid nanoparticles target glioblastoma. Chem Sci. 2017;8(8):5371–84.
Wang H, Xie H, Wu J, Wei X, Zhou L, Xu X, Zheng S. Structure-based rational design of prodrugs to enable their combination with polymeric nanoparticle delivery platforms for enhanced antitumor efficacy. Angew Chem Int Ed Engl. 2014;53(43):11532–7.
Parshad B, Prasad S, Bhatia S, Mittal A, Pan Y, Mishra PK, Sharma SK, Fruk L. Non-ionic small amphiphile based nanostructures for biomedical applications. RSC Advances. 2020;10(69):42098–115.
Maksimenko A, Dosio F, Mougin J, Ferrero A, Wack S, Reddy LH, Weyn AA, Lepeltier E, Bourgaux C, Stella B, Cattel L, Couvreur P. A unique squalenoylated and nonpegylated doxorubicin nanomedicine with systemic long-circulating properties and anticancer activity. Proc Natl Acad Sci U S A. 2014;111(2):E217–26.
Couvreur P, Reddy LH, Mangenot S, Poupaert JH, Desmaele D, Lepetre-Mouelhi S, Pili B, Bourgaux C, Amenitsch H, Ollivon M. Discovery of new hexagonal supramolecular nanostructures formed by squalenoylation of an anticancer nucleoside analogue. Small. 2008;4(2):247–53.
Sobot D, Mura S, Yesylevskyy SO, Dalbin L, Cayre F, Bort G, Mougin J, Desmaele D, Lepetre-Mouelhi S, Pieters G, Andreiuk B, Klymchenko AS, Paul JL, Ramseyer C, Couvreur P. Conjugation of squalene to gemcitabine as unique approach exploiting endogenous lipoproteins for drug delivery. Nat Commun. 2017;8:9.
Mura S, Zouhiri F, Lerondel S, Maksimenko A, Mougin J, Gueutin C, Brambilla D, Caron J, Sliwinski E, LePape A, Desmaele D, Couvreur P. Novel Isoprenoyl Nanoassembled Prodrug for Paclitaxel Delivery. Bioconjugate Chemistry. 2013;24(11):1840–9.
Dosio F, Reddy LH, Ferrero A, Stella B, Cattel L, Couvreur P. Novel Nanoassemblies Composed of Squalenoyl−Paclitaxel Derivatives: Synthesis, Characterization, and Biological Evaluation. Bioconjugate Chemistry. 2010;21(7):1349–61.
Wang H, Xie H, Wang J, Wu J, Ma X, Li L, Wei X, Ling Q, Song P, Zhou L, Xu X, Zheng S. Self-Assembling Prodrugs by Precise Programming of Molecular Structures that Contribute Distinct Stability, Pharmacokinetics, and Antitumor Efficacy. Advanced Functional Materials. 2015;25(31):4956–65.
Zuo J, Tong L, Du L, Yang M, Jin Y. Biomimetic nanoassemblies of 1-O-octodecyl-2-conjugated linoleoyl-sn-glycero-3-phosphatidyl gemcitabine with phospholipase A2-triggered degradation for the treatment of cancer. Colloids and Surfaces B: Biointerfaces. 2017;152:467–74.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Guest Editor: Yijun Pan
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Han, S., Mei, L., Quach, T. et al. Lipophilic Conjugates of Drugs: A Tool to Improve Drug Pharmacokinetic and Therapeutic Profiles. Pharm Res 38, 1497–1518 (2021). https://doi.org/10.1007/s11095-021-03093-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-021-03093-x