Abstract
Background
Adjuvant oral uracil-tegafur (UFT) has led to significantly longer postoperative survival among patients with non-small-cell lung cancer (NSCLC). Gemcitabine (GEM) monotherapy is also reportedly effective for NSCLC and has minor adverse events (AEs). This study compared the efficacy of GEM- versus UFT-based adjuvant regimens in patients with completely resected pathological stage (p-stage) IB–IIIA NSCLC.
Patients and methods
Patients with completely resected p-stage IB–IIIA NSCLC were randomly assigned to GEM or UFT. The primary endpoint was overall survival (OS); secondary endpoints were disease-free survival (DFS), and AEs.
Results
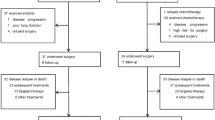
We assigned 305 patients to the GEM group and 303 to the UFT group. Baseline factors were balanced between the arms. Of the 608 patients, 293 (48.1%) had p-stage IB disease, 195 (32.0%) had p-stage II disease and 121 (19.9%) had p-stage IIIA disease. AEs were generally mild in both groups, and only one death occurred, in the GEM group. After a median follow-up of 6.8 years, the two groups did not significantly differ in survival: 5 year OS rates were GEM: 70.0%, UFT: 68.8% (hazard ratio 0.948; 95% confidence interval 0.73–1.23; P = 0.69).
Conclusion
Although GEM-based adjuvant therapy for patients with completely resected stage IB–IIIA NSCLC was associated with acceptable toxicity, it did not provide longer OS than did UFT.




Similar content being viewed by others
References
Non-small Cell Lung Cancer Collaborative Group (1995) Chemotherapy in non-small cell lung cancer: a meta-analysis using updated data on individual patients from 52 randomised clinical trials. BMJ 311(7010):899–909
Ohta M, Tsuchiya R, Shimoyama M et al (1993) Adjuvant chemotherapy for completely resected stage III non-small-cell lung cancer. Results of a randomized prospective study. The Japan Clinical Oncology Group. J Thorac Cardiovasc Surg 106(4):703–708
Tada H, Tsuchiya R, Ichinose Y et al (2004) A randomized trial comparing adjuvant chemotherapy versus surgery alone for completely resected pN2 non-small cell lung cancer (JCOG9304). Lung Cancer 43(2):167–173. https://doi.org/10.1016/j.lungcan.2003.08.028
Wada H, Hitomi S, Teramatsu T (1996) Adjuvant chemotherapy after complete resection in non-small-cell lung cancer. West Japan Study Group for Lung Cancer Surgery. J Clin Oncol 14(4):1048–1054. https://doi.org/10.1200/JCO.1996.14.4.1048
Kato H, Ichinose Y, Ohta M, Japan Lung Cancer Research Group on Postsurgical Adjuvant Chemotherapy et al (2004) A randomized trial of adjuvant chemotherapy with uracil-tegafur for adenocarcinoma of the lung. N Engl J Med 350(17):1713–1721. https://doi.org/10.1056/NEJMoa032792
Abratt RP, Bezwoda WR, Falkson G et al (1994) Efficacy and safety profile of gemcitabine in non-small-cell lung cancer: a phase II study. J Clin Oncol 12(8):1535–1540. https://doi.org/10.1200/JCO.1994.12.8.1535
Anderson H, Lund B, Bach F et al (1994) Single-agent activity of weekly gemcitabine in advanced non-small-cell lung cancer: a phase II study. J Clin Oncol 12(9):1821–1826. https://doi.org/10.1200/JCO.1994.12.9.1821
Gatzemeier U, Shepherd FA, Le Chevalier T et al (1996) Activity of gemcitabine in patients with non-small cell lung cancer: a multicentre, extended phase II study. Eur J Cancer 32A(2):243–248. https://doi.org/10.1016/0959-8049(95)00444-0
Vansteenkiste JF, Vandebroek JE, Nackaerts KL et al (2001) Clinical-benefit response in advanced non-small-cell lung cancer: a multicentre prospective randomised phase III study of single agent gemcitabine versus cisplatin-vindesine. Ann Oncol 12(9):1221–1230. https://doi.org/10.1023/a:1012208711013
Arriagada R, Bergman B, Dunant A et al (2004) Cisplatin-based adjuvant chemotherapy in patients with completely resected non-small-cell lung cancer. N Engl J Med 350(4):351–360. https://doi.org/10.1056/NEJMoa031644
Douillard JY, Rosell R, De Lena M et al (2006) Adjuvant vinorelbine plus cisplatin versus observation in patients with completely resected stage IB-IIIA non-small-cell lung cancer (Adjuvant Navelbine International Trialist Association [ANITA]): a randomised controlled trial. Lancet Oncol 7(9):719–727. https://doi.org/10.1016/S1470-2045(06)70804-X
Scagliotti GV, Fossati R, Torri V, Adjuvant Lung Project Italy/European Organisation for Research Treatment of Cancer-Lung Cancer Cooperative Group I et al (2003) Randomized study of adjuvant chemotherapy for completely resected stage I, II, or IIIA non-small-cell Lung cancer. J Natl Cancer Inst 95(19):1453–1461. https://doi.org/10.1093/jnci/djg059
Winton T, Livingston R, Johnson D, National Cancer Institute of Canada Clinical Trials G, National Cancer Institute of the United States Intergroup JBRTI et al (2005) Vinorelbine plus cisplatin vs. observation in resected non-small-cell lung cancer. N Engl J Med 352(25):2589–2597. https://doi.org/10.1056/NEJMoa043623
Pignon JP, Tribodet H, Scagliotti GV, LACE Collaborative Group et al (2008) Lung adjuvant cisplatin evaluation: a pooled analysis by the LACE Collaborative Group. J Clin Oncol 26(21):3552–3559. https://doi.org/10.1200/JCO.2007.13.9030
Toyooka S, Okumura N, Nakamura H et al (2018) A multicenter randomized controlled study of paclitaxel plus carboplatin versus oral uracil-tegafur as the adjuvant chemotherapy in resected non-small cell lung cancer. J Thorac Oncol 13(5):699–706. https://doi.org/10.1016/j.jtho.2018.02.015
Gridelli C, Perrone F, Gallo C, MILES Investigators et al (2003) Chemotherapy for elderly patients with advanced non-small-cell lung cancer: the multicenter Italian lung cancer in the elderly study (MILES) phase III randomized trial. J Natl Cancer Inst 95(5):362–372. https://doi.org/10.1093/jnci/95.5.362
Yamaguchi M, Takeo S, Suemitsu R et al (2010) Feasibility study for biweekly administration of cisplatin plus gemcitabine as adjuvant-chemotherapy for completely resected non-small cell lung cancer. Cancer Chemother Pharmacol 66(1):107–112. https://doi.org/10.1007/s00280-009-1139-x
Kunitoh H, Tsuboi M, Wakabayashi M, on behalf of the Japan Clinical Oncology Group Lung Cancer Surgical Study Group (JCOG-LCSSG) et al (2020) A phase III study of adjuvant chemotherapy in patients with completely resected, node-negative non–small cell lung cancer (JCOG 0707). JTCVS Open 4:90–102
Acknowledgements
We express our sincere gratitude and respect to the late Dr. Yukito Ichinose (1953-2018), National Hospital Organization Kyushu Cancer Center, Fukuoka, Japan who continuously supported this clinical trial and the West Japan Thoracic Oncology Group (WJTOG).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Yamaguchi, M., Tada, H., Mitsudomi, T. et al. Phase III study of adjuvant gemcitabine compared with adjuvant uracil-tegafur in patients with completely resected pathological stage IB–IIIA non-small cell lung cancer (WJTOG0101). Int J Clin Oncol 26, 2216–2223 (2021). https://doi.org/10.1007/s10147-021-02012-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-021-02012-9