Abstract
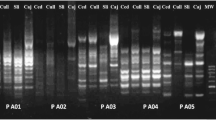
Artemisia anuua L. is an important economic medicinal plant well known for its bioactive compound artemisinin that is used as an antimalarial drug. Extraction of high-quality genomic DNA suitable for restriction enzyme reactions and PCR amplifications is very difficult due to the presence of high amounts of secondary metabolites in A. anuua. This study compared efficiency of Fermentas genomic DNA purification kit (FGDK), phenol–Chloroform, cetyltrimethyl ammonium bromide (CTAB), sodium dodecyl sulfate and Sarkosyl nitrogen methods for extraction of DNA. The quality and quantity DNA samples were determined by physical appearance, agarose gel electrophoresis, spectrophotometer and PCR amplifications. The study used Inter retrotransposon amplified polymorphism (IRAP), retrotransposon-microsatellite amplified polymorphism (REMAP) and start codon targeted-based primers. The results revealed that genomic DNA isolated by CTAB method was comparatively pure and produced clear polymorphic bands compared to the polymorphic bands noted on DNA extractions using other protocols.




Similar content being viewed by others
References
Aboul-Maaty NAF, Oraby HAS (2019) Extraction of high-quality genomic DNA from different plant orders applying a modified CTAB-based method. Bull Natl Res Cent 43:25
Agrawal A, Sharma A, Shukla NP (2016) Genomic DNA extraction protocol for Artemisia annua L. without using liquid nitrogen and phenol. Int J Appl Sci Biotechnol 4:448–451
Ali Q, Salisu IB, Raza A, Shahid AA, Roa AQ, Husnain T (2019) A modified protocol for rapid DNA isolation from cotton (Gossypium spp.). MethodsX 6:259–264
Arruda SR, Pereira DG, Silva-Castro MM, Brito MG, Waldschmidt AM (2017) An optimized protocol for DNA extraction in plants with a high content of secondary metabolites, based on leaves of Mimosa tenuiflora (Willd.) Poir. (Leguminosae). Genet Mol Res. https://doi.org/10.4238/gmr16039063
Bora KS, Sharma A (2011) The genus Artemisia: a comprehensive review. Pharm Biol 49(1):101–109
Chen S, Yu H, Luo HM, Wu Q, Li CF, Steinmetz A (2016) Conservation and sustainable use of medicinal plants: problems, progress, and prospects. Chin Med. https://doi.org/10.1186/s13020-016-0108-7
Croy EJ, Ikemura T, Shirsat A, Croy RRD (1993) Plant nucleic acids. Plant Molecular Biology Bios Scientific Publishers Blackwell Scientific Publications UK.
Dellaporta SL, Wood J, Hicks JB (1983) A plant DNA mini preparation: version II. Plant Mol Biol Rep 1:19–21
Devi KD, Punyarani K, Singh NS, Devi HS (2013) An efficient protocol for total DNA extraction from the members of order Zingiberales- suitable for diverse PCR based downstream applications. Springerplus 2:669
Doyle JJ, Doyle JL (1987) A rapid procedure for DNA purification from small quantities of fresh leaf tissue. Phytochem Bull 19(1):11–15
Ghaffariyan S, Mohammadi SA, Aharizad S (2012) DNA isolation protocol for the medicinal plant lemon balm (Melissa officinalis, Lamiaceae). Genet Mol Res 11:1049–1057
Hasan SMZ, Shafie MSB, Shah RM (2008) Efficient method for the extraction of genomic DNA from wormwood (Artemisia capllaris). Afr J Biotechnol 7:3211–3216
Hasan SMZ, Shafie MSB, Shah RM (2009) Genomic DNA extraction methods from wormwood capillary (Artemisia capillaris) for PCR-RAPD studies. NewYork Sci J 2:13–21
Healey A, Furtado A, Cooper T, Henry RJ (2014) Protocol: a simple method for extracting next-generation sequencing quality genomic DNA from recalcitrant plant species. Plant Methods 10:21. https://doi.org/10.1186/1746-4811-10-21
Ibtissam O, Mohamed G, Lahsen ELG, Benaissa K, Eddine BYS (2011) Genomic DNA extraction protocols from a Moroccan medicinal and aromatic plant Artemisia herba-alba Asso for RAPD-PCR studies. Int J Biotechnol Mol Biol Res 2:131–137
Kalbande BB, Patil AS, Chakrabarty PK (2016) An efficient, simple and high throughput protocol for cotton genomic DNA isolation. J Plant Biochem Biotechnol 25:437–441
Katterman H, Shattuck L (1983) An effective method of DNA isolation from the mature leaves of Gossypium species that contain large amounts of phenolic terpenoids and tannins. Prep Biochem 13:347–359
Kumar J, Bajaj P, Mishra GP, Singh SHB, Singh H, Naik PK (2014) Utilization of EST-derived SSRs in the genetic characterization of Artemisia Annua L. genotypes from Ladakh India. Indian J Biotechnol 13:464–472
Lade BD, Patil AS, Paikrao HM (2014) Efficient genomic DNA extraction protocol from medicinal rich Passiflora foetida containing high level of polysaccharide and polyphenol. Springerplus 3:457
Lucas MS, Carvalho CDS, Hypolito GB, Côrtes MC (2019) Optimized protocol to isolate high quality genomic DNA from different tissues of a palm species. Hoehnea 46(2):e942018
Maghni B, Bougoutaia Y, Abderrabi K, Adda A, Merah O (2016) Optimization of DNA extraction and PCR conditions for genetic diversity study in Artemisia Herba-Alba from Algeria. Sci J Genet Gene Therap 2:010–012
Michiels A, Van den Ende W, Tucker M, Van Riet L, VanLaere A (2003) Extraction of high-quality genomic DNA from latex-containing plants. Anal Biochem 315(1):85–89
Paul S, Khanuja SPS, Gupta M (2014) Breeding strategy for genetic improvement up to four generations in relation to artemisinin with canopy and other secondary metabolites in Artemisia annua L. Ind Crop Prod 56:67–73
Pirttila AM, Hirsikorpi M, Kamarainen T, Jaakola L, Hohtola A (2001) DNA isolation methods for medicinal and aromatic plants. Plant Mol Biol Rep 19(3):273–273
Ribeiro RA, Lovato MB (2007) Comparative analysis of different DNA extraction protocols in fresh and herbarium specimens of the genus Dalbergia. Genet Mol Res 6(1):173–187
Sahu SK, Thangaraj M, Kathiresan K (2012) DNA extraction protocol for plants with high levels of secondary metabolites and polysaccharides without using liquid nitrogen and phenol. ISRN Mol Biol. https://doi.org/10.5402/2012/205049
Sambrook J, Fritsch EF, Maniatis T (1989) Analysis and cloning of eukaryotic genomic DNA. In: Molecular cloning: a laboratory manual, 2nd ed. Cold Spring Harbor Laboratory Press, Cold Spring Harbor (NY)
Sarwat M, Nabi G, Das S, Srivastava PS (2012) Molecular markers in medicinal plant biotechnology: past and present. Crit Rev Biotechn 32(1):74–92. https://doi.org/10.3109/07388551.2011.551872
Sarwat M, Negi MS, Lakshmikumaran M, Tyagi AK, Das S, Srivastava PS (2006) A standardized protocol for genomic DNA isolation from Terminalia arjuna for genetic diversity analysis. Electron J Biotechnol 9(1):86–91
Sika KC, Kefela T, Adoukonou-Sagbadja H, Ahotone L, Saidou A, Baba-Moussa L, Baptiste LJ, Kotconi SO, Gachomoa EW (2015) A simple and efficient genomic DNA extraction protocol for large scale genetic analyses of plant biological systems. Plant Gene 1:43–45
Tel-zur N, Abbo S, Myslabodski D, Mizrahi Y (1999) Modified CTAB procedure for DNA isolation from epiphytic Cacti of the genera Hylocereus and Selenicereus (Cactaceae). Plant Mol Biol Rep 17(3):249–254
Xin Z, Chen J (2012) A high throughput DNA extraction method with high yield and quality. Plant Methods 8:26
Acknowledgements
The authors gratefully acknowledge the laboratory facilities provided by Genomics & Molecular Breeding Lab, Center of Excellence in Cereal Molecular Breeding, University of Tabriz, Iran. The research was financially supported by the University of Tabriz in co-operation with the Iran National Elites Foundation, under vote No. 102-1565.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Valizadeh, N., Holasou, H.A., Mohammadi, S.A. et al. A Comparison of Genomic DNA Extraction Protocols in Artemisia annua L. for Large Scale Genetic Analyses Studies. Iran J Sci Technol Trans Sci 45, 1587–1595 (2021). https://doi.org/10.1007/s40995-021-01170-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40995-021-01170-9